Anti-Human IL17A/IL17F Recombinant Antibody (TAB-H08)
CAT#: TAB-H08
Recombinant Humanized antibody to Human IL17A











Specifications
- Host Species
- Rat
- Derivation
- Humanized
- Type
- IgG1 - kappa
- Specificity
- IL17A (interleukin 17A, IL-17A) [Homo sapiens] ;
- Species Reactivity
- Human
- Applications
- ELISA, FC, IP, FuncS, IF, Neut, WB, Inhib, PK, Activ
- Related Disease
- Psoriasis
Product Property
- Purity
- >97%, by SDS-PAGE under reducing conditions and visualized by silver stain.
Applications
- Application Notes
- The IL17A antibody has been reported in applications of ELISA, FC, IP, FuncS, IF, Neut, WB, Inhib, PK, Activ.
Target
Customer Review
There are currently no Customer reviews or questions for TAB-H08. Click the button above to contact us or submit your feedback about this product.


Q&As
-
Does this antibody target only IL17A?
A: No, this antibody is specified to target both IL17A and IL17F. These are two closely related cytokines that are central to Th17 cell-mediated immune responses. This dual specificity makes it a useful reagent for research into inflammatory pathways where both cytokines may be present.
-
What is the host and type of TAB-H08?
A: This is a recombinant human antibody of the IgG1 isotype. It is produced in a mammalian CHO (Chinese Hamster Ovary) expression system. As a fully human antibody, it is a valuable tool for research in human systems, particularly for studying the IL17 signaling pathway.
View the frequently asked questions answered by Creative Biolabs Support.
Cite This Product
To accurately reference this product in your publication, please use the following citation information:
(Creative Biolabs Cat# TAB-H08, RRID: AB_3112023)
Submit Your Publication
Published with our product? Submit your paper and receive a 10% discount on your next order! Share your research to earn exclusive rewards.
Biosimilar Overview
Please refer to Bimekizumab Overview to learn more about the mechanism of action, clinical projects, and approved drugs of Bimekizumab.
Related Diseases
Downloadable Resources
Download resources about recombinant antibody development and antibody engineering to boost your research.
Product Notes
This is a product of Creative Biolabs' Hi-Affi™ recombinant antibody portfolio, which has several benefits including:
• Increased sensitivity
• Confirmed specificity
• High repeatability
• Excellent batch-to-batch consistency
• Sustainable supply
• Animal-free production
See more details about Hi-Affi™ recombinant antibody benefits.
Datasheet
MSDS
COA
Certificate of Analysis LookupTo download a Certificate of Analysis, please enter a lot number in the search box below. Note: Certificate of Analysis not available for kit components.
Protocol & Troubleshooting
We have outlined the assay protocols, covering reagents, solutions, procedures, and troubleshooting tips for common issues in order to better assist clients in conducting experiments with our products. View the full list of Protocol & Troubleshooting.
See other products for "IL17A"
Select a product category from the dropdown menu below to view related products.
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOB-1390z | Mouse Anti-IL17A Recombinant Antibody (clone 22B9) | FuncS | Mouse IgG2b |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-069 | Anti-Human IL17A Recombinant Antibody (TAB-069) | IF, IHC, ELISA, FC | IgG1 - kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-H57 | Anti-Human IL17A Recombinant Antibody (Perakizumab) | WB, ELISA, IP, FC, FuncS, Neut, IF | IgG1 - kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-399CL | Human Anti-IL17A Recombinant Antibody (TAB-399CL) | ELISA | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PABL-237 | Human Anti-IL17A Recombinant Antibody (clone CNTO7357) | WB, IHC, FuncS | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PABL-238 | Human Anti-IL17A Recombinant Antibody (clone Fab6785) | WB, ELISA, FuncS | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PSBL-237 | Human Anti-IL17A Recombinant Antibody (clone CNTO7357); scFv Fragment | WB, IHC, FuncS | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PSBL-238 | Human Anti-IL17A Recombinant Antibody (clone Fab6785); scFv Fragment | WB, ELISA, FuncS | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PFBL-237 | Human Anti-IL17A Recombinant Antibody (clone CNTO7357); Fab Fragment | WB, IHC, FuncS | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PFBL-238 | Human Anti-IL17A Recombinant Antibody (clone Fab6785); Fab Fragment | WB, ELISA, FuncS | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PABW-176 | Human Anti-IL17A Recombinant Antibody (PABW-176) | Neut, ELISA, WB | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PFBW-176 | Human Anti-IL17A Recombinant Antibody; Fab Fragment (PFBW-176) | Neut, ELISA, WB | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PSBW-176 | Human Anti-IL17A Recombinant Antibody; scFv Fragment (PSBW-176) | Neut, ELISA, WB | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-030ZJ | Mouse Anti-IL17A Recombinant Antibody (TAB-030ZJ) | ELISA, Neut | Mouse IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-048ZJ | Human Anti-IL17A Recombinant Antibody (TAB-048ZJ) | ELISA, Neut | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-078ZJ | Human Anti-IL17A Recombinant Antibody (TAB-078ZJ) | ELISA, Block, Neut | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-079ZJ | Human Anti-IL17A Recombinant Antibody (TAB-079ZJ) | ELISA, Block, Neut | Human IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-080ZJ | Human Anti-IL17A Recombinant Antibody (TAB-080ZJ) | ELISA, Block, Neut | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-030ZJ-S(P) | Mouse Anti-IL17A Recombinant Antibody; scFv Fragment (TAB-030ZJ-S(P)) | ELISA, Neut | Mouse scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-048ZJ-S(P) | Human Anti-IL17A Recombinant Antibody; scFv Fragment (TAB-048ZJ-S(P)) | ELISA, Neut | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-078ZJ-S(P) | Human Anti-IL17A Recombinant Antibody; scFv Fragment (TAB-078ZJ-S(P)) | ELISA, Block, Neut | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-030ZJ-F(E) | Mouse Anti-IL17A Recombinant Antibody; Fab Fragment (TAB-030ZJ-F(E)) | ELISA, Neut | Mouse Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-048ZJ-F(E) | Human Anti-IL17A Recombinant Antibody; Fab Fragment (TAB-048ZJ-F(E)) | ELISA, Neut | Humanized Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| Gly-077LC | Recombinant Anti-Human IL17A Antibody (Fc glycosylation) | ELISA | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOB-1196MZ | Recombinant Mouse Anti-Human IL17A Antibody (clone 2I7) | WB | Mouse antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| BRD-0289MZ | Chicken Anti-Interleukin-17a Polyclonal IgY | Indirect ELISA, WB | Chicken antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-1202CQ | Mouse Anti-IL17A Recombinant Antibody (NEUT-1202CQ) | Neut | Mouse IgG2a |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-1203CQ | Mouse Anti-IL17A Recombinant Antibody (NEUT-1203CQ) | Neut | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-1204CQ | Mouse Anti-IL17A Recombinant Antibody (clone 403D10.01) | Neut, FC | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-1205CQ | Mouse Anti-IL17A Recombinant Antibody (clone 408H6.01) | Neut, ELISA, FC | Mouse IgG2a |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-1206CQ | Mouse Anti-IL17A Recombinant Antibody (clone 409H7.05) | Neut, FC | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-458CQ | Anti-Human IL17A Recombinant Antibody (TAB-458CQ) | ELISA, IHC, FC, IP, IF, Inhib | IgG1, κ |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0061-YC-S(P) | Human Anti-IL17A Recombinant Antibody; scFv Fragment (HPAB-0061-YC-S(P)) | ELISA, Neut | Humanized scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0063-YC-S(P) | Human Anti-IL17A Recombinant Antibody (clone Antibody 7); scFv Fragment | FuncS | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0061-YC-F(E) | Human Anti-IL17A Recombinant Antibody; Fab Fragment (HPAB-0061-YC-F(E)) | ELISA, Neut | Humanized Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0063-YC-F(E) | Human Anti-IL17A Recombinant Antibody (clone Antibody 7); Fab Fragment | FuncS | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NS-042CN | Mouse Anti-IL17A Recombinant Antibody (NS-042CN) | ELISA, FC | Mouse IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NS-042CN-F(E) | Mouse Anti-IL17A Recombinant Antibody; Fab Fragment (NS-042CN-F(E)) | ELISA, FC | Mouse Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NS-042CN-S(P) | Mouse Anti-IL17A Recombinant Antibody; scFv Fragment (NS-042CN-S(P)) | ELISA, FC | Mouse scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-458CQ | Afuco™ Anti-IL17A ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-458CQ) | ELISA, IHC, FC, IP, IF, Inhib | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-069 | Afuco™ Anti-IL17A ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-069) | FC, IP, ELISA, Neut, FuncS, IF | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-H08 | Afuco™ Anti-IL17A ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-H08) | ELISA, FC, IP, FuncS, IF, Neut | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-H57 | Afuco™ Anti-IL17A ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-H57) | ELISA, IP, FC, FuncS, Neut | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0424-XY147 | AbPlus™ Anti-IL17A Magnetic Beads (41809) | IP, Protein Purification |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0225-XY130 | CytoStream™ Mouse Anti-IL17A Recombinant Antibody (VS-0225-XY130) | FC | Mouse IgG1, kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0325-FY96 | Human Anti-IL17A (clone Antibody 7) scFv-Fc Chimera | ELISA, Inhib | Human IgG1, scFv-Fc |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY3489 | Anti-IL17A Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY3490 | Anti-Human IL17A Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY3491 | Anti-Mouse IL17A Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0625-YC167 | Recombinant Anti-IL17A Eliminating Antibody, pH-Sensitive (VS-0625-YC167) | Antigen-Sweeping In Vivo. | Rat IgG1 kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0825-YC175 | SmartAb™ Recombinant Anti-IL17A pH-dependent Antibody (VS-0825-YC175) | IF, IHC, ELISA, FC | Human IgG1 kappa |
Popular Products

