Physicochemical Characterization
About Physicochemical Characterization Program
Biologics and biosimilars, also known as biotherapeutics, are produced by organisms such as bacteria, yeast, and mammalian cells. The structural characterization of therapeutic drugs is a key factor in ensuring efficacy and patient safety. It mainly includes from primary amino acid sequences to high order structures. Creative Biolabs has been committed to the development and research of biosimilars, and has a wide knowledge and experimental skills in physicochemical characterization of biosimilars. We are happy to share our valuable experience and research in biosimilars and seek partners who share the consensus with us. Ultimately gain a competitive advantage in the field of biosimilars and maximize the impact of physicochemical characterization programs.
Fig.1 Our physicochemical characterization program. (Creative Biolabs)
Primary Structure and Amino Acid Modification Characterization
| What We Can Do | Related Methods |
| Amino Acid Sequence |
|
| N/C-terminal Sequencing | |
| Intact Molecular Weight | |
| Disulfide Bonds | |
| Qualitative and quantitative determination of modifications |
- Published Data
Here are some typical figures about primary structure and amino acid modification characterization.
Analysis of Intact Molecular Mass
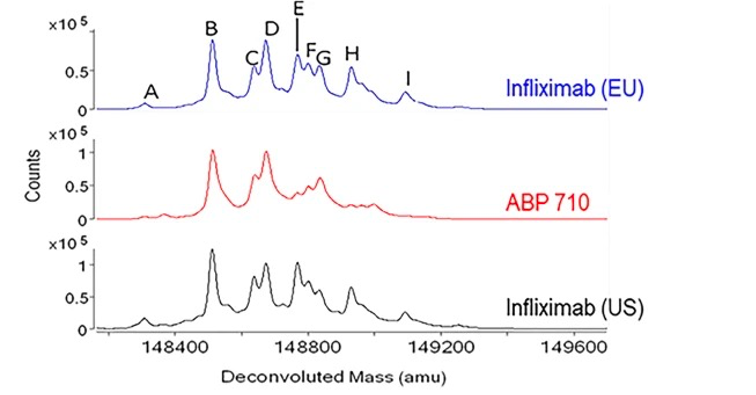 Fig.2 Intact molecular mass assessment of infliximab, ABP 710, and infliximab.1
Fig.2 Intact molecular mass assessment of infliximab, ABP 710, and infliximab.1
Analysis of Reduced Tryptic Peptide Map
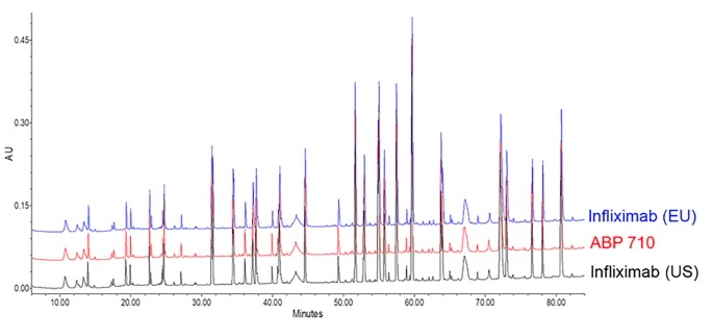 Fig.3 Reduced tryptic peptide map assessment of infliximab, ABP 710, and infliximab.1
Fig.3 Reduced tryptic peptide map assessment of infliximab, ABP 710, and infliximab.1
Higher Order Structure Characterization
| What We Can Do | Related Methods |
| Secondary Structure |
|
| Tertiary Structure | |
| Thermal Stability |
- Published Data
Here are some typical figures about higher order structure characterization.
