Palivizumab Biosimilar Pipeline Development Service
Comprehensive Solutions for RSV F protein-Targeted Biosimilar Development
Creative Biolabs provides end-to-end development solutions for Palivizumab biosimilars, guiding clients through every phase from initial analytical assessment to final regulatory filing. Leveraging extensive experience in antibody design and RSV F protein targeting technologies, we ensure a scientifically robust and compliant pathway through the biosimilar development process.
- At a glance of Palivizumab
Service Overview
Palivizumab is a humanized monoclonal antibody specifically designed to target the fusion (F) protein of respiratory syncytial virus (RSV), a key factor in viral entry into host cells. By binding to a highly conserved epitope on the F protein, Palivizumab effectively neutralizes the virus and prevents infection of respiratory epithelial cells. It is widely used for the prevention of severe RSV disease in high-risk pediatric populations, including premature infants and those with chronic lung or congenital heart conditions. Engineered to combine human and murine antibody elements, Palivizumab demonstrates reduced immunogenicity and favorable pharmacokinetic properties, making it a valuable tool in RSV prophylaxis.
Basic Information
Drug Type: Monoclonal antibody
Synonyms: Palivizumab (Genetical Recombination), Palivizumab (genetical recombination) (JAN), Palivizumab (USAN/INN)
Target: RSV F protein
Action: Inhibitors
Mechanism: Respiratory syncytial virus F protein inhibitors
Inactive Indication: Aortic Coarctation; Chronic lung disease; Ductus Arteriosus, Patent
Originator Organization: AbbVie, Inc.
Inactive Organization: Abbott Laboratories; AbbVie, Inc.; MedImmune LLC
First Approval Date: United States (19 Jun 1998)
Most Recent Events
| Date | Events |
| 17 Dec 2024 | Merck Announces Acceptance of Biologics License Application for Clesrovimab, an Investigational Long-Acting Monoclonal Antibody Designed to Protect Infants from RSV Disease During their First RSV Season |
| 17 Dec 2024 | Merck Announces Acceptance of Biologics License Application for Clesrovimab, an Investigational Long-Acting Monoclonal Antibody Designed to Protect Infants from RSV Disease During their First RSV Season |
- Pipeline Status
Our Palivizumab Biosimilar Pipeline Offers
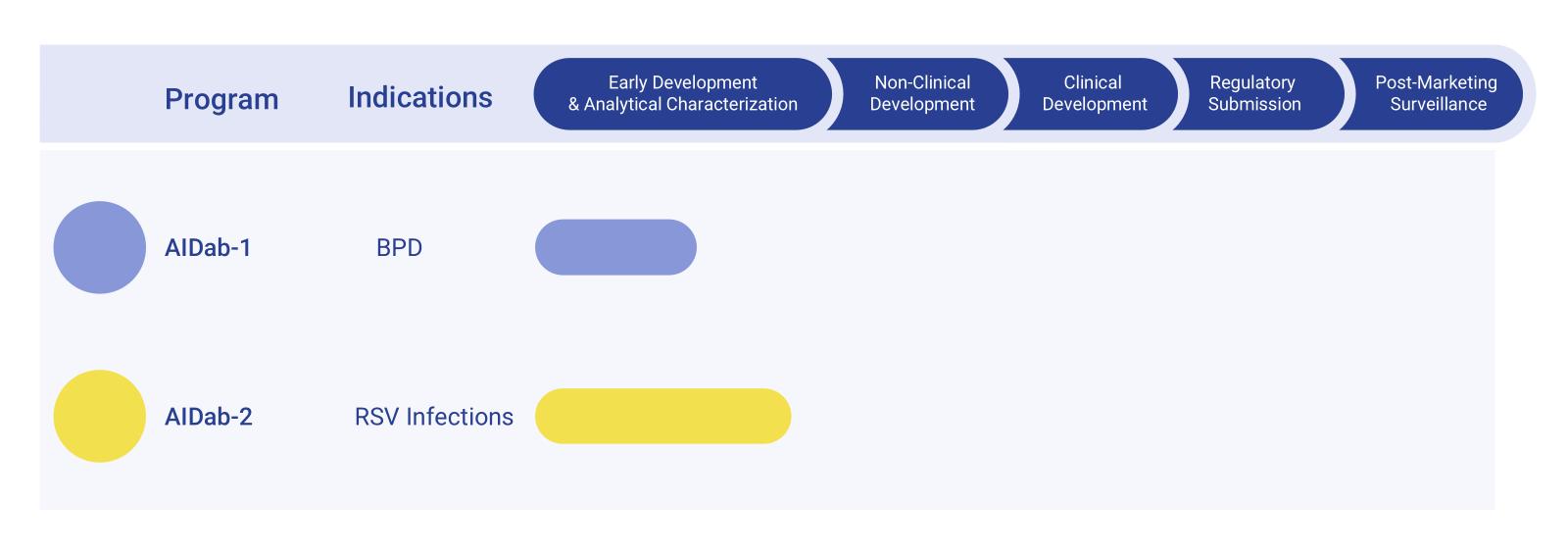
Development Workflow of Palivizumab Biosimilar Pipeline
| Early Development & Analytical Characterization | ||
| 1 | Project Initiation & Feasibility Assessment |
|
| 2 | Analytical Characterization |
|
| 3 | Cell Line Development |
|
| 4 | Process Development |
|
| 5 | Formulation Development |
|
| Non-Clinical Development | ||
| 6 | Preclinical Studies |
|
- Available Packages
Creative Biolabs provides customizable and scalable service solutions tailored to support every phase of your Palivizumab biosimilar development journey. Whether you're initiating analytical comparability studies or approaching regulatory submission, you can select from modular service options aligned with your specific project stage—or work with us to design a fully personalized development plan.
| Items | classification | What we can offer |
| 1 | Antibody Product | Fully characterized Palivizumab biosimilar antibody available in research-grade or GMP-grade formats, suitable for analytical testing and preclinical studies. |
| 2 | Cell Line | High-producing, stable CHO cell line expressing the biosimilar antibody, with detailed documentation including clone selection history, productivity profiles, and stability data. |
| 3 | Analytical & Biological Data |
|
- Why Choose Us
Integrated Development Platform
End-to-end services covering antibody production, cell line development, and comprehensive analytical characterization—all under one roof.
High Quality & Customization
Stable high-yield CHO cell lines, research- and GMP-grade antibodies, and flexible service packages tailored to your project stage and goals.
Expertise You Can Trust
Decades of antibody engineering experience with strong capabilities in biosimilarity assessment, ensuring scientific precision and efficient progress.
- Ready to Advance Your Palivizumab Biosimilar Program
Contact us to explore how we can create a personalized development strategy aligned with your regulatory objectives and project schedule. Reach out today to request a quote, book a consultation, or access our detailed service brochure.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

