Antibody-Drug Conjugate Development Services
Creative Biolabs offers a wide range of Antibody-drug Conjugates (ADCs) that can be produced, including monoclonal antibody-based ADCs, polyclonal antibody-based ADCs, and bispecific antibody-based ADCs. Our main production process involves conjugating cytotoxic drugs to monoclonal antibodies through a linker molecule, ensuring targeted delivery of the drug to specific cancer cells while minimizing harm to healthy cells.
Services
Antibody-drug conjugate (ADC) services offered by Creative Biolabs typically involve the development, production, and characterization of ADCs for therapeutic purposes. Our services may include antibody selection and optimization, linker chemistry design, payload selection, and conjugation, in vitro/in vivo characterization testing, and scale-up production. Meanwhile, we also offer custom ADC development services tailored to specific client needs, as well as contract manufacturing services for ADC production. We aim to facilitate the development and production of highly targeted and potent ADC therapeutics for various diseases.
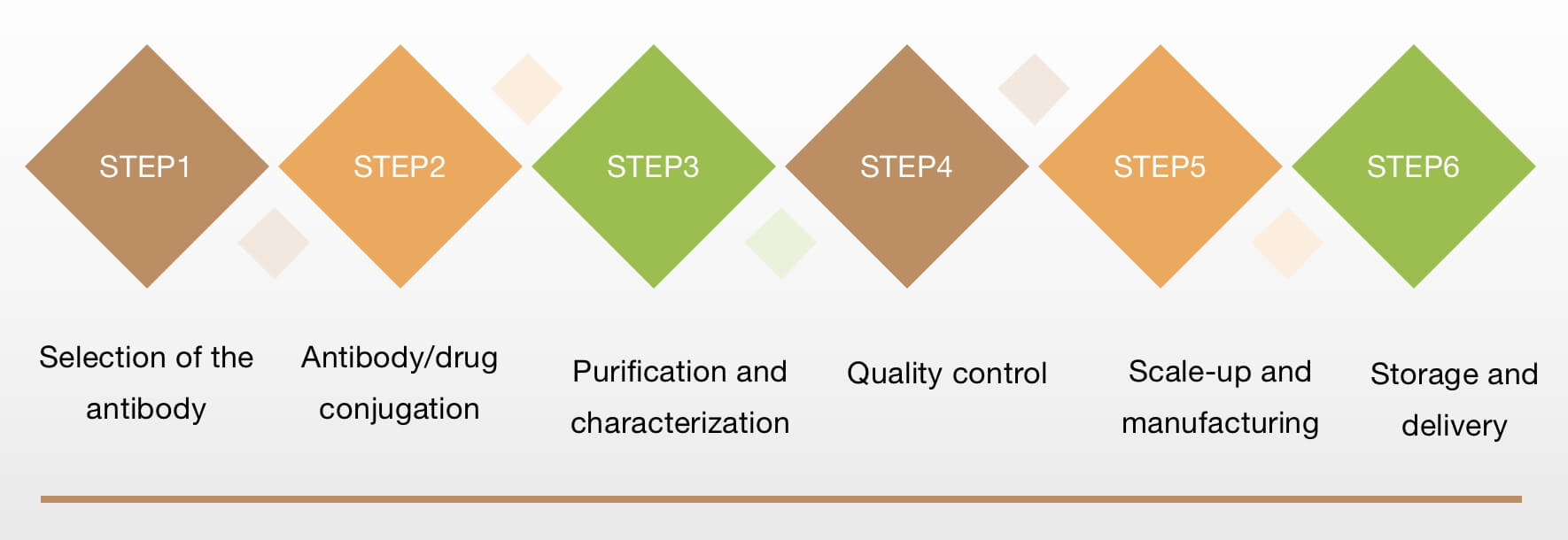 Fig.1 ADC Production Service Procedure. (Creative Biolabs Original)
Fig.1 ADC Production Service Procedure. (Creative Biolabs Original)
Platform
Creative Biolabs has established several key platforms relevant to ADC development, including but not limited to:
- Antibody selection and engineering: This platform involves selecting the right monoclonal antibody that targets cancer cells specifically and engineering it to improve its binding affinity, stability, and drug conjugation capacity.