Analysis of Secondary Structure
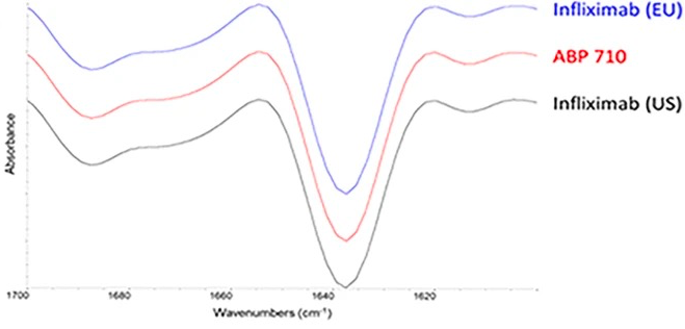 Fig.4 Secondary structure assessment of infliximab, ABP 710, and infliximab by FTIR.1
Fig.4 Secondary structure assessment of infliximab, ABP 710, and infliximab by FTIR.1
Analysis of Tertiary Structure
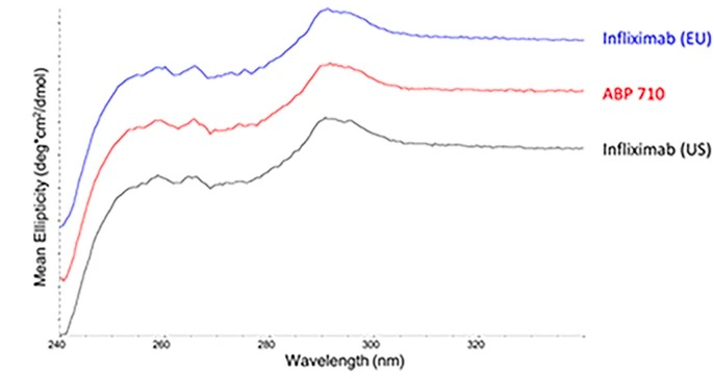 Fig.5 Tertiary structure assessment of infliximab, ABP 710, and infliximab by Near UV-CD.1
Fig.5 Tertiary structure assessment of infliximab, ABP 710, and infliximab by Near UV-CD.1
Analysis of Thermal Stability
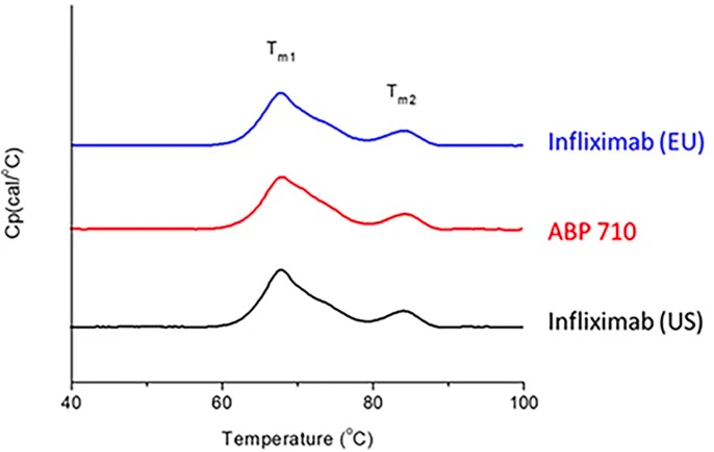 Fig.6 Thermal stability assessment of infliximab, ABP 710, and infliximab by DSC.1
Fig.6 Thermal stability assessment of infliximab, ABP 710, and infliximab by DSC.1
Heterogeneity Characterization
| What Can We Do | Related Methods |
| Charge (acidic, main, basic) |
|
| Size (dimers, high-molecular weight species, heavy chain and light chain fragments) | |
| Aggregates | |
| Hydrophobicity |
- Published Data
Here are some typical figures about heterogeneity characterization.
Analysis of Charge Heterogeneity
Analysis of Size Heterogeneity
Glycosylation Characterization
| What Can We Do | Related Methods |
| Afucosylation |
|
| Galactosylation | |
| High Mannose | |
| Sialylation |
- Published Data
Here are some typical figures about glycosylation characterization.
Analysis of Glycosylation Site
Analysis of Mannose
Analysis of Sialylation
Drug Product Attributes Characterization
| What Can We Do | Related Methods |
| Purity and Impurities |
|
| Protein Content | |
| Sub-visible Particles | |
| Deliverable volume | |
| Appearance, pH, osmolality |
- Published Data
Here are some typical figures about drug product attributes characterization.
Analysis of Purity
Do It Better Together
Creative Biolabs has a professional R&D team in biosimilar, which has strong professional knowledge and skills. We are aim to help you with customized services and comprehensive analysis of your biosimilar project. We are looking for outstanding scientists and professional technology platforms to collaborate. Through close collaboration, our partners can identify promising biosimilar drug candidates and determine accurate preclinical results. Creative Biolabs is committed to helping everyone from discovery to preclinical research and even IND applications. If you are interested in our Physicochemical Characterization program, contact us today to learn more!
References
- Saleem, Ramsey et al. “Analytical and Functional Similarity Assessment of ABP 710, a Biosimilar to Infliximab Reference Product.” Pharmaceutical research vol. 37,6 114. 31 May. 2020, doi:10.1007/s11095-020-02816-w. Distributed under Open Access license CC BY 4.0, without modification.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

