E. coli Cell-Free based Antibody Production and QC Service
E. coli systems are the most common cell-free expression systems. E. coli-based systems typically produce samples that are more homogeneous and have greater yields, making them ideal for structural studies. The protein yields of E. coli-based systems range from a few micrograms up to several milligrams per milliliter of reaction. E. coli cell extracts contain ribosomes, translational factors, and enzymes, all of which are essential for translation. Amino acids, ATP, GTP, an energy regeneration system, and a template are added to the cell lysate for in vitro translation. To promote the formation of disulfide bonds in generated antibodies, E. coli cell-free systems can be supplemented with mixtures of reduced and oxidized glutathione, disulfide bond isomerase C, or chaperones.
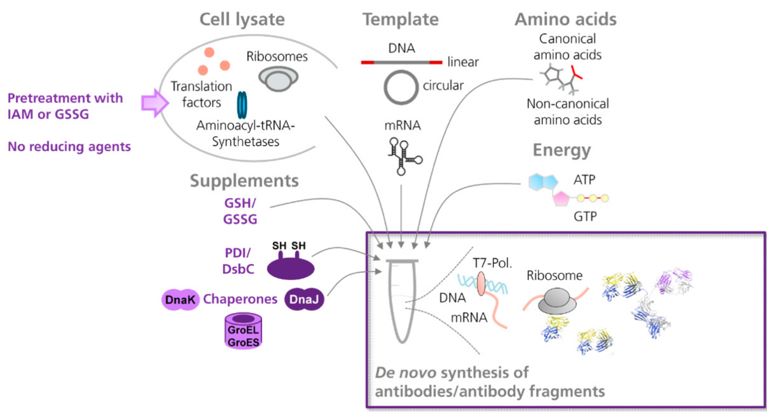 Fig.1. Recombinant Antibody Production in E. coli Cell-Free Systems.1
Fig.1. Recombinant Antibody Production in E. coli Cell-Free Systems.1
Advantages of E. coli Cell-Free Systems
- High protein yield
- Simple lysate preparation
- Cost-efficient
- Easy genetic engineering
- Well-established
Applications of E. coli Cell-Free Systems
- scFv Antibody Fragments Production in E. coli Cell-Free Systems
Numerous studies have shown that Escherichia coli cells can produce single-chain antibodies (scFv). Despite the high amount of scFvLH expression in vivo, the scFvHL was synthesized in a very low yield and no active product could be produced. While scFvLH is largely synthesized as a product with the anticipated molecular weight, scFvHL is produced with extra shorter fragments. This suggests that the reason for the low yield separation through the expression in vivo is an erroneous translation or ribonucleolytic cleavage of the transcript. The proportion of scFv rose from 25% to 90% in the cell-free expression with the presence of chaperones. However, there was little impact on the degree of biological activity and total expression.
- Fab Antibody Fragments Production in E. coli Cell-Free Systems
Fab has a larger potential for usage in clinical therapy and molecular diagnostics due to its binding affinity and stability equivalent to full-length antibodies. Cell-free protein synthesis is seen to be a viable alternate strategy that can get beyond the limitations of traditional in vivo gene expression techniques because of its excellent speed and adaptability. For instance, it has been demonstrated that disulfide linkages develop inside the cell-free produced proteins as a result of chemically treating the cell extract with a sulfhydryl-specific alkylating agent. These adaptable characteristics make it possible to use cell-free protein synthesis for the quick and parallel production of various antibody molecules.
- Full-length Antibodies Production in E. coli Cell-Free Systems
Antibodies are higher-order protein complexes that feature an interchain disulfide bridge-supported quaternary structure. Due to their versatility and ease of use, bacterial expression systems are commonly utilized for antibody discovery methods like phage display or ribosome display. This has led to the need for bacterial systems that are specifically designed for the folding of eukaryotic disulfide-bonded proteins. A process called Open-Cell Free Synthesis (OCFS) uses a linked transcription-translation system made from cell extracts of fast-diverging E. coli strains to produce full-length antibodies quickly.
Creative Biolabs can offer the most comprehensive services for antibody production. We can produce recombinant antibodies and antibody fragments in E. coli cell-free systems. Please contact us for more detailed information.
Reference
- Stech, Marlitt, and Stefan Kubick. "Cell-free synthesis meets antibody production: a review." Antibodies 4.1 (2015): 12-33. Distributed under Open Access license CC BY 4.0, without modification.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

