Antibody-Long-Circulating Liposome Production Service
In today's rapidly evolving landscape of antibody engineering, the need for antibody-long-circulating liposomes has never been more critical. This is where our top Antibody-Long-Circulating Liposome Production Services come into play. By fusing monoclonal antibodies, Creative Biolabs is capable of selectively binding to specific antigens, with a powerful liposome modification, they can exhibit a unique ability to target and destroy malignants with unparalleled precision.
Services
Long-circulating liposomes are a specialized type of liposome that have an extended circulation time in the body, allowing them to effectively transport drugs or other bioactive substances. These liposomes are typically modified on their surface with materials such as polyethylene glycol (PEG), which makes them less recognizable by the immune system, thereby prolonging their lifespan in the bloodstream.
Creative Biolabs offers a comprehensive range of long-circulating liposome services. Our services typically include the following components:
- Formulation Development: Design and optimization services for the preparation of long-circulating liposomes, including the selection of lipid components and the development of manufacturing processes.
- Characterization and Analysis: Utilizing various techniques (such as dynamic light scattering and transmission electron microscopy) to analyze the physicochemical properties of liposomes, assessing parameters like particle size, morphology, and drug encapsulation efficiency.
- Efficacy Evaluation: Assessing the efficacy of long-circulating liposomes in both in vitro and in vivo models, including studies on biodistribution, pharmacokinetics, and therapeutic effects.
- Preclinical Research: Providing relevant toxicology studies and safety assessments to long-circulating liposomes.
- Antibody Conjugation: Linking specific antibodies to long-circulating liposomes, enabling these liposomes to selectively target cells that express particular antigens. The conjugation can be achieved using chemical crosslinkers or other biotechnological methods. Our antibody-long-circulating liposomes can effectively deliver drugs (such as chemotherapy agents or biologics) to the target cells, enhancing therapeutic efficacy.
