

Bladder Cancer Biomarkers
Bladder cancer is a significant global health concern, characterized by distinct forms such as non-muscle-invasive bladder cancer (NMIBC), muscle-invasive bladder cancer (MIBC), and metastatic disease. Advances in molecular biology and sequencing technologies have unveiled a wealth of DNA, RNA, and protein biomarkers that enhance our understanding of bladder cancer's heterogeneity. These biomarkers are crucial for diagnosing, predicting disease progression, and tailoring individualized treatment strategies. The identification of molecular subtypes and their associated biomarkers represents a paradigm shift in bladder cancer management, aiming for early detection, monitoring recurrence, and improving therapeutic outcomes.
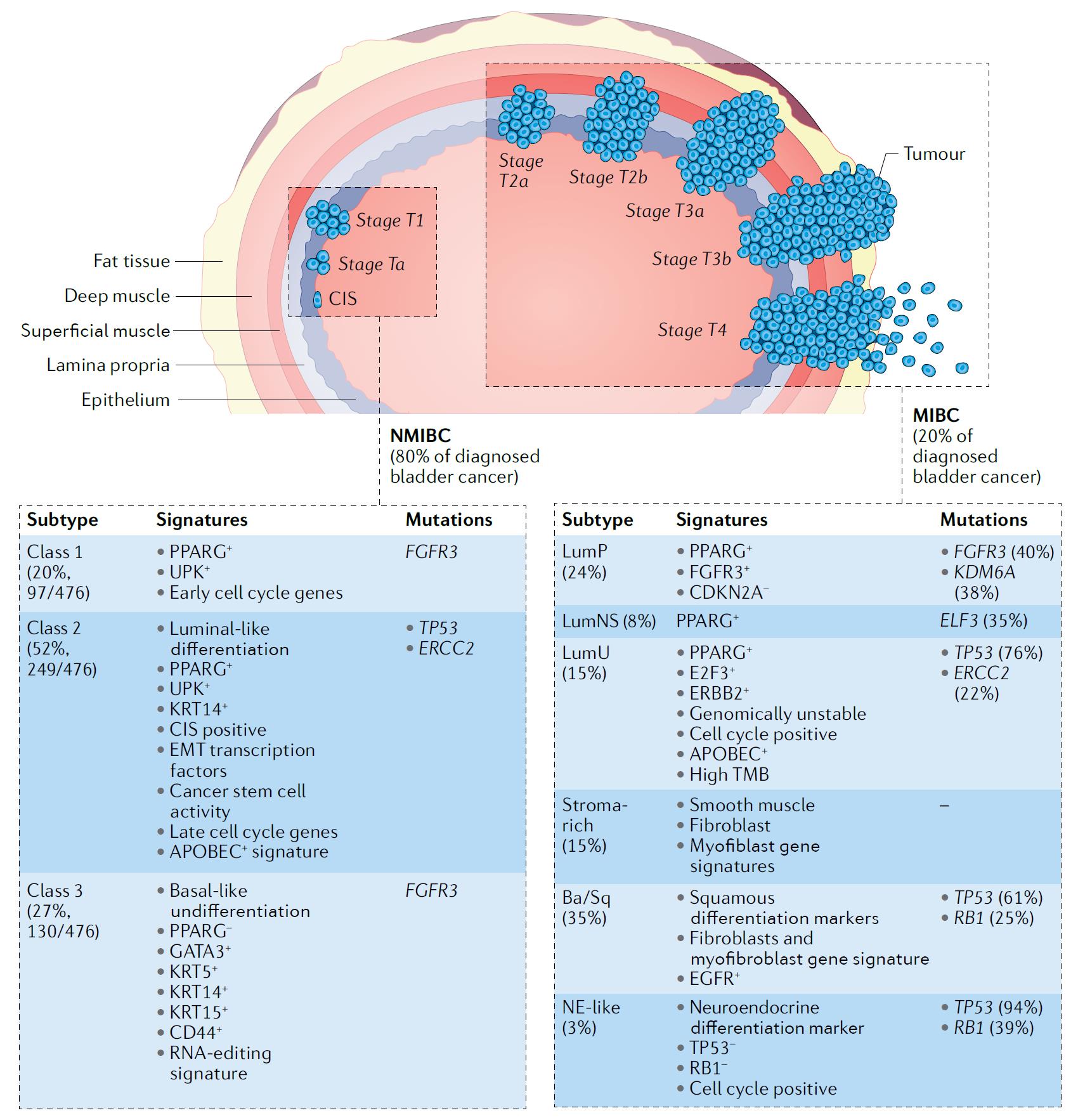 Figure 1 Pathological and molecular features of human bladder cancer. (Tran, 2021)
Figure 1 Pathological and molecular features of human bladder cancer. (Tran, 2021)
Representative Biomarkers of Bladder Cancer
ERCC1
DNA excision repair protein ERCC-1 plays a critical role in the nucleotide excision repair pathway, responsible for repairing bulky DNA adducts and crosslinks, which is vital for maintaining genome integrity. Its overexpression in solid tumors is linked to increased DNA repair capacity and resistance to platinum-based chemotherapy, commonly used in treating urothelial cancer, including bladder cancer. ERCC1's expression and polymorphisms have been extensively studied for their prognostic and predictive value in bladder cancer treatment. High ERCC1 expression is associated with a favorable outcome in muscle-invasive bladder cancer (MIBC) treated with surgery alone, suggesting a reduced DNA repair capacity and greater tumor mutational burden in ERCC1-low expressing tumors. Conversely, ERCC1-low expressing tumors show increased sensitivity to platinum-based chemotherapy, indicating ERCC1's potential as a biomarker to predict treatment response. Recent studies have focused on targeting ERCC1 to enhance cisplatin sensitivity, highlighting its importance in bladder cancer therapeutics and personalized medicine approaches.