- Drug-linker chemistry: This platform focuses on developing linkers that can stably attach potent cytotoxic drugs to the antibody while also allowing for the efficient release of the drug inside cancer cells.
- Conjugation: Different conjugation methods have been used by our labs to attach the drug to the antibody, such as site-specific conjugation, enzymatic conjugation, and chemical conjugation.
- Payloads: The choice of cytotoxic drug payload is critical for the efficacy of ADCs. In Creative Biolabs, different classes of drugs, including cytotoxins, radioisotopes, and immunomodulators, can be used as payloads.
- Manufacturing and formulation: This platform involves complex processes to ensure the stability, purity, and bioactivity of the final product. Formulation technologies are also important for optimizing pharmacokinetics and drug delivery.
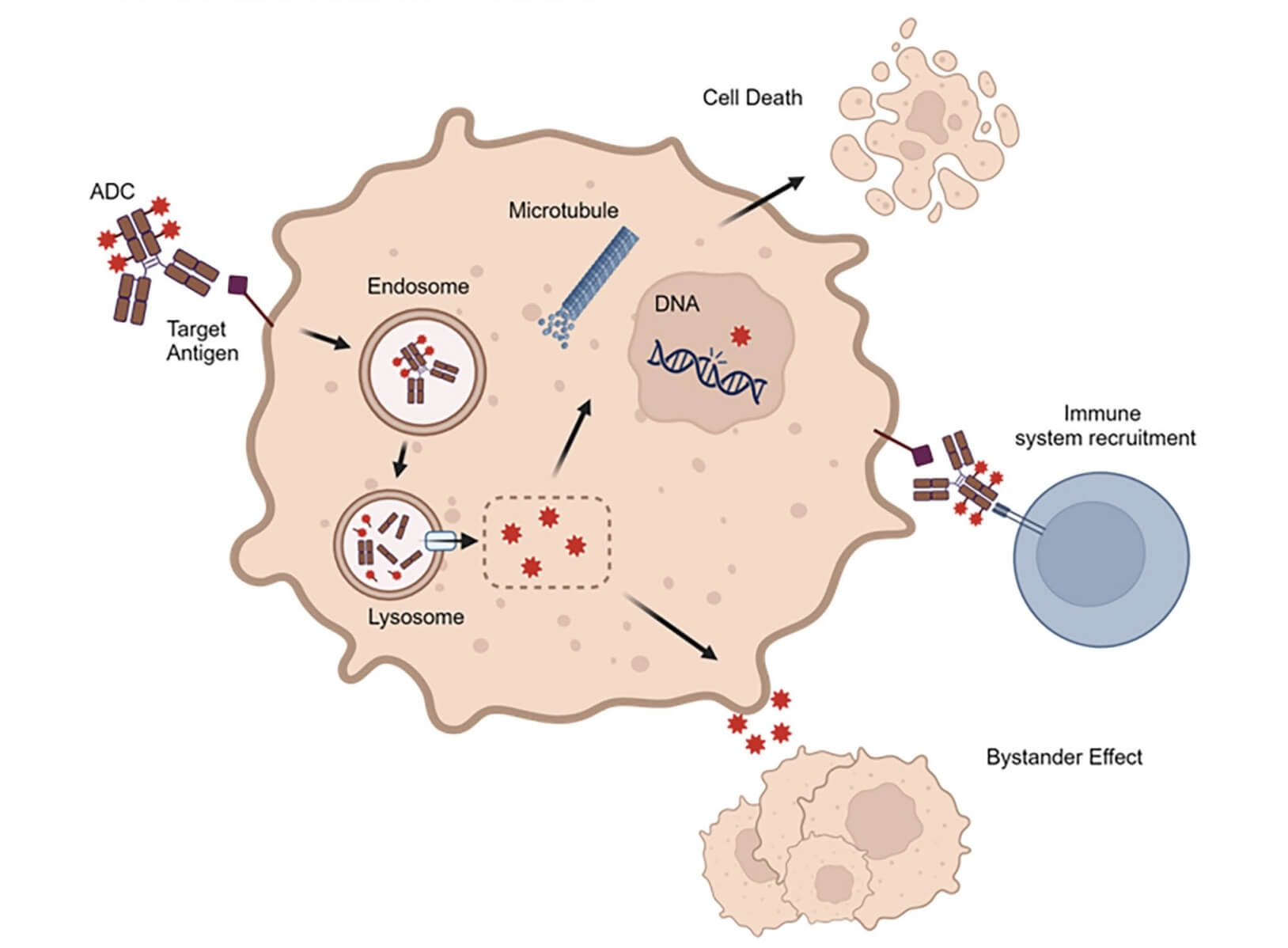 Fig.2 ADC Mechanisms of Action.1,3
Fig.2 ADC Mechanisms of Action.1,3
Advantages
Introducing our cutting-edge services for the production and characterization, analysis, and validation of Antibody Drug Conjugates (ADCs). Our Antibody-drug conjugates (ADCs) services have several advantages:
- Targeted delivery: ADCs use specific antibodies to target and deliver cytotoxic drugs directly to cancer cells, minimizing damage to healthy cells.
- Enhanced efficacy: By delivering high concentrations of potent drugs directly to tumor cells, ADCs can be more effective in killing cancer cells and reducing tumor growth.
- Lower toxicity: Targeted delivery reduces the exposure of healthy tissues to toxic chemotherapy drugs, leading to fewer side effects and improved patient outcomes.
