Capromab Pendetide Overview
Introduction of Capromab Pendetide
Capromab is a Murine IgG1 monoclonal 7E11-C5.3 antibody that recognizes prostate specific membrane antigen (PSMA, also known as glutamate carboxypeptidase 2) from prostate cancer cells and normal prostate tissue. When linked to pendetide, capromab is used as a chelating agent for the radionuclide Indium In-111 during imaging to evaluate the extent of prostate cancer. Capromab pendetide also called ProstaScint® or CYT-365. A gamma camera (a special camera that detects radioactivity) could be used to find prostate cancer cells in the body by 111In-capromab pendetide. 111In-capromab pendetide was approved by the US Food and Drug Administration in 1996 for the detection of prostate carcinoma and soft tissue metastases in prostate cancer patients. The image performance of ProstaScint has been assessed in the pretherapy evaluation of patients with clinically localized prostate cancer who are considered at high risk for disease spread to regional nodes and in the post-therapy setting when occult residual or recurrent disease is suspected. Surgical series suggest the diagnostic accuracy in the 70% range. In a series of 255 men undergoing RP, Raj and associates405 reported that ProstaScint was able to differentiate between locally confined and metastatic disease, to detect soft tissue defects as small as 5 mm, and to have a sensitivity, specificity, and positive predictive value of 73%, 53%, and 89%, respectively. In the setting of a rising PSA value after prostatectomy, ProstaScint imaging has been used to evaluate patients for the presence of local or lymph node recurrence of disease after prostatectomy.
Mechanism of Action of Capromab Pendetide
Prostate cancer usually shows no symptoms in early stages, when it is most treatable. To detect the disease early, physicians usually recommend that every man 50 years and older have an annual examination consisting of a digital rectal examination and a prostate specific antigen (PSA) blood test. Conventional treatments are most successful for early stage prostate cancer but have limited effectiveness in advanced stages of the disease. For this reason, accurate staging of primary and recurrent prostate cancer is mandatory for proper therapeutic decisions. Prostate-specific membrane antigen (PSMA) is a unique type II, transmembrane-bound glycoprotein that is overexpressed on prostate tumor cells and in the neovasculature of most of the solid tumors, but not in the vasculature of normal tissues. This unique expression of PSMA makes it an important biomarker as well as a large extracellular target of imaging agents. Capromab, a monoclonal antibody against the cytoplasmic domain of PSMA, was found to be specific to prostate tumor tissues in humans when conjugated with glycyl-tyrosyl-(N-ε-diethylenetriamine pentacetic acid)-lysine (GYK-DTPA). Tumors often contain dying or necrotic cells with permeable membranes, allowing the binding of antibodies to the intracellular epitope of PSMA. The antibody conjugate, Capromab pendetide, was radiolabeled with 111Indium for the detection of prostate carcinoma and soft tissue metastases in prostate cancer patients.
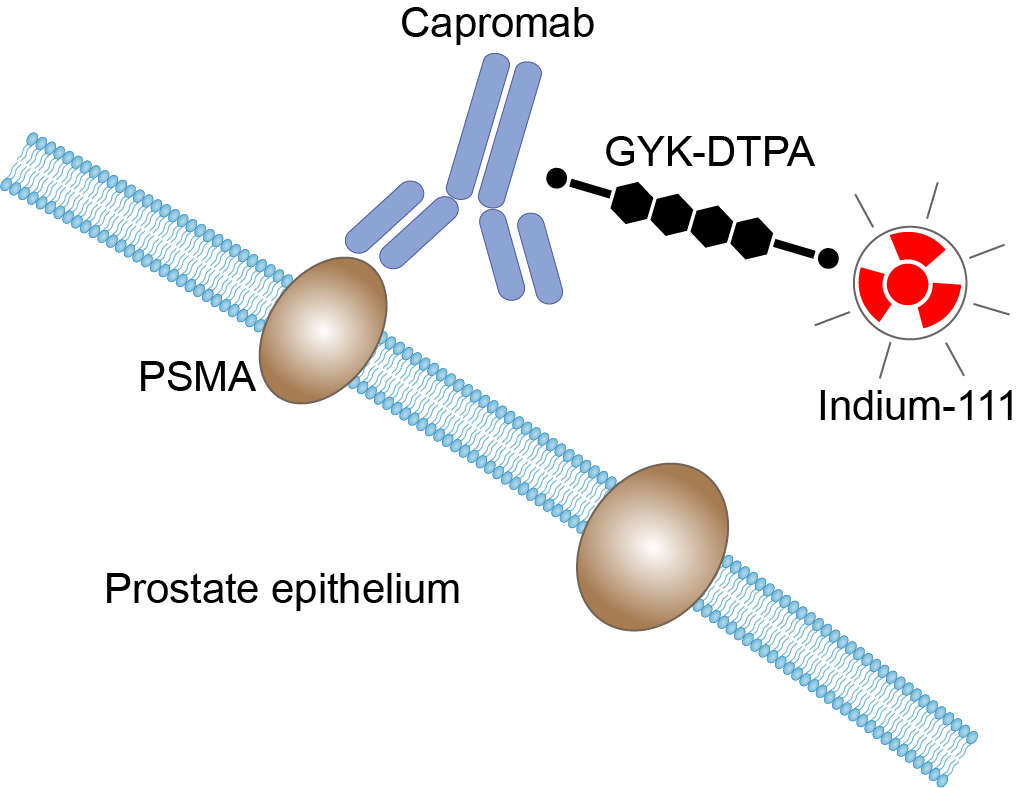 Fig 1. Mechanism of Action of Capromab Pendetide
Fig 1. Mechanism of Action of Capromab Pendetide
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

