KD & KO Validation Antibody Products
Product List
 Loading...
Loading...- Knock-Valid™ Rabbit Anti-CHD3 Monoclonal Antibody (VS-1225-XU5)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-C1QBP Antibody (VS-1225-XU127)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AMPKα1 Antibody (VS-1225-XU126)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-SETD7 Antibody (VS-1225-XU125)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-p53 (Ab-15) Antibody (VS-1225-XU124)
-
- Species Reactivity: Human; Mouse; Rat; Monkey
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AKT1S1 Antibody (VS-1225-XU122)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-APAF1 Antibody (VS-1225-XU121)
-
- Species Reactivity: Human; Mouse
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ERK1/2 Antibody (VS-1225-XU120)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-c-Jun (Phospho-Ser249) Antibody (VS-1225-XU119)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GARS Monoclonal Antibody (VS-1225-XU118)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Atg7 (Apg7) Monoclonal Antibody (VS-1225-XU117)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Cathepsin L/V/K/H Monoclonal Antibody (VS-1225-XU116)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-EEF2 Monoclonal Antibody (VS-1225-XU114)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD82 Monoclonal Antibody (VS-1225-XU113)
-
- Species Reactivity: Human; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Dynamin 1 Monoclonal Antibody (VS-1225-XU112)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Emerin Monoclonal Antibody (VS-1225-XU111)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Grp75 Monoclonal Antibody (VS-1225-XU110)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AMBP Monoclonal Antibody (VS-1225-XU109)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-DHFR Monoclonal Antibody (VS-1225-XU108)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Atg4B Monoclonal Antibody (VS-1225-XU107)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD276 Monoclonal Antibody (VS-1225-XU106)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Calpain 1 Monoclonal Antibody (VS-1225-XU105)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CHD4 Monoclonal Antibody (VS-1225-XU104)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-TGF beta 1 Monoclonal Antibody (VS-1225-XU102)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Cdc25C Monoclonal Antibody (VS-1225-XU101)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Cyclin A1/A2 Monoclonal Antibody (VS-1225-XU100)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-EEA1 Monoclonal Antibody (VS-1225-XU99)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-p62/SQSTM1 Monoclonal Antibody (VS-1225-XU98)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GATA2/3 Monoclonal Antibody (VS-1225-XU97)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CLIC4 Monoclonal Antibody (VS-1225-XU96)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AKAP95 Monoclonal Antibody (VS-1225-XU95)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CLIP170 Monoclonal Antibody (VS-1225-XU94)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Bag1 Monoclonal Antibody (VS-1225-XU93)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CCDC98 Monoclonal Antibody (VS-1225-XU92)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AGL Monoclonal Antibody (VS-1225-XU91)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CLPTM1 Monoclonal Antibody (VS-1225-XU90)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CAP1 Monoclonal Antibody (VS-1225-XU89)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-HEAB Monoclonal Antibody (VS-1225-XU88)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ACAT2 Monoclonal Antibody (VS-1225-XU86)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ARPC2 Monoclonal Antibody (VS-1225-XU85)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-COASY Monoclonal Antibody (VS-1225-XU84)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD133 Monoclonal Antibody (VS-1225-XU83)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD58 Monoclonal Antibody (VS-1225-XU82)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-BRMS1 Monoclonal Antibody (VS-1225-XU81)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ACAA2 Monoclonal Antibody (VS-1225-XU80)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-FACL4 Monoclonal Antibody (VS-1225-XU79)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CIRBP Monoclonal Antibody (VS-1225-XU77)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-EHMT2 Monoclonal Antibody (VS-1225-XU76)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CC2D1A Monoclonal Antibody (VS-1225-XU75)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-AP1G1 Monoclonal Antibody (VS-1225-XU74)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Gemin 2 Monoclonal Antibody (VS-1225-XU73)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CHMP2B Monoclonal Antibody (VS-1225-XU72)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-RanBP9 