Vatelizumab Overview
Introduction of Vatelizumab
Vatelizumab is an immunomodulator for some autoimmune disorders. It is a humanized monoclonal antibody binds to integrin alpha 2, which is an intergrin alpha subunit. The originator of vatelizumab is Chormos Molecular Systems, a biotechnology company. And then, it was developed by Glenmark Pharmaceuticals S.A. and Sanofi. Vatelizumab has been used in trials studying the treatment of various diseases including chronic obstructive pulmonary disease, multiple sclerosis, retinal disorders, rheumatoid arthritis and ulcerative colitis. On 01 Jan 2016, Glenmark announced that is available for licensing for autoimmune disorders. However, in Jul 2016, Sanofi terminates a phase II/III trial in multiple sclerosis based on the planned interim analysis. In addition, it was withdrawn from phase II trials for inflammatory bowel disease as the treatment was not as effective as expected. Recent study of vatelizumab has been reported in May 2019. The safety and efficacy of vatelizumab during a phase 2 clinical study in multiple sclerosis has been investigated.
Mechanism of Action of Vatelizumab
Integrin alpha 2 (ITGA2), the target of vatelizumab, is a subunit of integrin α2β1. Integrin α2β1 is also known as very late antigen (VLA)-2 which expressed on a wide variety of cell types including T cells, NK cells, fibroblasts and platelets. It was first identified as an extracellular matrix receptor for collagens and/or laminins, while now recognized as a receptor for many matrix and nonmatrix molecules. Integrin α2β1 preferentially binds fibril-forming collagens and plays important roles in platelet adhesion, branching morphogenesis, mast cell activation, and the regulation of cell migration. The heterodimeric integrin α2β1 is composed of a distinct α2 chain and a common β1 chain. The humanized monoclonal immunoglobulin G (IgG) 4 antibody, vatelizumab, is designed to bind the α2 subunit domain of integrin α2β1. The clear mechanism of action of vatelizumab is not known, but it is hypothesized to block the interaction of integrin α2β1 with collagen through binding to the α2 subunit of integrin α2β1.
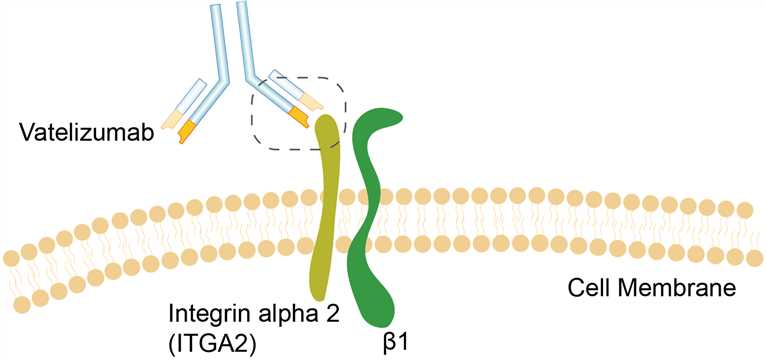 Fig.1 Mechanism of Action of Vatelizumab
Fig.1 Mechanism of Action of Vatelizumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

