Pancreatic Cancer Overview
About Pancreatic Cancer
Pancreatic cancer is a highly malignant digestive system tumor whose etiology and pathogenesis are still unknown. Pancreatic cancer is a cancer that develops from pancreatic tissue. It is a disease in which pancreatic cells become cancerous and can invade other tissues and spread to other parts of the body. The clinical characteristics of pancreatic cancer are short course of disease, rapid development and rapid deterioration of the disease. Its most common symptoms include emaciation, jaundice, abdominal mass, hepatosplenomegaly and so on. The early stage of the pancreas usually does not have obvious and specific symptoms, so it is difficult to be detected in time, and the diagnosis is usually in the late stage of the disease. The risk factors of pancreatic cancer can be divided into two types, congenital factors and acquired factors. Congenital factors are inherited by family genes. Acquired factors include smoking, drinking, obesity, associated diabetes and chronic pancreatitis. Early clinical symptoms of pancreatic cancer can sometimes be characterized by diabetes and chronic pancreatitis. Epidemiological studies have confirmed that pancreatic cancer has the characteristics of familial aggregation, and the mutations of CDKN2A, BRCA1/2, PALB2 and other genes are closely related to the pathogenesis of familial pancreatic cancer. According to the classification of the World Health Organization, pancreatic malignant tumors can be divided into epithelial source and non-epithelial source according to the origin of tissue. Epithelial tumors include ductal adenocarcinoma, acinar cell carcinoma and neuroendocrine tumors from ductal epithelium, acinar cells and neuroendocrine cells, as well as a variety of mixed tumors. The diagnosis of pancreatic cancer is usually based on imaging and biomarker detection. CA19-9 is the most commonly used diagnostic marker of pancreatic cancer.
Main Signaling Pathways in Colorectal Cancer
Diagnosis of Pancreatic Cancer
