Bavituximab Overview
Introduction of Bavituximab
Bavituximab is a human-mouse chimeric IgG1 monoclonal antibody (mAb), which binds to a basic component of the cell structure called a phospholipid that is exposed only on the surface of tumor blood vessel cells or on cells infected with certain viruses. Bavituximab was developed for the treatment of non-small cell lung cancer (NSCLC), breast cancer, pancreatic cancer, hepatocellular carcinoma and cell infection such as hepatitis C. However, there is currently no relevant clinically approved information disclosure.
Mechanism of Action of Bavituximab
Under normal cellular homeostasis, phosphatidylserine (PS) is an anionic membrane phospholipid preferentially located on the inner leaflet of the plasma membrane. This asymmetry is maintained by ATP-dependent aminophospholipid translocases that catalyze the transfer of aminophospholipids from the external to the internal plasma membrane. In certain physiologic states, such as apoptosis and cell activation, PS asymmetry is disrupted, resulting in exposure of PS on the external membrane leaflet. This process occurs due to inactivation of aminophospholipid translocases or the activation of scramblases and ABC floppases. Stresses within tumors, such as hypoxia and acidity, also trigger these events through the generation of reactive oxygen species. Exposed PS in the cellular microenvironment restricts the production of pro-inflammatory cytokines and increases the production of anti-inflammatory cytokines. This role is consistent with PS exposure during apoptosis, a class of cell death that does not induce inflammatory responses. In addition, PS exposure suppresses immune responses by reducing dendritic cell maturation and antigen presentation. Bavituximab is a human-mouse chimeric IgG1 monoclonal antibody that targets PS by recognizing the PS-binding protein β2 glycoprotein-1 (β2GP1). Specifically, by cross-linking two β2GP1 molecules at surface of cells with exposed PS, Bavituximab localizes to tumors. Once bound, Bavituximab activates host effector (immune) functions, such as antibody dependent cellular cytotoxicity (ADCC), resulting in tumor vessel destruction. Additionally, Bavituximab conveys anticancer effects by reversing the immune and inflammatory suppression normally exerted by tumors. In normal cells, including vascular endothelium, PS is segregated to the inner membrane leaflet and is therefore not available for antibody binding. In contrast, the exposure of PS on cancer cells and vascular endothelial cells in the tumor microenvironment permits this antibody to target the tumor microenvironment. This targets the bavituximab to the malignant cells and potentially minimizes unwanted side effects.
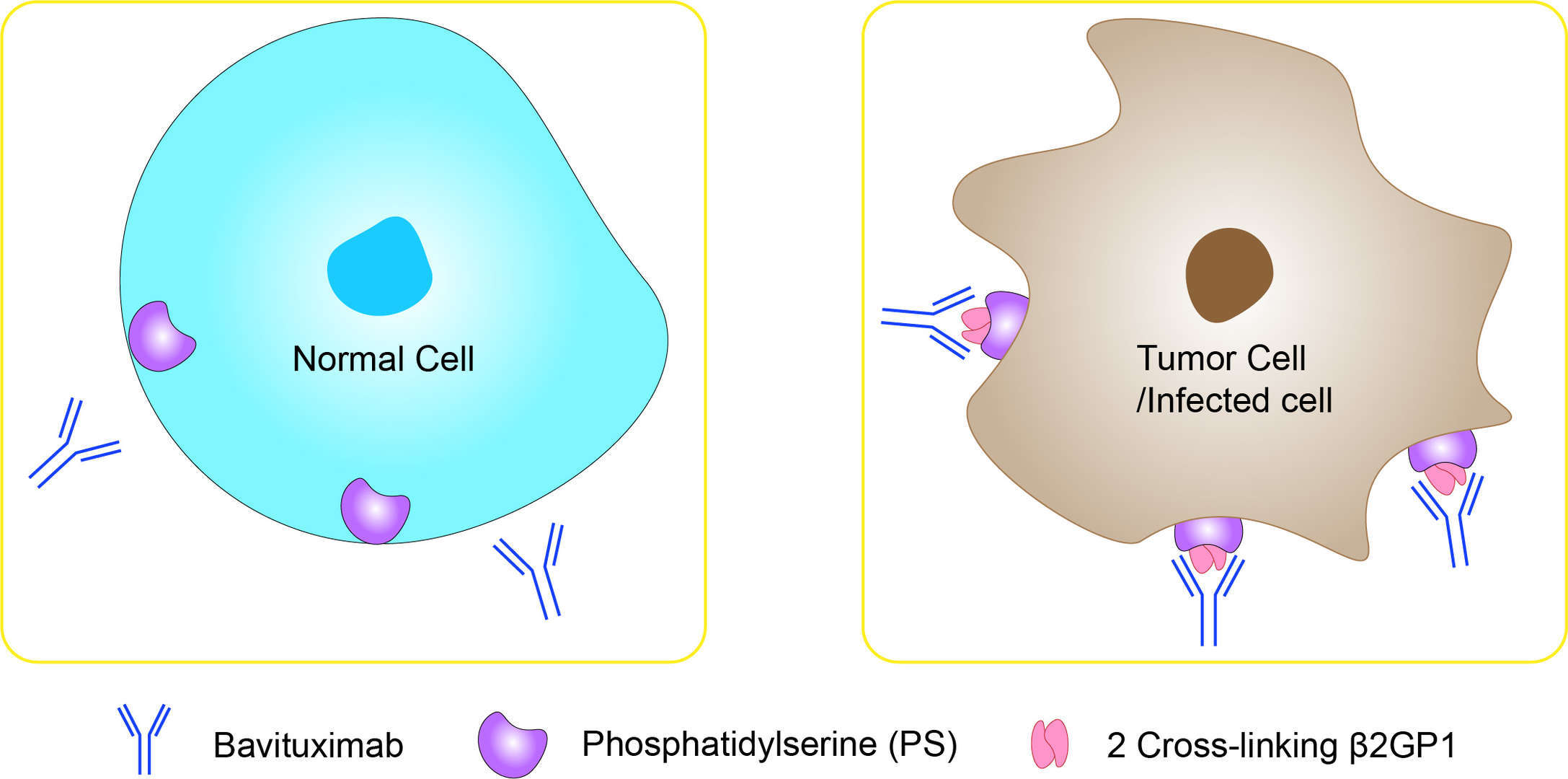 Fig 1. Mechanism of Action of Bavituximab
Fig 1. Mechanism of Action of Bavituximab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