Monoclonal Antibody (VS-1225-XU71)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Arg2 Monoclonal Antibody (VS-1225-XU70)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ARF5 Monoclonal Antibody (VS-1225-XU69)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-RhoGAP Monoclonal Antibody (VS-1225-XU68)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-DLDH Monoclonal Antibody (VS-1225-XU67)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CSK Monoclonal Antibody (VS-1225-XU66)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CRKII Monoclonal Antibody (VS-1225-XU65)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ATP7b Monoclonal Antibody (VS-1225-XU64)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-FE65 Monoclonal Antibody (VS-1225-XU63)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-BCAR1 Monoclonal Antibody (VS-1225-XU62)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-DGAT1 Monoclonal Antibody (VS-1225-XU60)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-DLAT Monoclonal Antibody (VS-1225-XU59)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CDKN2C Monoclonal Antibody (VS-1225-XU58)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ABCE1 Monoclonal Antibody (VS-1225-XU57)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-PHAPI2/APRIL Monoclonal Antibody (VS-1225-XU56)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CBX2 Monoclonal Antibody (VS-1225-XU55)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-RING1 Monoclonal Antibody (VS-1225-XU54)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-BCL2L12 Monoclonal Antibody (VS-1225-XU53)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Desmoplakin Monoclonal Antibody (VS-1225-XU52)
-
- Species Reactivity: Human; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD97 Monoclonal Antibody (VS-1225-XU51)
-
- Species Reactivity: Human; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-COPS3 Monoclonal Antibody (VS-1225-XU49)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GOLPH4 Monoclonal Antibody (VS-1225-XU48)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GDF3 Monoclonal Antibody (VS-1225-XU47)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-BMP6 Monoclonal Antibody (VS-1225-XU46)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CLASP1 Monoclonal Antibody (VS-1225-XU44)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Eph receptor B1 Monoclonal Antibody (VS-1225-XU43)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-eIF4A1 Monoclonal Antibody (VS-1225-XU42)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ABHD5 Monoclonal Antibody (VS-1225-XU41)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-CD166 Monoclonal Antibody (VS-1225-XU39)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-Caspase-2 Monoclonal Antibody (VS-1225-XU37)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ERK1/2 Monoclonal Antibody (VS-1225-XU36)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-ErbB2 (HER2) Monoclonal Antibody (VS-1225-XU34)
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GRP78 BiP Monoclonal Antibody (VS-1225-XU33)
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
- Knock-Valid™ Rabbit Anti-GM130 Monoclonal Antibody (VS-1225-XU32)
-
- Species Reactivity: Human; Mouse; Rat; Monkey; Cow; Dog
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human; Mouse; Rat
- Application: For research on KD and KO validation.
-
- Species Reactivity: Human
- Application: For research on KD and KO validation.
Our customer service representatives are available 24 hours a day, from Monday to Sunday. Contact Us
Can't find the products you're looking for? Try to filter in the left sidebar.Filter By Tag
The reproducibility crisis in life science research is often rooted in poorly characterized antibodies that yield ambiguous results and cross-reactivity, compromising data integrity and therapeutic development. Creative Biolabs' KD/KO Validation Antibody Products are engineered to overcome these challenges through definitive genetic validation using CRISPR-Cas9 knockout and RNAi mediated knockdown models. We provide a complete validation service that delivers application-ready antibodies paired with side by side "Signal vs. No Signal" evidence. This rigorous approach ensures exceptional specificity and reliability, equipping researchers with validated tools to accelerate discovery and de-risk clinical translation.
Introduction
The widespread challenge of experimental reproducibility has brought to light the profound consequences of using inadequately validated antibodies. Genetic validation employing Knockout (KO) or Knockdown (KD) methodologies stands as the definitive standard for confirming antibody specificity. These approaches directly eliminate the target protein at the genetic or RNA level, enabling conclusive verification that an antibody's binding is exclusive to its designated epitope. Creative Biolabs utilizes this rigorous validation paradigm to deliver proteomic research tools with uncompromising reliability.