The early symptoms of pancreatic cancer are atypical, often manifested as upper abdominal discomfort, low back pain, dyspepsia or diarrhea, which is easy to be confused with other digestive system diseases. Because pancreatic cancer has no specific clinical manifestations, it is difficult to detect it in time during the physical examination. Usually, the patient's medical history will provide some clues for the diagnosis of pancreatic cancer. The diagnosis and staging of pancreatic cancer depend on a variety of laboratory detection methods and imaging diagnosis methods. Laboratory testing methods are usually used to detect the physiological indicators and tumor markers of patients. Serum CA19-9 is the only pancreatic cancer marker confirmed and widely used in the clinic. The diagnosis of pancreatic cancer mainly depends on imaging and histological examination. The imaging methods of pancreatic cancer include percutaneous or internal ultrasound, Computed tomography (CT), endoscopic retrograde cholangiopancreatography (ERCP), magnetic resonance imaging (MRI), magnetic resonance cholangiopancreatography (MRCP) and positron emission tomography (PET). In addition, pathological examination is still the final standard for the detection of pancreatic cancer. Pathological examination of paraffin sections can be used to determine the benign and malignant tissues. Immunohistochemistry can be used to classify the origin and prognosis of tumors. In addition, puncture cytology and exfoliative cytology are also an effective method to obtain the pathological diagnosis of pancreatic cancer. In theory, tumor puncture can lead to the spread of abdominal cavity and needle passage. Therefore, if all kinds of preoperative diagnostic results are highly suspected to be pancreatic tumors, and the mass can be respectable, puncture examination is not recommended. However, if the mass is unresectable and clinically highly suspected of pancreatic cancer, puncture cytology can be performed before radiotherapy or chemotherapy to obtain pathological confirmation.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


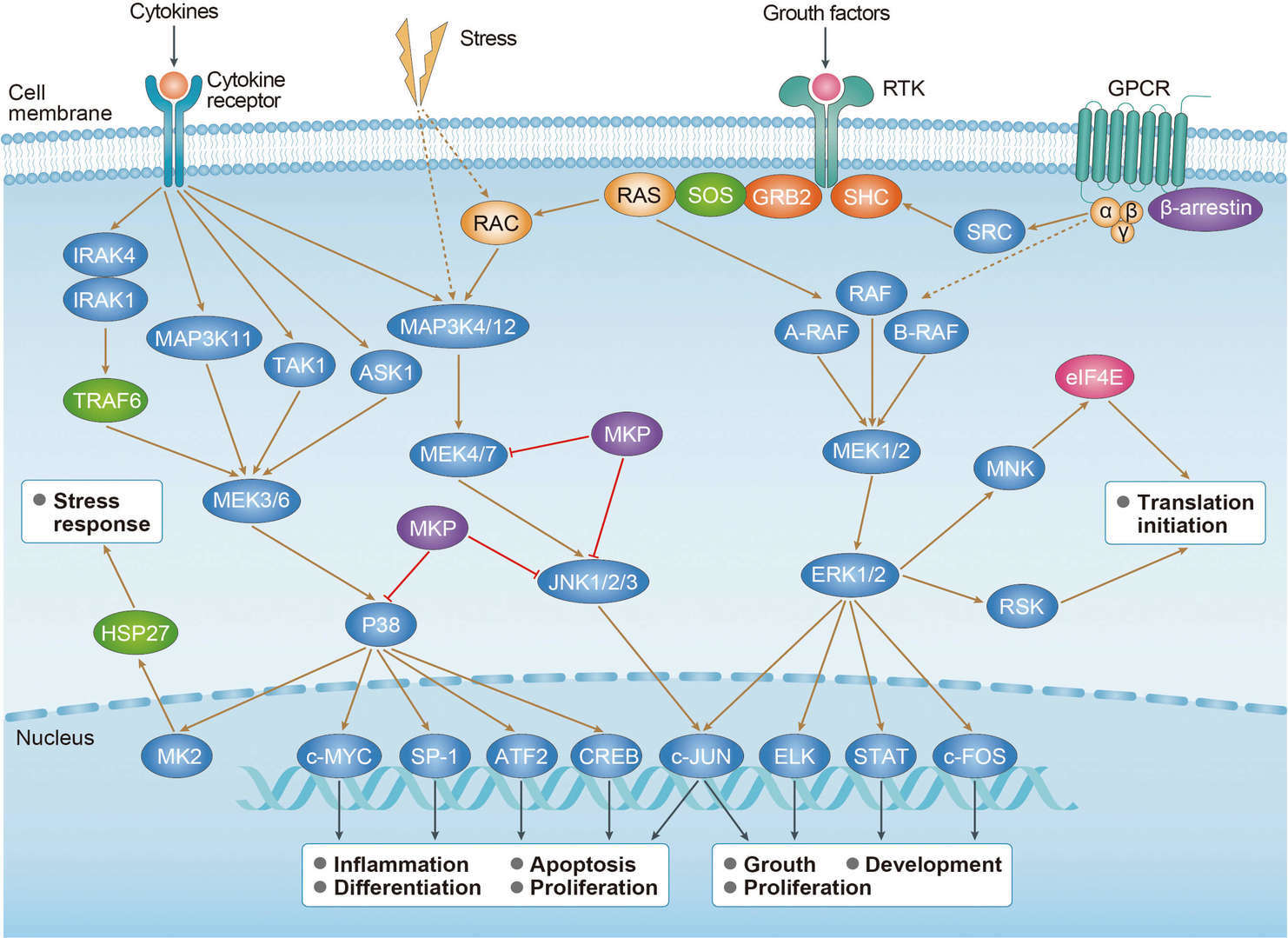 MAPK Pathway
MAPK Pathway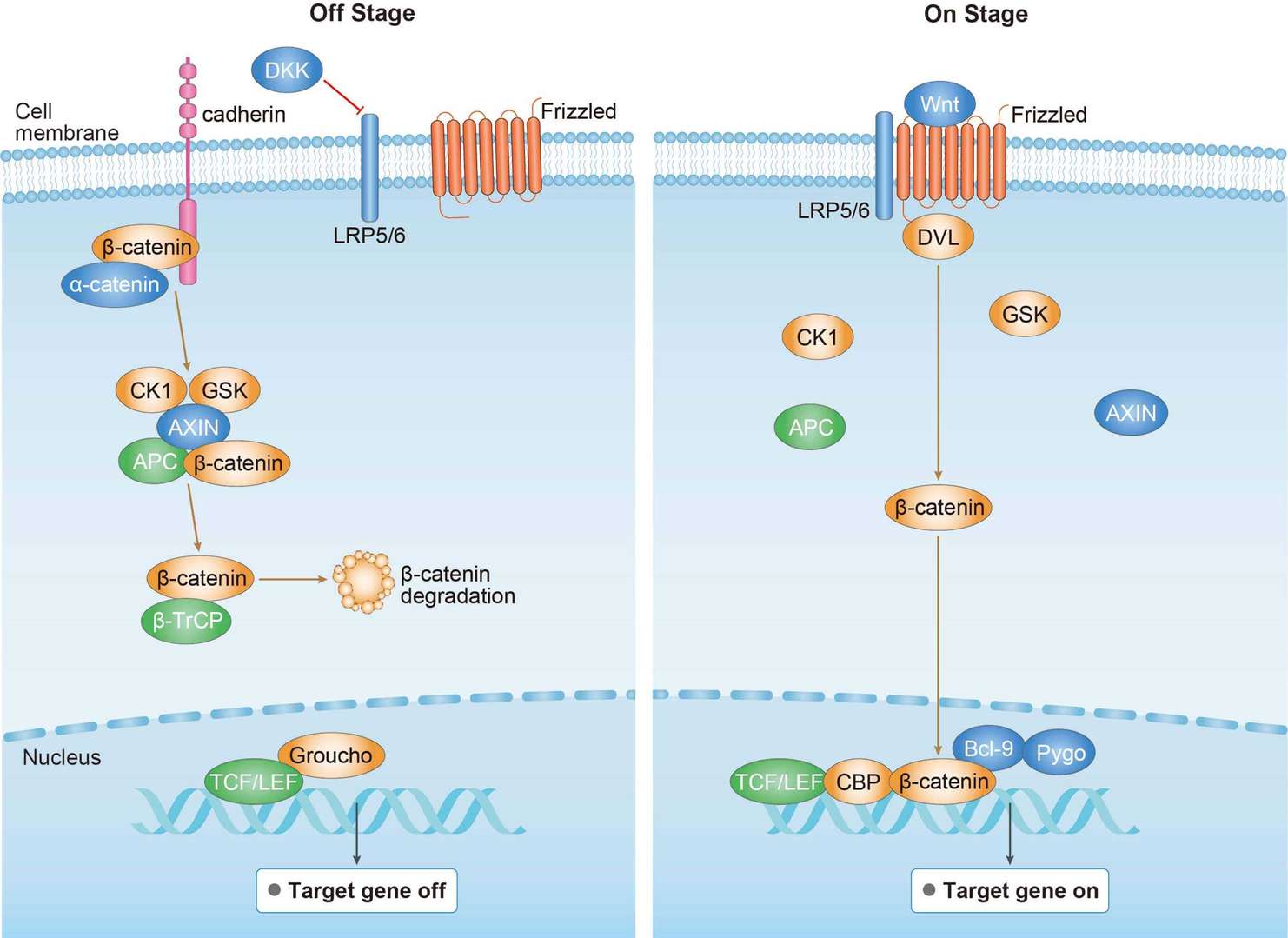 Wnt Pathway
Wnt Pathway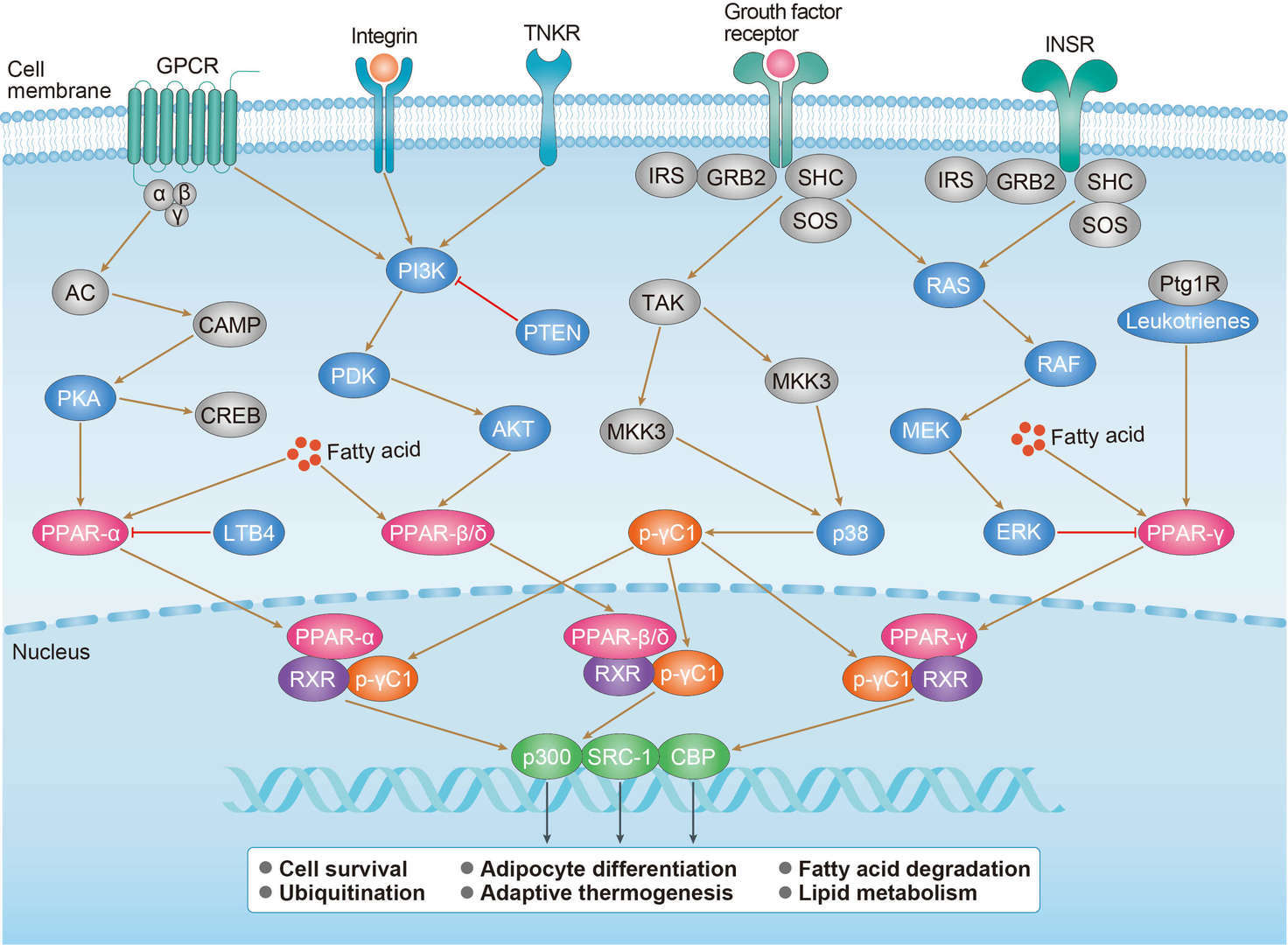 PPAR Pathway
PPAR Pathway