MMP9
Matrix metalloproteinase-9 (MMP9) is pivotal in extracellular matrix remodeling, crucial for tissue repair and disease processes, including cancer. It specifically degrades type IV collagen, integral to basement membranes, and its regulation is crucial; overexpression can lead to cancer progression, facilitating tumor growth and spread. In bladder cancer, MMP9's role is particularly pronounced; its heightened expression is linked to the tumor's invasive and metastatic capabilities. This association points to MMP9 not just as a facilitator of cancer cell movement through tissue barriers but also as a participant in tumor angiogenesis and immune system evasion. Consequently, MMP9 is a focus for identifying aggressive tumors and a target for therapeutic strategies in bladder cancer treatment. By targeting MMP9, there's potential to block tumor growth and spread, offering a promising avenue for improving patient outcomes.


Interleukin 8 (IL-8)
Interleukin-8 (IL-8) is a crucial chemokine produced by various types of cells, including macrophages, epithelial cells, and endothelial cells, playing a pivotal role in the immune response by attracting neutrophils to sites of infection or injury. Beyond its fundamental role in inflammation, IL-8 is involved in the pathogenesis of numerous diseases, including cancer, where it promotes angiogenesis, tumor growth, and metastasis. In the context of bladder cancer, IL-8's functions become particularly significant. It has been found to contribute to the aggressive behavior of bladder tumors by enhancing cancer cell proliferation, survival, and migration, as well as by facilitating the formation of new blood vessels, which tumors need for oxygen and nutrients. This makes IL-8 not only a marker of disease progression but also a potential therapeutic target, offering new avenues for treatment strategies aimed at blocking its effects to inhibit tumor growth and spread.
MKI67
Ki-67, a marker for cell proliferation, is crucial in assessing growth rates across tissues, highlighting its significance in oncology. Present in the nucleus during active cell phases but not in dormant cells, Ki-67's expression is a telltale of cellular activity. In bladder cancer, its levels correlate with tumor aggressiveness, making it a valuable prognostic tool. Elevated Ki-67 suggests rapid tumor growth, signaling a potentially aggressive cancer type. Thus, measuring Ki-67 aids in risk stratification and informs treatment choices, from surgery to chemotherapy. This use of Ki-67 not only enhances understanding of tumor biology but also fosters personalized treatment approaches in bladder cancer, reflecting its importance in improving patient management and outcomes.



