Insect Cell-Free based Antibody Production and QC Service
Antibodies are critical elements of detection in research, diagnosis, and treatment. Cell extracts from Escherichia coli have been used to make antibody fragments and antibody fragment fusion proteins. These lysates usually have high production yields, however, only a fraction of the generated antibodies might be in a functional state. It has been established that soluble and functional scFv molecules can be expressed using a cell-free translation system based on Spodoptera frugiperda (Sf21) cells. Translation factors, enzymes, and translationally active ribosomes are sourced from the cell extract. The lysate is distinguished by the existence of endogenous vesicles, which originate from the extracellular reticulum of the cultured insect cells. This enables target proteins to be translocated into the lumen of these vesicles through co-translation, or in the case of membrane proteins, to be embedded into microsomal membranes.
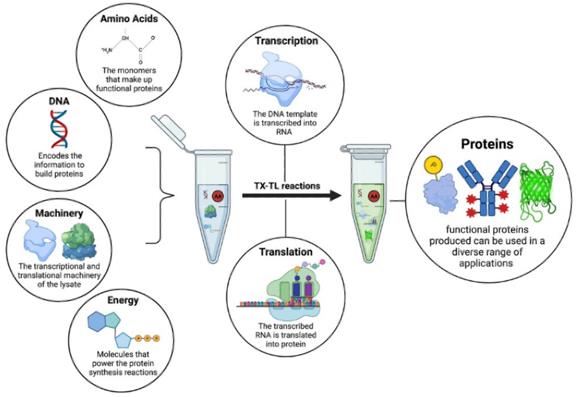 Fig.1. The components of a cell-free system.1
Fig.1. The components of a cell-free system.1
Features of Insect Cell-Free Systems
The insect cell-free system was produced using cultured Spodoptera frugiperda cells. The insect cell lysate does not remove the ER. Instead, the remaining components reorganize to form distinct microsomes. Protein translocation and embedding are made possible by the indigenous microsomes in the insect cell extract, which are translocationally active and produce a lipid membrane that is very similar to the natural one. There is evidence of modifications such as phosphorylation, lipidation, glycosylation, peptide cleavage, and disulfide bond formation. Eukaryotic proteins frequently exhibit these covalent changes, which include glycosylation and the creation of disulfide bonds, and are typically necessary for proper protein folding and function. Proteins were translocated into microsomes by mixing the target protein with the honeybee melittin signal peptide.
Advantages of Insect Cell-Free Systems
- Simple and quick preparation of cell lysate.
- Post-translational modifications could occur.
- There are endogenous microsomes accessible.
Applications of Insect Cell-Free Systems
The functional scFv antibody fragments were produced in the insect cell-free translation system. DNA templates of two distinct types were produced: scFv genes fused to the N-terminus of the melittin signal sequence and scFv genes lacking a signal sequence. Posttranslational modification of proteins requires their translocation into the ER. Consequently, to investigate whether the translocation of de novo produced scFv molecules would improve their functionality, scFv genes were N-terminally fused to the melittin signal sequence. De novo produced antibody fragments were detected in the autoradiograph as discrete and homogenous bands, exhibiting the anticipated migration pattern, and reached protein yields of 10–15 μg/mL.
Creative Biolabs provides recombinant antibody production services in Insect cell-free systems. Our one-stop cell-free protein production services, which are supported by cutting-edge technology platforms and a wealth of experience, can assist our clients in advancing their varied research and projects. Please feel free to contact us for more details.
Reference
- Brookwell, August et al. “Biotechnology Applications of Cell-Free Expression Systems.” Life (Basel, Switzerland) vol. 11,12 1367. 8 Dec. 2021, doi:10.3390/life11121367. Distributed under Open Access license CC BY 4.0, without modification.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