- Improved specificity: ADCs can be designed to target specific cancer cell markers, increasing the specificity of the treatment and reducing the risk of off-target effects.
- Potential for combination therapy: ADCs can be used in combination with other cancer treatments, such as chemotherapy or immunotherapy, to enhance the overall effectiveness of the treatment regimen.
- Versatility: ADCs can be customized with different antibodies, cytotoxic drugs, and linker technologies to target a wide range of cancer types and patient populations.
Case Study
In a recent project, we utilized our expertise in bioconjugation techniques to attach a potent cytotoxic drug to a monoclonal antibody that specifically targets cancer cells. This antibody-drug conjugate can deliver the toxic payload directly to the cancer cells, leading to more effective and less toxic treatment for patients. Our state-of-the-art facilities and experienced team ensure the production of high-quality antibody-drug conjugates that meet the rigorous standards of regulatory agencies. The details are described below:
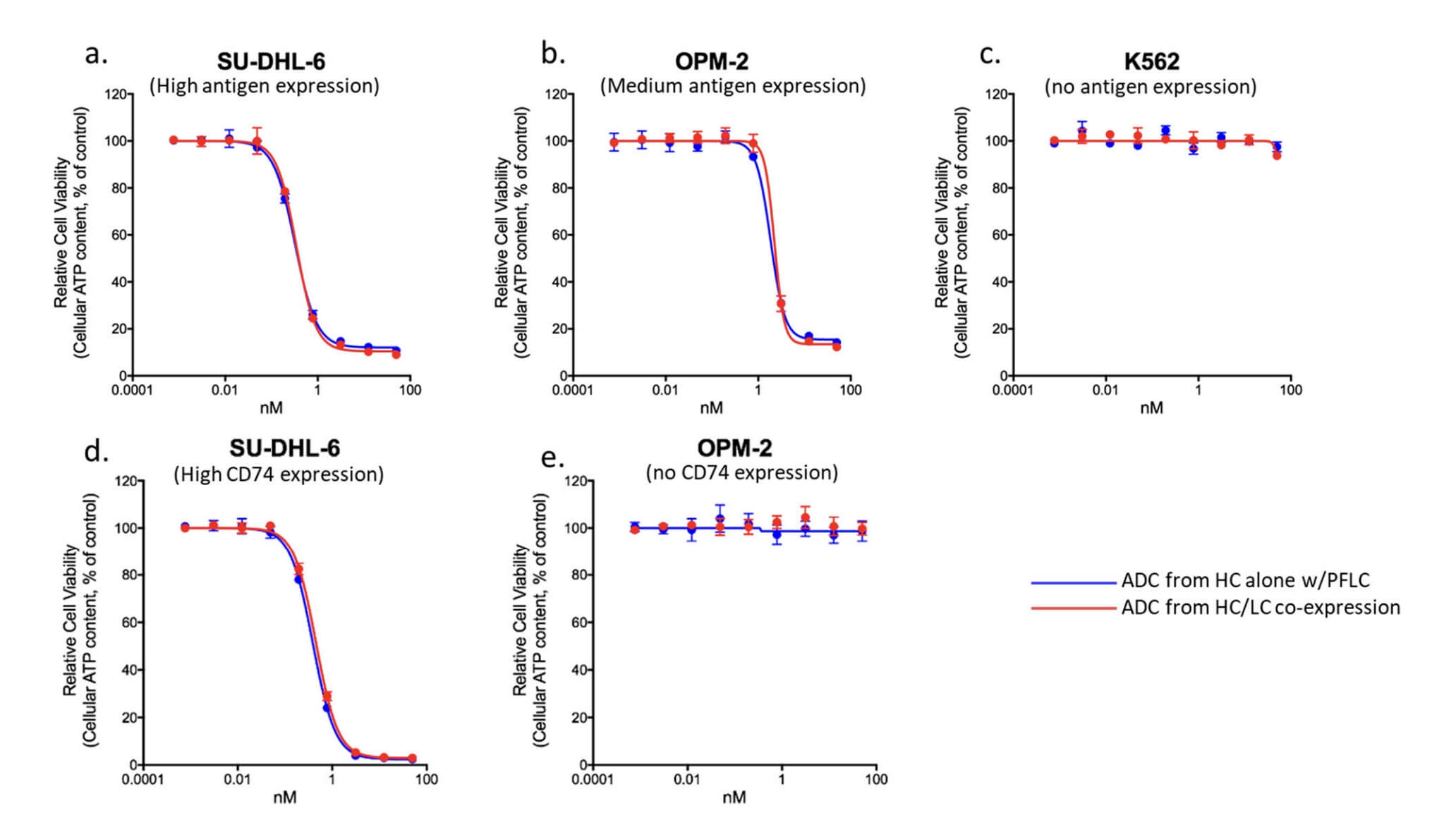 Fig.3 The Cell Killing Results for CD74 ADCs.2,3
Fig.3 The Cell Killing Results for CD74 ADCs.2,3
A monoclonal antibody specific for the cancer antigen of interest was produced using CHO cells in a mammalian cell expression system. The antibody was purified to homogeneity using protein A chromatography. The cytotoxic drug payload was attached to the antibody using a stable linker through a site-specific conjugation method. The resulting ADC was characterized for drug-to-antibody ratio, drug stability, and cytotoxic activity in vitro using cancer cell lines expressing the target antigen.
The ADC was successfully produced with a drug-to-antibody ratio of 3:1, indicating efficient conjugation of the cytotoxic drug to the antibody. The conjugation method used resulted in stable linkages between the antibody and drug payload, ensuring minimal premature drug release during circulation. In vitro studies showed that the ADC selectively targeted and killed cancer cells expressing the target antigen, with an IC50 value in the nanomolar range.
In Creative Biolabs, we specialize in the development and production of custom ADCs for various types of cancer. Our team of experts utilizes state-of-the-art technology and innovative techniques to create highly specific and potent ADCs that target and destroy cancer cells with a high level of precision. If you are interested in learning more about our ADCs or collaborating with us, please contact us and one of our sales representatives will be in touch with you shortly.
References
- Koukoutzeli, Chrysanthi, et al. "Use of Antibody-Drug Conjugates in the Early Setting of Breast Cancer." Clinical Medicine Insights: Oncology 18 (2024): 11795549241260418.
- Hanson, Jeffrey, et al. "An integrated in vivo/in vitro protein production platform for site-specific antibody drug conjugates." Bioengineering 10.3 (2023): 304.
- under Open Access License CC BY 4.0, without modification.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

