Afuco™ Anti-Human TNF ADCC Recombinant Antibody (AVX-470), ADCC Enhanced
CAT#: AFC-601CL
Anti-TNF ADCC Enhanced Antibody (AVX-470) is an ADCC enhanced antibody produced by our Afuco™ platform. AVX-470 is a novel polyclonal antibody specific for human TNF used for Inflammatory bowel disease (IBD). IBD is a chronic inflammatory disease of the gastrointestinal tract, which is currently treated with injected monoclonal antibodies specific for tumor necrosis factor (TNF).


Specifications
- Host Species
- Cow
- Derivation
- Cow
- Type
- ADCC enhanced antibody
- Species Reactivity
- Human
- Applications
- Neut, FuncS
- Related Disease
- Ulcerative Colitis
Product Property
- Purity
- >95% as determined by analysis by SDS-PAGE
- Storage
- ≤1 year at -20°C.
Applications
- Application Notes
- AVX-470 and AVX-470m TNF-Binding and Neutralization Activities
The ability of AVX-470 and AVX-470m to bind to TNF was measured in a direct enzyme-linked immunosorbent assay (ELISA). Microtiter plates were coated with human or mouse TNF at 1 µg/mL and binding was detected using horseradish peroxidase-conjugated sheep anti-bovine antibody using standard techniques. Titers, expressed as µg/mL, were defined as the concentration of test article resulting in an absorbance value of 0.2. The ability of antibody to neutralize TNF was determined using the murine fibroblast cell line L929. These cells are killed by TNF, especially in the presence of actinomycin D, and a neutralizing anti-TNF antibody will prevent this cytotoxicity15. IC50 values were calculated as the concentration of AVX-470/AVX-470m resulting in 50% inhibition of TNF-mediated killing of L929 cells. Affinity of AVX-470A and AVX-470mA for TNF by Competition ELISA Serial dilutions of human or mouse TNF were incubated with defined levels of anti-TNF antibodies or control bovine Ig. The levels of TNF-specific antibodies used in these assays were previously determined to be in the linear portions of titration curves in direct ELISAs: AVX-470A (35 ng/mL), AVX-470mA (50 ng/mL), infliximab (2 ng/mL) and TN3 (TN3-19.12, 10 ng/mL). The antibody/TNF dilution mixtures were then added to the TNF-coated plates which were incubated, washed, incubated with horseradish peroxidase-labeled anti-bovine, human or hamster IgG secondary antibodies, washed and read on a plate reader. Data were analyzed using Gen5 Data Analysis Software and affinities were estimated as dissociation constants (KD) calculated from the amount of soluble TNF required to inhibit 50% of the binding of the antibodies to plate-bound TNF. Mouse models of colitis Animal experiments were conducted in the AAALAC-approved facility of Biomodels, LLC with ethical review approval from Biomodels' Institutional Animal Care and Use Committee. Colitis was induced in male C57BL/6 mice by exposure to 3% DSS in drinking water from Day 0 to Day 512 or by intrarectal administration of 4 mg TNBS in 50% ethanol on Day 0. Treatments with vehicle (saline), AVX-470m (1-10 mg/day), control Ig (3 or 10 mg/day), or prednisolone (3 mg/kg/day) were administered in volumes of 0.2 mL/mouse twice a day by oral gavage. Treatments with etanercept (10 mg/kg) were administered by intraperitoneal injection every other day. In prevention studies, treatment began one day before the induction of colitis and continued through day 3 for TNBS colitis or through day 12 for DSS colitis. In treatment studies, treatment began on day 6 and continued through day 19. Colitis severity was assessed using video endoscopy (Karl Storz Endoskope, Tuttlingen, Germany) a method that provides a robust clinical readout of disease severity14,16. Images were scored by a blinded observer: 0 = normal, 1 = loss of vascularity, 2 = loss of vascularity and friability, 3 = friability and mucosal erosions, and 4 = ulcerations and bleeding.
Target
Customer Review
There are currently no Customer reviews or questions for AFC-601CL. Click the button above to contact us or submit your feedback about this product.
Submit Your Publication
Published with our product? Submit your paper and receive a 10% discount on your next order! Share your research to earn exclusive rewards.
