Denosumab Overview
Introduction of Denosumab
Denosumab is a novel, fully human IgG2 monoclonal antibody specific to receptor activator of nuclear factor kappa-B ligand (RANKL, also known as tumor necrosis factor ligand superfamily member 11, TNFSF11), suppresses bone resorption markers in patients with a variety of metastatic tumors. It was developed for the treatment of osteoporosis, treatment-induced bone loss, metastases to bone, and giant cell tumor of bone. On June 1, 2010, denosumab was approved by the US Food and Drug Administration (FDA) for use in postmenopausal women with risk of osteoporosis, and in November 2010, for the prevention of skeleton-related events in patients with bone metastases from solid tumors. Denosumab is the first RANKL inhibitor to be approved by the FDA. In addition, the drug was approved for marketing by the European Commission on May 28, 2010. At present, denosumab is an efficacious and safe osteoporosis treatment option, with current data up to 8 years of continued use showing continued improvement in bone density with sustained fracture risk reduction. Safety profiles overall are similar to placebo, with no new safety concerns in extension trials, though a theoretical increased risk of infection exists with RANKL inhibition.
Mechanism of Action of Denosumab
Osteoporosis is a systemic skeletal disease that increases with age and is common among postmenopausal women. Characterized by reduced bone mineral density (BMD) and weakened bone structure, osteoporosis decreases bone resistance to low-energy trauma and increases bone fragility and fracture risk. A complex mechanism controls bone remodeling in human. This mechanism includes the receptor activator of nuclear factor kappa B ligand (RANKL), its natural receptor (RANK) and osteoprotegerin (OPG). The RANK/RANKL pathway is a key that promotes osteoclast formation and activation and prolongs osteoclast survival. OPG acts as a decoy receptor for RANKL and prevents its interaction with RANK thereby inhibiting osteoclast formation, function and survival. Alteration of the RANK/RANKL/OPG system in favor of increased osteoclastic activity and enhanced osteoblastic dysfunction is proposed as an important mechanism in the etiology of osteoporosis. Denosumab is a antiresorptive agent, with a novel mechanism of action. Briefly, denosumab is a fully human monoclonal antibody that inhibits RANKL and helps regulate turnover in healthy bone. Denosumab binds with high specificity and affinity to the cytokine RANKL, inhibiting its action; as a result, osteoclast recruitment, maturation and action are inhibited, and bone resorption slows. Denosumab is indicated for postmenopausal women with osteoporosis at high risk of fracture, or for patients who have failed or are intolerant to other available osteoporosis therapies.
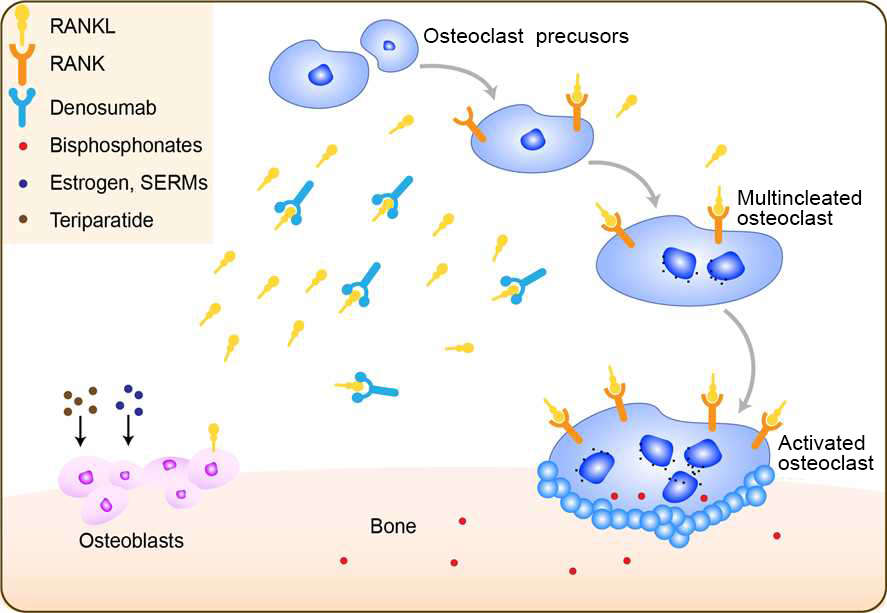
Fig 1. Mechanism of Action of Denosumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