Application: Neut, ELISA, IF, IP, FuncS, FC, ICC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: ELISA, FC, IP, FuncS, IF, Neut, ICC

Application: IP, IF, FuncS, FC, Neut, ELISA, ICC

Application: ELISA, FC, IP, FuncS, IF, Neut, ICC

Application: IP, IF, FuncS, FC, Neut, ELISA, ICC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: FC, IP, ELISA, Neut, FuncS, IF, IHC

Application: ELISA, WB, BLI, SPR
For research use only. Not intended for any clinical use. No products from Creative Biolabs may be resold, modified for resale or used to manufacture commercial products without prior written approval from Creative Biolabs.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.














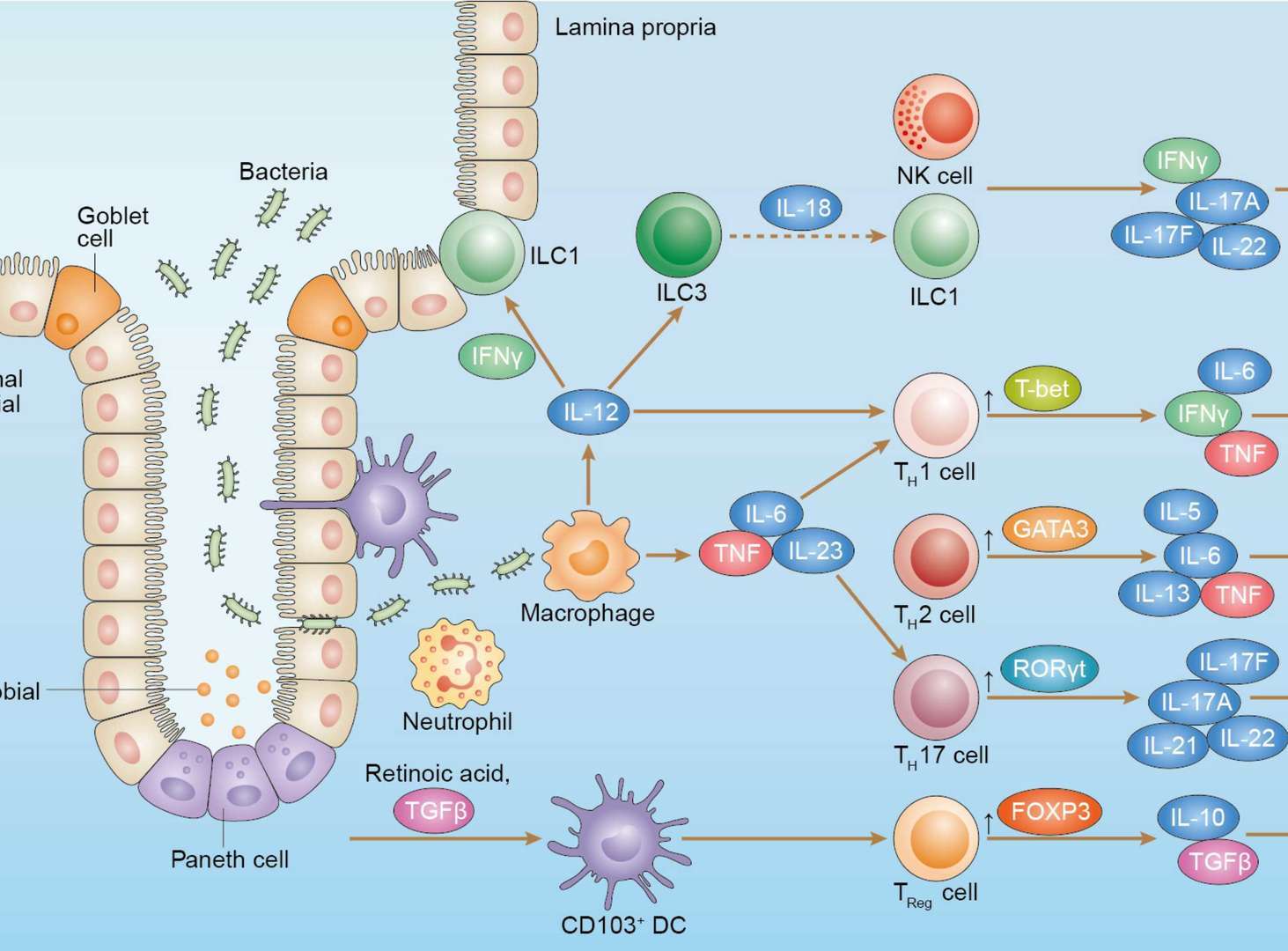 Inflammatory Bowel Diseases
Inflammatory Bowel Diseases