Related Diseases
Related Signaling Pathways
Downloadable Resources
Download resources about recombinant antibody development and antibody engineering to boost your research.
Product Notes
This is a product of Creative Biolabs' Hi-Affi™ recombinant antibody portfolio, which has several benefits including:
• Increased sensitivity
• Confirmed specificity
• High repeatability
• Excellent batch-to-batch consistency
• Sustainable supply
• Animal-free production
See more details about Hi-Affi™ recombinant antibody benefits.
Datasheet
MSDS
COA
Certificate of Analysis LookupTo download a Certificate of Analysis, please enter a lot number in the search box below. Note: Certificate of Analysis not available for kit components.
See other products for "TNF"
Select a product category from the dropdown menu below to view related products.
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOB-1076z | Mouse Anti-TNF Recombinant Antibody (clone 18C3) | ELISA, FC, FuncS | Mouse IgG1, κ |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NABL-051 | Recombinant Anti-human TNF VHH Single Domain Antibody | WB, ELISA, IHC, FC, FuncS | Llama VHH |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-010 | Anti-Human TNF Recombinant Antibody (TAB-010) | WB, FC, IP, ELISA, Neut, FuncS, IF | IgG1, κ |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-768 | Anti-TNF alpha Recombinant Antibody (TAB-768) | FuncS, IF, Neut, ELISA, FC, IP, ICC | IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-006 | Anti-Human TNF Recombinant Antibody (Infliximab) | IF, IP, Neut, FuncS, ELISA, FC, WB | IgG1 - kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-034-F(E) | Anti-Human TNF alpha Recombinant Antibody Fab Fragment (TAB-034-F(E)) | ELISA, FC, IP, FuncS, IF, Neut, IHC | Fab' |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-254 | Anti-Human TNF Recombinant Antibody (TAB-254) | Neut, ELISA, IF, IP, FuncS, FC, ICC | [V-kappa]2 - Fc |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-718 | Anti-TNFα Recombinant Antibody (TAB-718) | Neut, ELISA, IF, IP, FuncS, FC, ICC | F(ab')2 - G3 - kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-623CL | Bovine Anti-TNF Recombinant Antibody (TAB-623CL) | Neut | Bovine IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PABL-349 | Human Anti-TNF Recombinant Antibody (clone A2) | WB, ELISA, FuncS | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PSBL-349 | Human Anti-TNF Recombinant Antibody (clone A2); scFv Fragment | WB, ELISA, FuncS | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| PFBL-349 | Human Anti-TNF Recombinant Antibody (clone A2); Fab Fragment | WB, ELISA, FuncS | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-311CT | Anti-Human TNFα/RANKL Recombinant Antibody (8G12) | Neut, ELISA, FC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-311CT-S(P) | Anti-Human TNFα/RANKL Recombinant Antibody scFv Fragment (8G12) | Neut, ELISA, FC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-311CT-F(E) | Anti-Human TNFα/RANKL Recombinant Antibody Fab Fragment (8G12) | Neut, ELISA, FC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-110CQ | Anti-Human TNF Recombinant Antibody (CDP571) | ELISA | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-111CQ | Anti-Human TNF Recombinant Antibody (B5) | ELISA, WB | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-114CQ | Human Anti-TNF Recombinant Antibody (TAB-114CQ) | ELISA, FC, Inhib | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-115CQ | Human Anti-TNF Recombinant Antibody (TAB-115CQ) | ELISA, FC, Neut | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-118CQ | Anti-Human TNF Recombinant Antibody (TAB-118CQ) | FC, ELISA | Humanized antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-119CQ | Human Anti-TNF Recombinant Antibody (TAB-119CQ) | ELISA, BL | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-120CQ | Anti-Human TNF Recombinant Antibody (TAB-120CQ) | ELISA, Neut | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-121CQ | Anti-Human TNF Recombinant Antibody (TAB-121CQ) | ELISA | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-148CQ | Anti-Human TNF Recombinant Antibody | ELISA | Chimeric antibody (mouse/human) |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-402CQ | Human Anti-TNF Recombinant Antibody (TAB-402CQ) | ELISA | Chimeric (mouse/human) IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-412CQ | Anti-Human TNF Single Domain Antibody (TAB-412CQ), Research