Antibody Process Impurity Measurement Service
Safeguarding Biologic Integrity Through Precise Impurity Profiling
During antibody biomanufacturing, a wide array of process-related impurities can be introduced, including host cell proteins (HCPs), DNA residues, endotoxins, leachables, and residual chromatography agents. These impurities, even at trace levels, may impact product safety, efficacy, or immunogenicity, and thus require careful quantification and control to meet regulatory expectations. At Creative Biolabs, our antibody process impurity measurement service is designed to provide thorough, accurate, and sensitive impurity profiling to support quality assurance and regulatory compliance across all stages of therapeutic antibody development.
Comprehensive Impurity Profiling: What We Offer
Our impurity analysis services encompass a broad spectrum of process-related contaminants that may persist after purification steps. We offer both platform-based and customized testing strategies for the following impurity classes:
- Host Cell Protein (HCP) Detection: Using ELISA or LC-MS/MS approaches tailored to the production cell line (e.g., CHO, HEK293).
- Host Cell DNA Quantification: Real-time PCR and PicoGreen assays to quantify residual DNA within regulatory thresholds.
- Endotoxin Analysis: LAL or recombinant factor C (rFC) assays for endotoxin detection in parenteral products.
- Residual Protein A and Other Ligands: HPLC and ELISA-based measurement of leached affinity chromatography ligands.
- Residual Detergents and Buffers: Assessment of surfactants (e.g., polysorbate 20/80), chelators, and buffer components.
- Chemical Leachables and Extractables: GC-MS and LC-MS profiling of materials derived from process equipment or packaging.
- Antifoam Agents and Media Components: Targeted quantification of additives carried over from upstream processing.
All assays are conducted under strict quality controls, and assay design is customized based on the antibody format, expression system, and purification process.
Precision-Driven Technologies at the Core
To ensure reliable and regulatory-compliant impurity quantification, we apply advanced technologies that are validated for sensitivity, specificity, and robustness:
- Quantitative PCR (qPCR): For ultra-sensitive DNA quantification down to picogram levels, with cell-line specific primers.
- LC-MS/MS Proteomics: Enables in-depth HCP profiling when ELISA kits are insufficient or unvalidated for novel hosts.
- Chromogenic and Fluorometric Endotoxin Detection: Compliant with FDA and USP guidelines for injectable biologics.
- Size-Exclusion and Reverse Phase HPLC: To measure residual ligands, detergents, and other process-related chemicals.
- Spectrophotometric and Fluorescence Assays: For general residual impurities and buffer component analysis.
- Extractables & Leachables Testing: Utilizing validated GC-MS and LC-MS workflows per ICH and USP standards.
Each assay is developed and executed with a focus on minimizing matrix interference and ensuring reproducibility across sample batches.
A Stepwise, Client-Centric Workflow
Our service workflow is designed to integrate seamlessly with your development timeline, ensuring transparency and scientific accuracy at every stage:
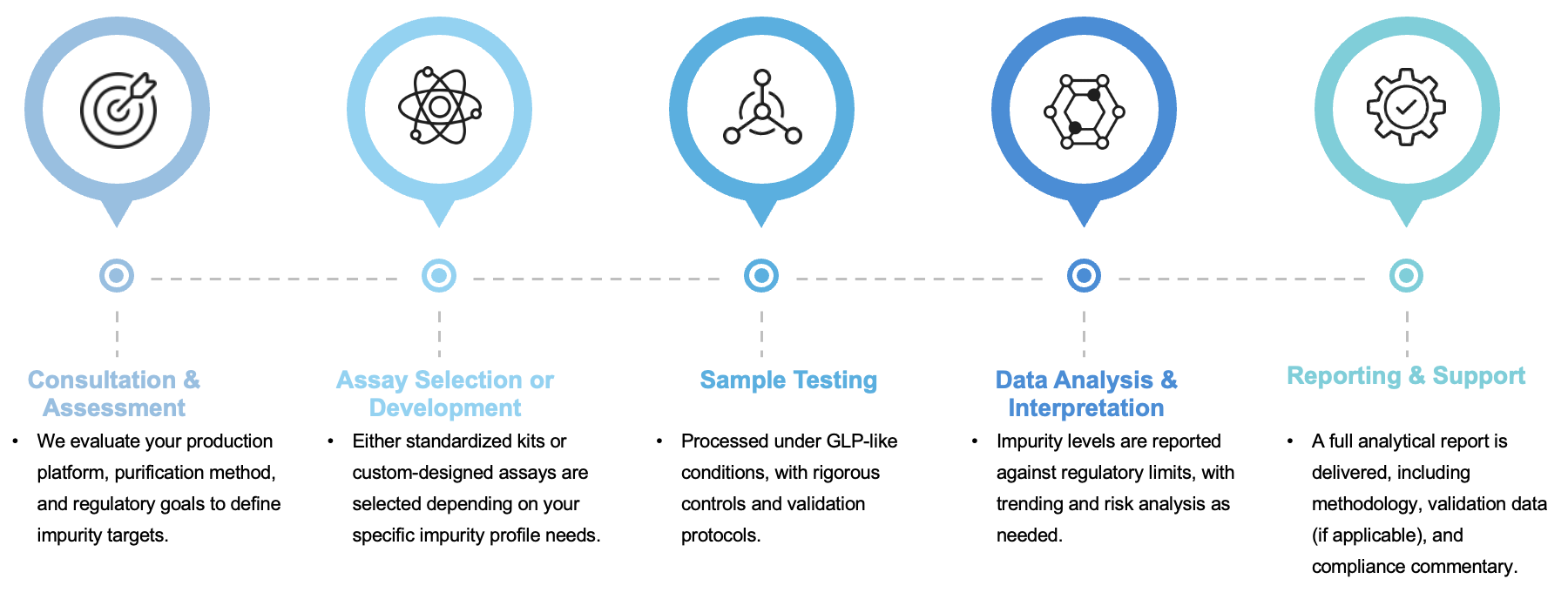
Why Choose Us: Scientific Rigor Meets Operational Flexibility
Creative Biolabs offers distinct advantages that position us as a trusted partner in impurity profiling for antibody therapeutics:
Broad Coverage Across Impurity Classes
One-stop testing for proteins, nucleic acids, endotoxins, residual ligands, and extractables.
Cell Line-Specific Expertise
Extensive experience with CHO, HEK293, NS0, and other expression systems enhances assay reliability.
Customized Assay Development
We build tailor-made assays when off-the-shelf kits do not meet specificity or sensitivity requirements.
Responsive Project Management
Our dedicated scientific and client service teams ensure timely communication and fast turnaround.
Collaborate with Confidence
With a robust portfolio of process impurity assays and a proven record in supporting preclinical and clinical-stage antibody programs, Creative Biolabs stands ready to help ensure the purity and compliance of your biologic product. Whether you're preparing for conducting routine release testing or other needs, we provide the scientific insight and technical precision needed to safeguard your therapeutic's integrity.
Reach out today to explore how our antibody process impurity measurement service can be integrated into your development strategy. Let us help you achieve quality benchmarks with confidence and clarity.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