Full List of Bladder Cancer Biomarkers
| Biomarker | Alternative Names | Gene ID | UniProt ID | Roles |
| APOA1 | Apolipoprotein A1; Apolipoprotein A-I; Apo-AI; Apo(A); ApoA-I | 335 | A0A024R3E3 | This gene encodes apolipoprotein A-I, which is the major protein component of high density lipoprotein (HDL) in plasma. The encoded preproprotein is proteolytically processed to generate the mature protein, which promotes cholesterol efflux from tissues to the liver for excretion, and is a cofactor for lecithin cholesterolacyltransferase (LCAT), an enzyme responsible for the formation of most plasma cholesteryl esters. This gene is closely linked with two other apolipoprotein genes on chromosome 11. Defects in this gene are associated with HDL deficiencies, including Tangier disease, and with systemic non-neuropathic amyloidosis. Alternative splicing results in multiple transcript variants, at least one of which encodes a preproprotein. [provided by RefSeq, Dec 2015] |
| ERCC1 | ERCC Excision Repair 1, Endonuclease Non-Catalytic Subunit; Excision Repair Cross-Complementing Rodent Repair Deficiency, Complementation Group 1 (Includes Overlapping Antisense Sequence); Excision Repair Cross-Complementation Group 1; DNA Excision Repair Protein ERCC-1; COFS4; RAD10; UV20; | 2067 | P07992 | The product of this gene functions in the nucleotide excision repair pathway, and is required for the repair of DNA lesions such as those induced by UV light or formed by electrophilic compounds including cisplatin. The encoded protein forms a heterodimer with the XPF endonuclease (also known as ERCC4), and the heterodimeric endonuclease catalyzes the 5' incision in the process of excising the DNA lesion. The heterodimeric endonuclease is also involved in recombinational DNA repair and in the repair of inter-strand crosslinks. Mutations in this gene result in cerebrooculofacioskeletal syndrome, and polymorphisms that alter expression of this gene may play a role in carcinogenesis. Multiple transcript variants encoding different isoforms have been found for this gene. The last exon of this gene overlaps with the CD3e molecule, epsilon associated protein gene on the opposite strand. |
| IL-8 | Interleukin-8 | |||
| MKI67 | Marker Of Proliferation Ki-67; Antigen Identified By Monoclonal Antibody Ki-67; Protein Phosphatase 1, Regulatory Subunit 105; Antigen KI-67; Antigen Ki67; Proliferation-Related Ki-67 Antigen | 4288 | P46013 | This gene encodes a nuclear protein that is associated with and may be necessary for cellular proliferation. Alternatively spliced transcript variants have been described. A related pseudogene exists on chromosome X. [provided by RefSeq, Mar 2009] |
| MMP9 | Matrix Metallopeptidase 9; Matrix Metalloproteinase 9 (Gelatinase B, 92kDa Gelatinase, 92kDa Type IV Collagenase); EC 3.4.24.35; CLG4B; MMP-9; GELB; Matrix Metallopeptidase 9 (Gelatinase B, 92kDa Gelatinase, 92kDa Type IV Collagenase); Matrix Metalloproteinase-9 | 4318 | P14780 | Proteins of the matrix metalloproteinase (MMP) family are involved in the breakdown of extracellular matrix in normal physiological processes, such as embryonic development, reproduction, and tissue remodeling, as well as in disease processes, such as arthritis and metastasis. Most MMP's are secreted as inactive proproteins which are activated when cleaved by extracellular proteinases. The enzyme encoded by this gene degrades type IV and V collagens. Studies in rhesus monkeys suggest that the enzyme is involved in IL-8-induced mobilization of hematopoietic progenitor cells from bone marrow, and murine studies suggest a role in tumor-associated tissue remodeling. [provided by RefSeq, Jul 2008] |
Tested Data-Supported Products Targeting Bladder Cancer Biomarkers
| CAT | Product Name | Biomarker | Assay | Image |
| ZG-0020C | Mouse Anti-ERCC1 Recombinant Antibody (clone 1B10) | ERCC1 | WB |

|
| MOB-0066F | Mouse Anti-MKI67 Recombinant Antibody (clone 4A8) | MKI67 | IF |

|
| MOB-0067F | Mouse Anti-MKI67 Recombinant Antibody (MOB-0067F) | MKI67 | IHC |

|
| ZG-0296F | Mouse Anti-ApoA1 Recombinant Antibody (ZG-0296F) | ApoA1 | WB |

|
| ZG-0240C | Mouse Anti-MKI67 Recombinant Antibody (ZG-0240C) | MKI67 | IHC |

|
| ZG-0242C | Mouse Anti-MKI67 Recombinant Antibody (ZG-0242C) | MKI67 | WB |

|
| ZG-0404U | Rabbit Anti-MKI67 Recombinant Antibody (clone M17-I) | MKI67 | IHC-P |

|
- Tran, Linda, et al. "Advances in bladder cancer biology and therapy." Nature Reviews Cancer 21.2 (2021): 104-121.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