Grade Biosimilar | ELISA, Inhib | Single domain antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-413CQ | Anti-Human TNF Single Domain Antibody (TAB-413CQ), Research Grade Biosimilar | ELISA, WB, Neut | Single domain antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-414CQ | Anti-Human TNF Single Domain Antibody (TAB-414CQ), Research Grade Biosimilar | ELISA, WB, Neut | Single domain antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-415CQ | Anti-Human TNF Single Domain Antibody (TAB-415CQ), Research Grade Biosimilar | ELISA, WB, Neut, FunS | Single domain antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-148CQ-S(P) | Anti-Human TNF Recombinant Antibody scFv Fragment | ELISA | Chimeric antibody (mouse/human) |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| TAB-402CQ-S(P) | Mouse Anti-TNF Recombinant Antibody; scFv Fragment (TAB-402CQ-S(P)) | ELISA | Mouse scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| Gly-083LC | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Non-hyper galactosylated) | ELISA, FC | Human antibody |
| Gly-083LC-1 | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Non-hyper galactosylated) | ELISA, FC | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| Gly-085LC | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Sialylated) | ELISA, FC | Human antibody |
| Gly-085LC-1 | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Sialylated) | ELISA, FC | Human antibody |
| Gly-163LC | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Sialylated) | ELISA | Chimeric antibody (mouse/human) |
| Gly-163LC-1 | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Sialylated) | ELISA | Chimeric antibody (mouse/human) |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| Gly-086LC-1 | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Hyper-galactosylated) | ELISA, FC | Human antibody |
| Gly-087LC-1 | Recombinant Anti-Human TNF Antibody (Fc glycosylation/Hyper-galactosylated) | ELISA, FC | Human antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| Gly-088LC | Recombinant Anti-Human TNF Antibody (Fc glycosylation) | ELISA, FC | Human antibody |
| Gly-100LC | Recombinant Anti-Human TNF Antibody (Fc glycosylation) | ELISA | Chimeric antibody (mouse/human) |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOB-0229MZ | Recombinant Mouse Anti-Human TNF Antibody (clone G7C6) | ELISA, IHC | Mouse antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| BRD-0609MZ | Chicken Anti-Tumor Necrosis Factor-α Polyclonal IgY | Indirect ELISA, WB | Chicken antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2051CQ | Mouse Anti-TNF Recombinant Antibody (clone CBL589) | ELISA, ICC, Neut | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2052CQ | Mouse Anti-TNF Recombinant Antibody (clone CBL570) | WB, Neut | Mouse IgG2b |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2053CQ | Mouse Anti-TNF Recombinant Antibody (clone CBL571) | WB, Neut | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2054CQ | Rat Anti-Tnf Recombinant Antibody (NEUT-2054CQ) | Neut | Rat IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2055CQ | Rat Anti-Tnf Recombinant Antibody (NEUT-2055CQ) | Neut, FC | Rat IgD |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2057CQ | Rat Anti-Tnf Recombinant Antibody (NEUT-2057CQ) | Block, ELISA, IHC, WB | Rat IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2070CQ | Mouse Anti-TNF Recombinant Antibody (clone 4F4) | Block, ELISA, ICC, IF, IHC, WB | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2071CQ | Rabbit Anti-TNF Recombinant Antibody (clone 6E10) | Block, ELISA, IHC, WB | Rabbit IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| NEUT-2082CQ | Mouse Anti-TNF Recombinant Antibody (clone B-C7) | Block | Mouse IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOR-3602 | Rabbit Anti-Tnf Recombinant Antibody (clone DS3602AB) | ELISA, WB | Rabbit IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOR-4460 | Rabbit Anti-TNF Recombinant Antibody (clone SI554DS) | ELISA | Rabbit IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOR-4461 | Rabbit Anti-TNF Recombinant Antibody (clone SI555DS) | Neut | Rabbit IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOR-4462 | Rabbit Anti-TNF Recombinant Antibody (clone SI556DS) | FC | Rabbit IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| MOR-4463 | Rabbit Anti-TNF Recombinant Antibody (clone SI557DS) | FC | Rabbit IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0007-YC | Human Anti-TNF Recombinant Antibody (clone cA2) | ELISA, Neut | Chimeric (mouse/human) IgG1 |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0151-YC | Human Anti-TNF Recombinant Antibody (HPAB-0151-YC) | ELISA, Inhib, FuncS | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0007-YC-S(P) | Human Anti-TNF Recombinant Antibody (clone cA2); scFv Fragment | ELISA, Neut | Mouse scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0151-YC-S(P) | Human Anti-TNF Recombinant Antibody; scFv Fragment (HPAB-0151-YC-S(P)) | ELISA, Inhib, FuncS | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0007-YC-F(E) | Human Anti-TNF Recombinant Antibody (clone cA2); Fab Fragment | ELISA, Neut | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0151-YC-F(E) | Human Anti-TNF Recombinant Antibody; Fab Fragment (HPAB-0151-YC-F(E)) | ELISA, Inhib, FuncS | Human Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0046-FY-S(P) | Mouse Anti-TNF Recombinant Antibody (clone 1B7); scFv Fragment | Neut | Mouse scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0046-FY-F(E) | Mouse Anti-TNF Recombinant Antibody (clone 1B7); Fab Fragment | Neut | Mouse Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0675-CN-S(P) | Human Anti-TNF Recombinant Antibody; scFv Fragment (HPAB-0675-CN-S(P)) | ELISA, Neut | Human scFv |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| HPAB-0675-CN-F(E) | Human Anti-TNF Recombinant Antibody; Fab Fragment (HPAB-0675-CN-F(E)) | ELISA, Neut | Humanized Fab |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-768 | Afuco™ Anti-TNF ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-768) | FuncS, IF, Neut, ELISA, FC, IP | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-006 | Afuco™ Anti-TNF ADCC Recombinant Antibody, ADCC Enhanced (AFC-TAB-006) | IF, IP, Neut, FuncS, ELISA, FC | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| AFC-TAB-010 | Afuco™ Anti-TNF ADCC Recombinant Antibody (AFC-TAB-010), ADCC Enhanced | FC, IP, ELISA, Neut, FuncS | ADCC enhanced antibody |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0424-XY256 | AbPlus™ Anti-TNF Magnetic Beads (4H31) | IP, Protein Purification |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0724-YC1214 | AbPlus™ Anti-TNF Magnetic Beads (VS-0724-YC1214) | IP, Protein Purification |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0125-FY32 | Human Anti-TNF (clone BPC1500) scFv-Fc Chimera | ELISA, Neut | Human IgG1, scFv-Fc |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0225-XY215 | CytoStream™ Mouse Anti-TNF Recombinant Antibody (clone MAb11) | FC | Mouse IgG1, kappa |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0225-XY216 | CytoStream™ Armenian Hamster Anti-TNF Recombinant Antibody (clone TN3-19.12) | ELISA, FC, Neut, IP, WB | Armenian Hamster IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0325-XY2313 | Anti-TNF Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY7348 | Anti-Mouse TNF Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY7352 | Anti-Human TNF Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY7353 | Anti-Rat TNF Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0525-XY7355 | Anti-Fish TNF Immunohistochemistry Kit | IHC |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0625-YC314 | Recombinant Anti-TNF Eliminating Antibody, pH-Sensitive (VS-0625-YC314) | Antigen-Sweeping In Vivo. | Human IgG |
| CAT | Product Name | Application | Type |
|---|---|---|---|
| VS-0825-YC332 | SmartAb™ Recombinant Anti-TNF pH-dependent Antibody (VS-0825-YC332) | IF, Neut, ELISA, FC, IP, ICC | Mouse IgG1 |
Popular Products

Application: Neut, ELISA, IF, IP, FuncS, FC, IHC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: WB, IF, IP, Neut, FuncS, ELISA, FC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: IP, IF, FuncS, FC, Neut, ELISA, ICC

Application: IP, IF, FuncS, FC, Neut, ELISA, ICC

Application: WB, FuncS, IF, Neut, ELISA, FC, IP

Application: ELISA, IP, FC, FuncS, Neut, IF, ICC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: IP, IF, FuncS, FC, Neut, ELISA, ICC

Application: IF, IP, Neut, FuncS, ELISA, FC, ICC

Application: ELISA, WB, BLI, SPR
For research use only. Not intended for any clinical use. No products from Creative Biolabs may be resold, modified for resale or used to manufacture commercial products without prior written approval from Creative Biolabs.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.