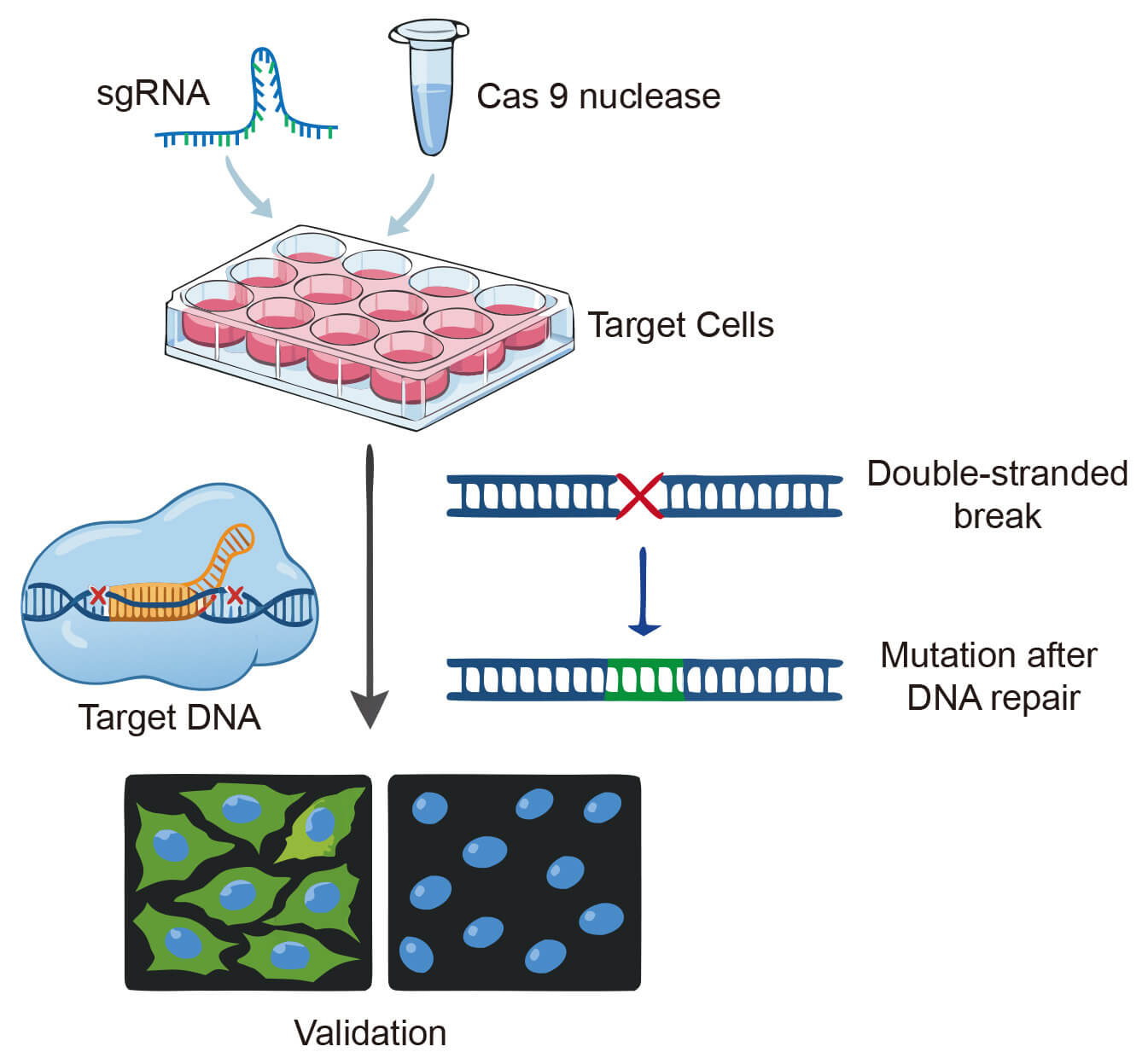 Fig.1 CRISPR-Cas9 knockout-mediated antibody specificity validation.
Fig.1 CRISPR-Cas9 knockout-mediated antibody specificity validation.
KD KO Validation Antibody Products at Creative Biolabs
In high-stakes biopharmaceutical research, the specificity of your primary detection tool determines the validity of your entire project. Creative Biolabs provides a suite of antibodies that have undergone rigorous genetic scrutiny, effectively removing the "guesswork" from proteomics. By using our validated products, you ensure that your therapeutic targets are correctly identified and that your lead compounds are interacting with the intended protein. This precision prevents the costly pursuit of false positives during the early stages of drug development.
Our Service Process
Required Starting Materials:
- The Target Protein Accession Number.
- Preferred Cell Line Background.
- The intended Application Parameters (e.g., WB, IHC, or Flow Cytometry).