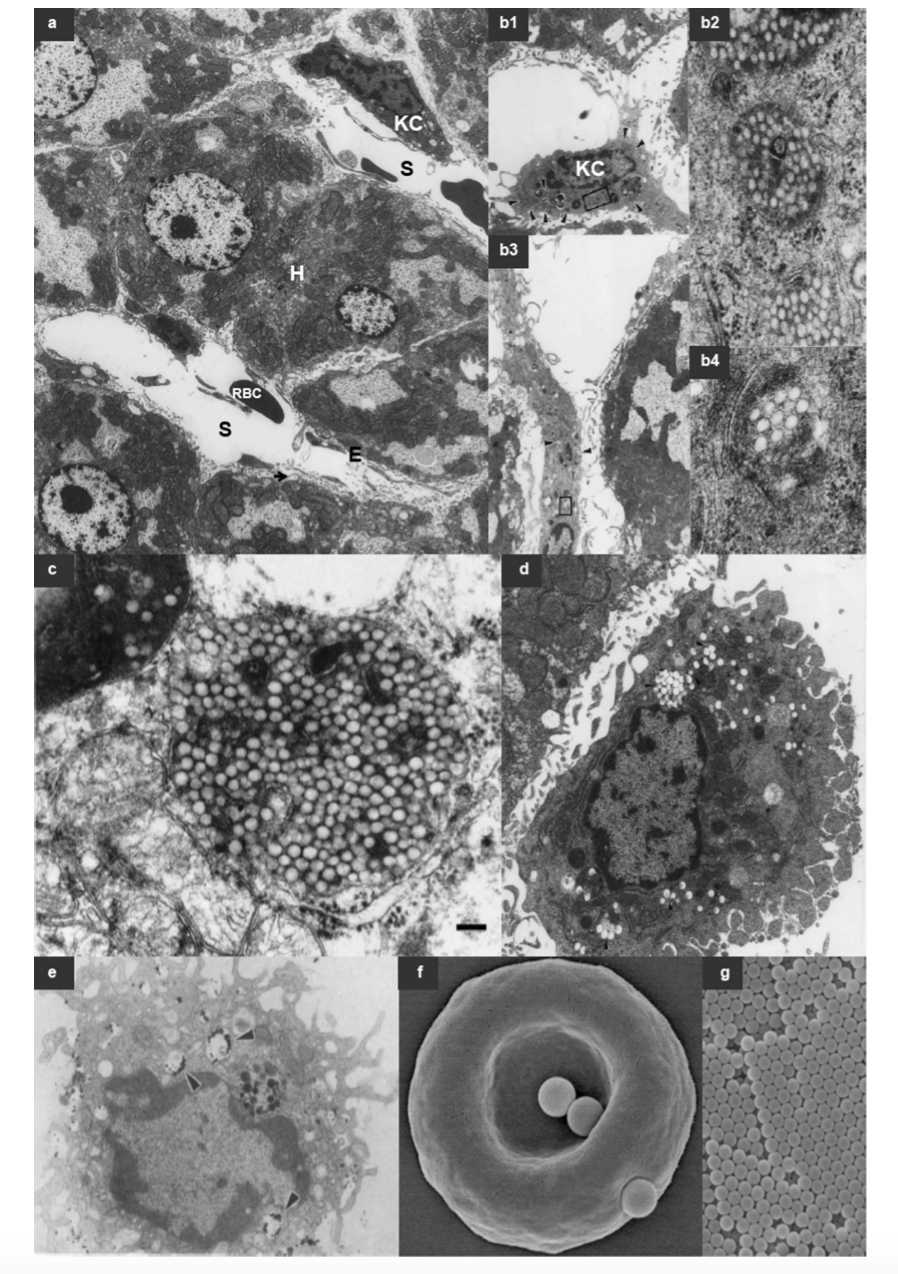 Fig.1 The Long Circulating Nanoparticles.1,3
Fig.1 The Long Circulating Nanoparticles.1,3
Procedures
Creative Biolabs utilizes specific antibody modification and long-circulating liposome production techniques to extend the circulation time of long-circulating liposomes in the bloodstream, thereby enhancing the bioavailability and targeting of drugs. Our antibody-long-circulating liposome products play a significant role in drug delivery systems, particularly in targeted therapies and the reduction of drug side effects. In our labs, we typically follow these steps:
1. Preparation of Liposomes
Material Selection: Choose appropriate phospholipids (such as phosphatidylcholine or hydrogenated phosphatidylcholine) and cholesterol.
Preparation Methods: Common methods include film hydration, sonication, and extrusion, which yield initial liposomal formulations.
Characterization: Assess the physicochemical properties of the liposomes, including particle size and morphology.
2. Antibody Selection and Purification
Antibody Selection: Select a suitable monoclonal antibody based on the target, typically opting for antibodies with high specificity and affinity.
Antibody Purification: Purify the antibody using methods such as affinity chromatography, ensuring its purity and activity.
3. Conjugation of Liposomes and Antibodies
Conjugation Reaction: Link the antibodies to the liposomes using chemical crosslinkers (EDC/NHS). Optimize reaction conditions to maximize the activity of the antibodies.
Evaluation of Conjugation Efficiency: Assess the conjugation efficiency of the antibodies to the liposomes using immunological techniques ( ELISA) or other methods.
4. Long-Circulation Modification
Modifying Lipid Composition: Selecting appropriate lipid components, such as saturated fatty acids and cholesterol, can enhance the stability of lipid bilayers and prolong circulation time in the bloodstream.
Structural Optimization: Optimize the composition and structure of the liposomes based on pharmacokinetic studies to enhance their stability and biocompatibility.
5. In Vitro and In Vivo Evaluation
In Vitro Assessment: Test the cellular uptake, cytotoxicity, and targeting ability of LCLCA in cell culture models.
In vivo assessment: Evaluate the pharmacokinetics, efficacy, and safety of the drug using animal models.