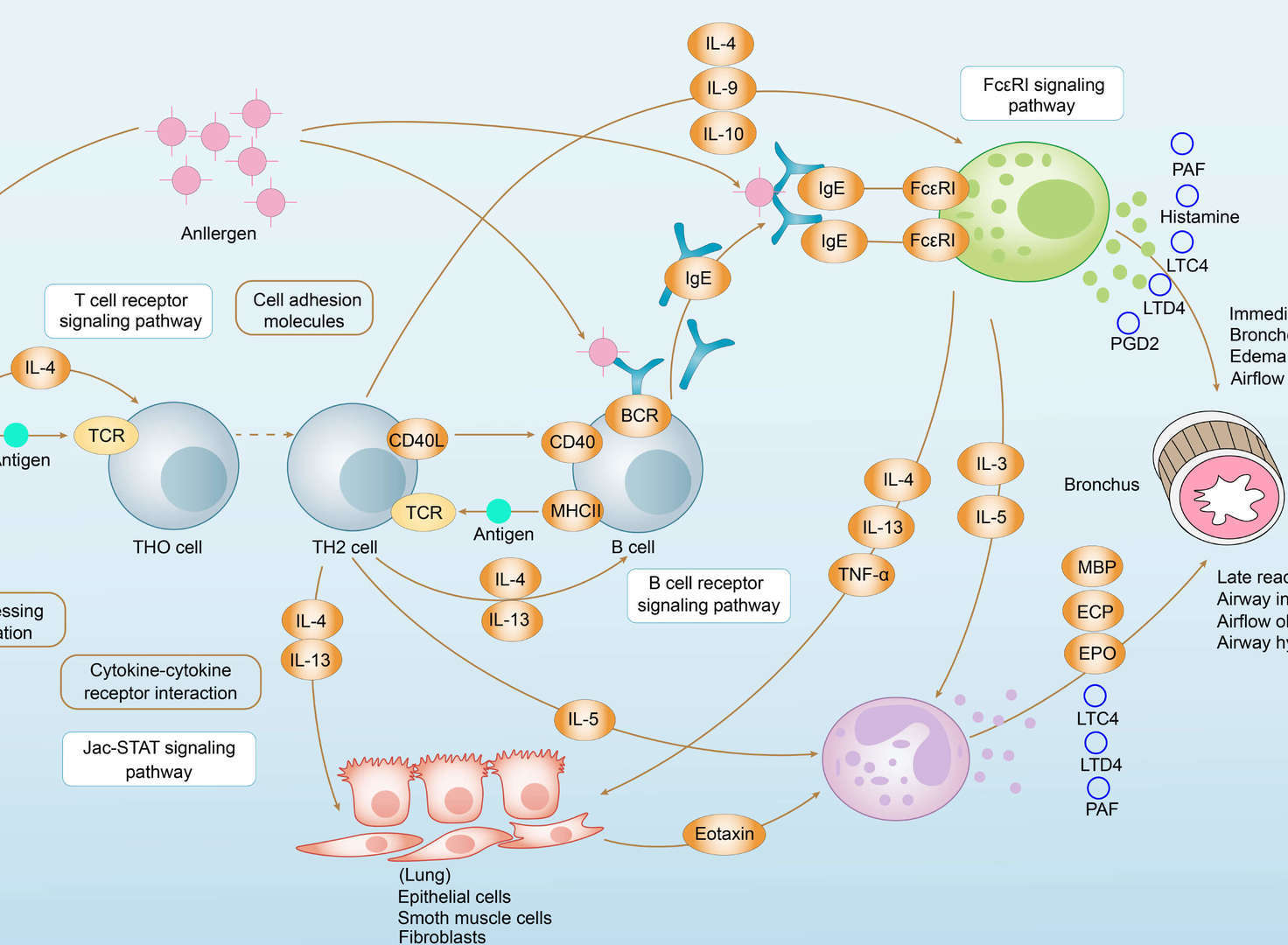 Asthma
Asthma
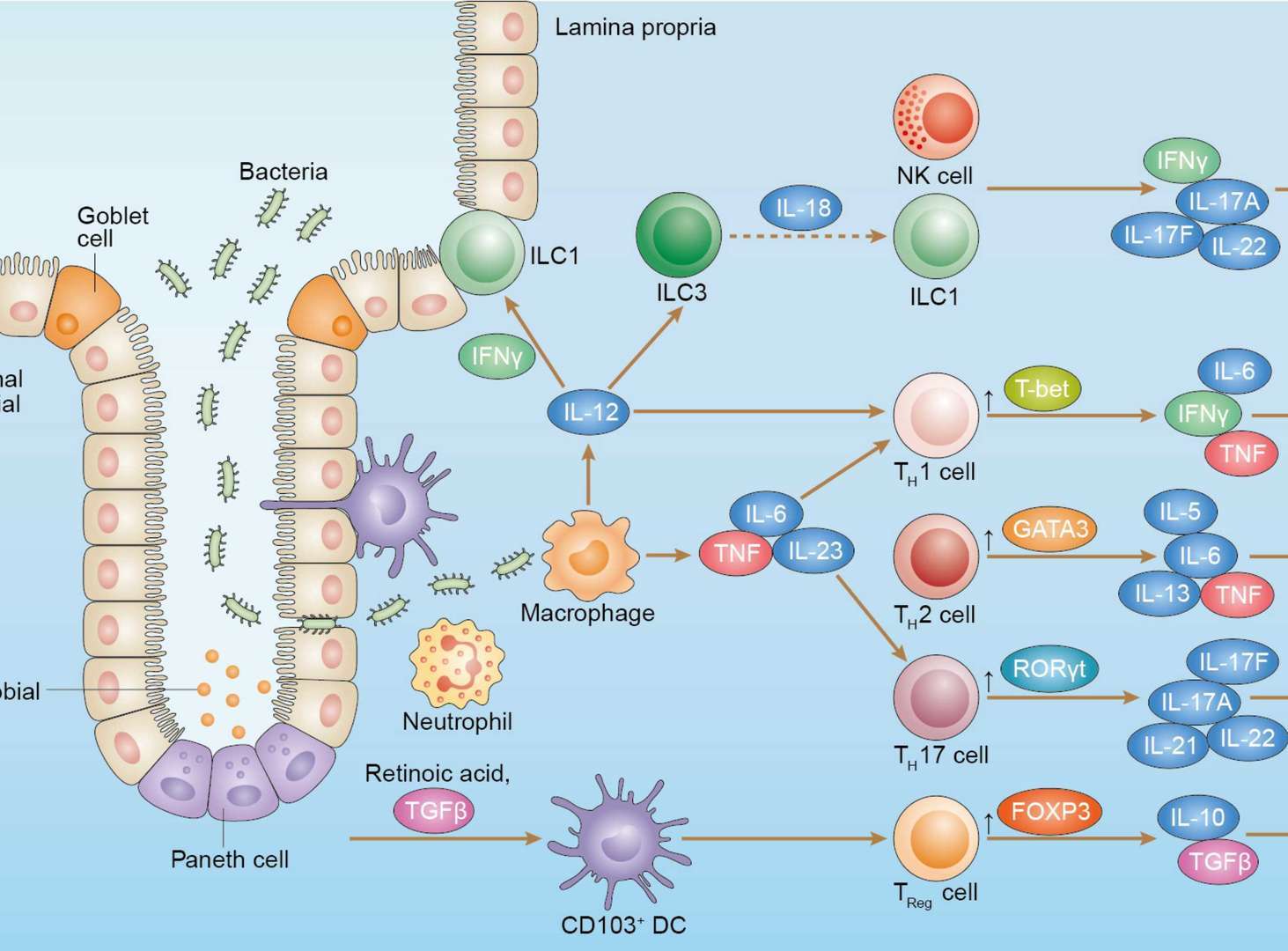