Key Steps:
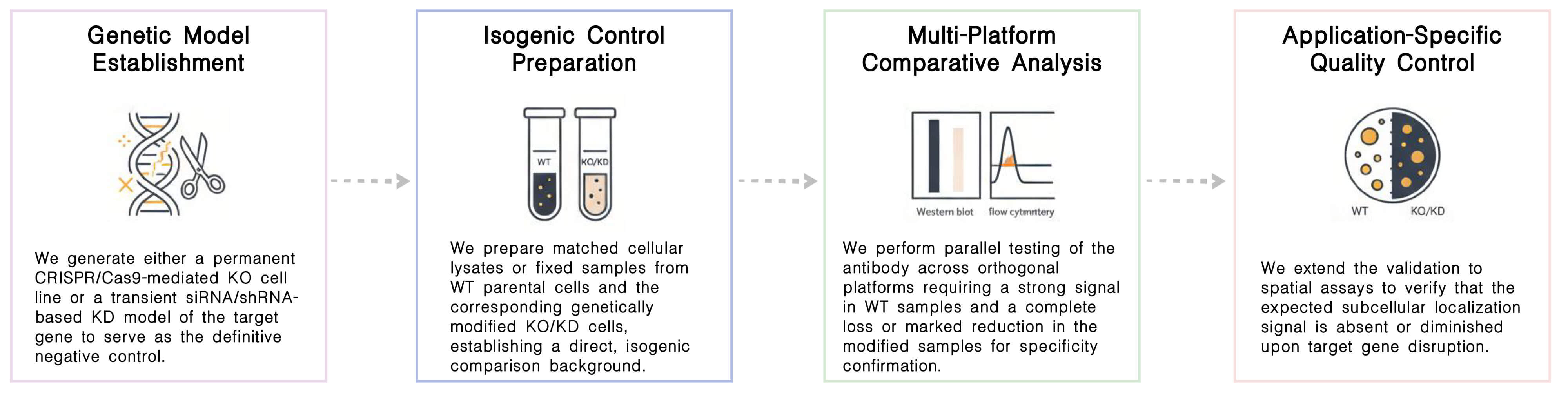
Final Deliverables: Upon completion of the validation process, you will be provided with a complete report detailing all experimental outcomes. This report includes high resolution documentation demonstrating specific signal detection, quantitative PCR data verifying successful gene silencing in knockdown experiments, and a formal Certificate of Analysis for the referenced antibody lot.
Key Advantages
- Complete Genetic Verification: Documentation of strain origin and cell line isogenic profiles assessed and approved by our expert Quality Assurance team.
- Stability Guarantee: We guarantee the stability of gene editing across cell banks and large-scale validation cultures to ensure consistent results.
- Custom Validation Optimization: We optimize codon usage and transfection protocols to facilitate the expression and validation of even the most challenging microorganisms and rare targets.
FAQ
What is the main difference between KO and KD validation for my project?
KO provides a permanent, binary "zero signal" control, while KD is used for essential genes where a complete knockout would be lethal to the cell. Both provide rigorous evidence of specificity.
Are these antibodies validated for Immunohistochemistry (IHC)?
Yes, many of our KO/KD validated antibodies are tested across multiple applications including IHC, WB, and Flow Cytometry. We encourage you to check the specific product page for your application.
Why Choose Us?
Creative Biolabs pioneers a transformative standard in antibody validation by advancing beyond conventional "peptide competition" methodologies. Our genetic validation framework screens every antibody against rigorously defined true negative controls, eliminating potential cross-reactivity from structural homologs.
Customer Reviews
"Our adoption of Creative Biolabs' KD/KO validated antibodies has resolved long-standing issues with antibody cross-reactivity in Western blotting. Specifically, a persistent 50 kDa band was confirmed as a homologous protein through KO lysate analysis, which definitively demonstrated the non-specific binding of our former antibody." Dr. Luc M.
"These antibodies were instrumental in securing acceptance of our manuscript. Comparative immunofluorescence imaging of wild type versus knockout cells supplied conclusive validation, directly addressing reviewer requirements." Sarah W.
"Incorporating KD validated antibodies from Creative Biolabs has markedly enhanced the reliability of our siRNA mediated knockdown assays. We observed a strict correspondence between mRNA depletion and reduction in target protein signal, confirming assay specificity." Prof. Rob T.
How to contact us?
Creative Biolabs provides the industry's most robust KD KO Validation Antibody Products, designed to solve the challenges of irreproducibility and cross-reactivity. Our genetic validation platform ensures that your scientific conclusions are based on accurate protein detection.
For detailed technical specifications or to discuss a custom validation project, please contact our team of specialists.
For Research Use Only. Not For Clinical Use.