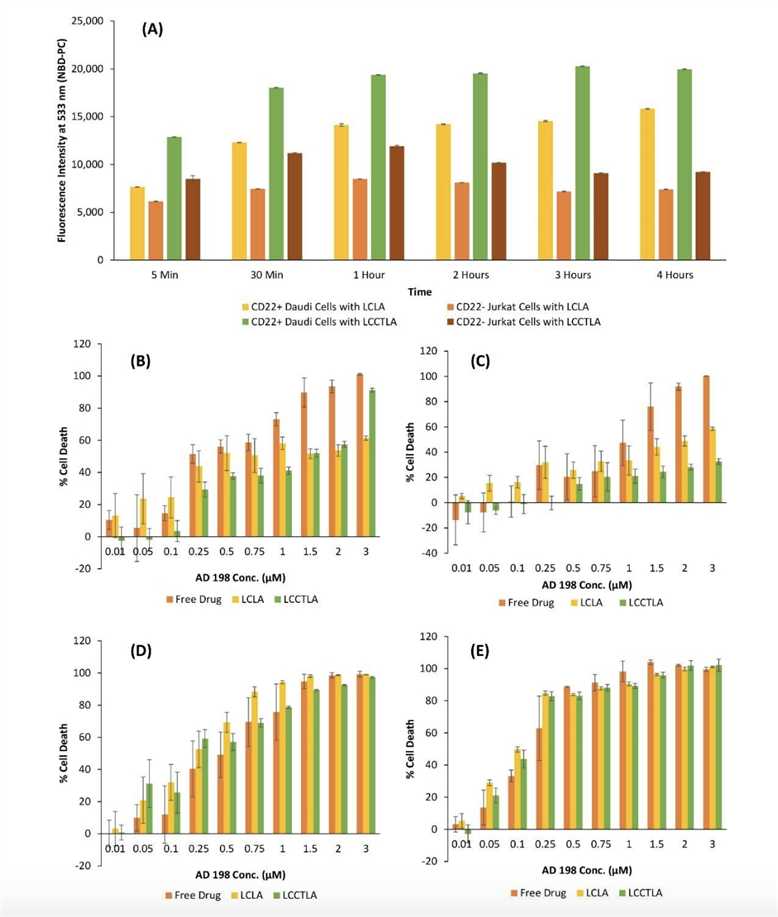 Fig.2 Cellular Uptake Analysis of LCLA and LCCTLA.2,3
Fig.2 Cellular Uptake Analysis of LCLA and LCCTLA.2,3
Case Study
Recently, Creative Biolabs has developed a long-circulating liposome formulation for enhanced stability and bioavailability. Tumor-specific antibodies (anti-HER2 or anti-CD20) have been modified with maleimide groups to facilitate conjugation to thiolated liposomes through thiol-maleimide click chemistry. We have finally evaluated the effectiveness of antibody-long-circulating liposomes in vitro and in vivo.
- In Vitro Evaluation Assay:
Cellular Uptake Studies
Fluorescence Microscopy/Flow Cytometry: These techniques can be employed to evaluate the uptake of antibody-conjugated liposomes by cancer cells. A higher uptake rate in comparison to non-conjugated liposomes would suggest effective targeting, as the antibodies enhance recognition and binding to specific tumor antigens.
Cytotoxicity Assays
MTT, XTT: These assays assess the cytotoxic effects of the drug delivered by the antibody-conjugated liposomes. Effectiveness can be determined by comparing the IC50 values (the concentration required to inhibit cell growth by 50%) between conjugated and non-conjugated liposomes. A lower IC50 indicates improved therapeutic efficacy due to targeted delivery.
Cell Signaling and Apoptosis Assay
Western Blotting/ELISA: Investigating downstream signaling pathways and apoptosis markers (e.g., cleaved caspases) offers insights into the mechanisms of action facilitated by liposomal formulation. An increase in apoptosis markers suggests effective drug delivery and action.
- In Vivo Evaluation Assay:
Biodistribution Studies
Radiolabeling or Fluorescent Imaging: Monitoring the distribution of antibody-conjugated liposomes in animal models (e.g., tumor-bearing mice) through imaging techniques will reveal a more significant accumulation of the conjugated liposomes in tumor tissues as opposed to normal tissues, thereby demonstrating their targeting capability.
Tumor Growth Inhibition
Tumor Xenograft Models: Administering antibody-conjugated liposomes in tumor-bearing mouse models enables the assessment of therapeutic efficacy through measurements of tumor growth.
Creative Biolabs with a wealth of knowledge in antibody development, linker chemistry, and long-circulating liposome formulation. We are committed to pushing the boundaries of what is possible in the antibody-long-circulating liposomes space. Our facilities are equipped with the latest tools for characterization and quality control, ensuring the safety and efficacy of your product. We are here to assist you don't hesitate to contact us for more information or to order.
- Haroon, H. B., et al. "A brief history of long-circulating nanoparticles." Advanced Drug Delivery Reviews 188 (2022): 114396.
- Mittal, Nivesh K., et al. "Formulation, development, and in vitro evaluation of a CD22 targeted liposomal system containing a non-cardiotoxic anthracycline for B cell malignancies." Pharmaceutics 10.2 (2018): 50.
- Distributed under Open Access license CC BY 4.0, without modification.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