 Inflammatory Bowel Diseases
Inflammatory Bowel Diseases
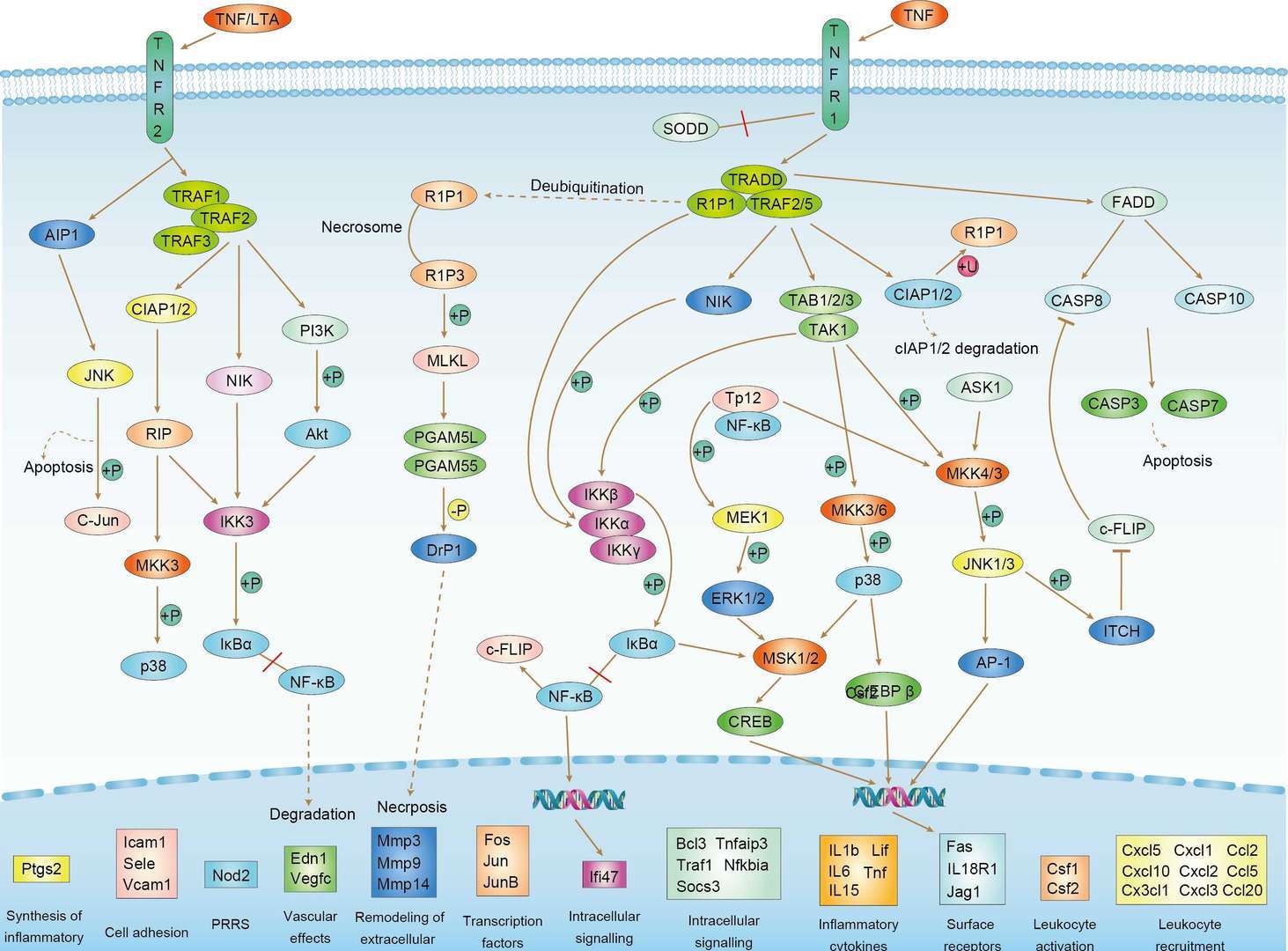 TNF Signaling Pathway
TNF Signaling Pathway










