

In Vitro Enzymic and Chemo-Enzymatic Antibody Glycosylation Remodeling Service
Creative Biolabs has proudly established the unique GlycoOpitimize™ platform based on our extensive experience on the development of antibody glycoengineering. Clients all over the world can collaborate with our staff of experts to get specialized, reliable, high-quality services and solutions. Our in vitro enzymic and chemo-enzymatic antibody glycosylation remodeling service cover a broad range of options from enzyme catalyzed breaking or forming of specific glycosidic bonds to chemo-enzymatic modifying, remodeling the antibody with homogeneous N-glycans, analyzing the site and structure of oligosaccharide components and more.
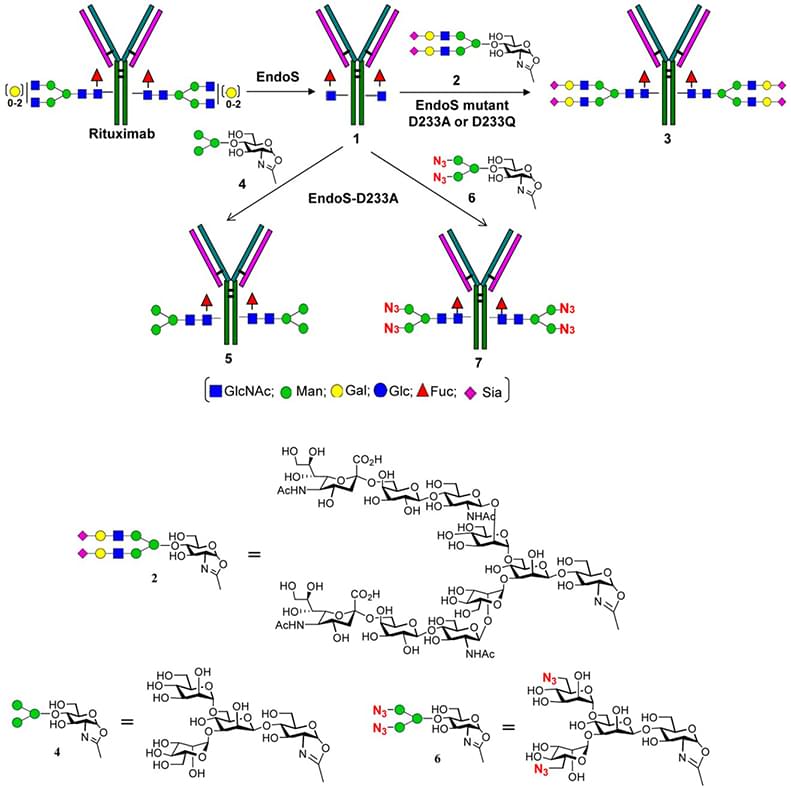
The main method of antibody glycoengineering is manipulation of host biosynthetic pathways. However, in vitro enzymic and chemo-enzymatic antibody glycosylation remodeling is another attractive method and gaining more and more attentions. This method involves modification of the glycoform with specific enzymes and chemicals after the expression of antibody. For example, incubating the target antibody with highly specific β-1,4--galactosyltransferase and α-2,6-sialyltransferase and the corresponding activated sugar substrates (UDP-Gal, CMP-sialic acid) can lead to the formation of over- or even per-sialylated antibodies. Another impressive example comprises three main steps. First, an endoglycosidase, such as the wild type endoglycosidase S (Endo S) from Streptococcus pyogenes is used to cleave the bulk of the oligosaccharide from the Fc glycan while the innermost GlcNAc or the fucosylated GlcNAc moieties are retained. The product contains a homogeneous well-defined glycan and is a suitable substrate for the chemo-enzymatic addition. Second, oxazoline derivatives of N-glycans are obtained by chemical synthesis or isolation methods. The third step is extension of the glycan using glycosyltransfer enzymes. These enzymic and chemo-enzymatic processes lead to predictable and reproducible outcomes and reduce the complexity of antibodies.
Creative Biolabs has many advanced techniques in the field of glyco-engineering. Our wholly dedicated scientist are proficiency in using various enzymic and chemo-enzymatic technologies to obtain the glyco-engineered antibodies according to your unique requirements. All products are verified by our antibody glycan analysis systems and only those that match the requested structure are shipped. Send us your project description and needs and you can get a tailored all-in-one service to meet your R&D or cGMP production needs.
References
- Washburn, N, Schwab, I, et al. (2015) Controlled tetra-Fc sialylation of IVIg results in a drug candidate with consistent enhanced anti-inflammatory activity. Proceedings of the National Academy of Sciences, 112(11), E1297-E1306.
- Giddens, JP, & Wang, LX. (2015). Chemoenzymatic glyco-engineering of monoclonal antibodies. Glyco-Engineering: Methods and Protocols, 375-387.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
