Immunohistology-Resin Section Protocol & Troubleshooting
In addition to traditional paraffin and frozen sections, resin sections are also a good choice for commonly used sections for immunohistochemistry. Compared to the two traditional protocols, the resin embedding procedure is similar to but slightly different from paraffin embedding. And it also offers excellent resolution, the advantage of good tissue morphology preservation and the ability to obtain multiple sections from a small biopsy sample.
Resin embedding is a viable alternative to paraffin and freezing and can overcome a number of technical problems. Creative Biolabs describes resin embedding and immunostaining protocols, provides problems and solutions that may be encountered in resin sectioning protocols, and assists you in obtaining sections with improved morphology for immunoassays.
Solutions and Reagents
| Stages | Solutions and Reagents |
| Section Preparation | Washing buffer, fixative, ethanol solution, resin solution |
| Antibody Staining | Blocking buffer, primary antibody, labeled secondary antibody, dilution buffer, washing buffer, incubation buffer, chromogen |
| Mounting | Mounting solution, washing buffer |
Immunohistology-Resin Sections Procedure
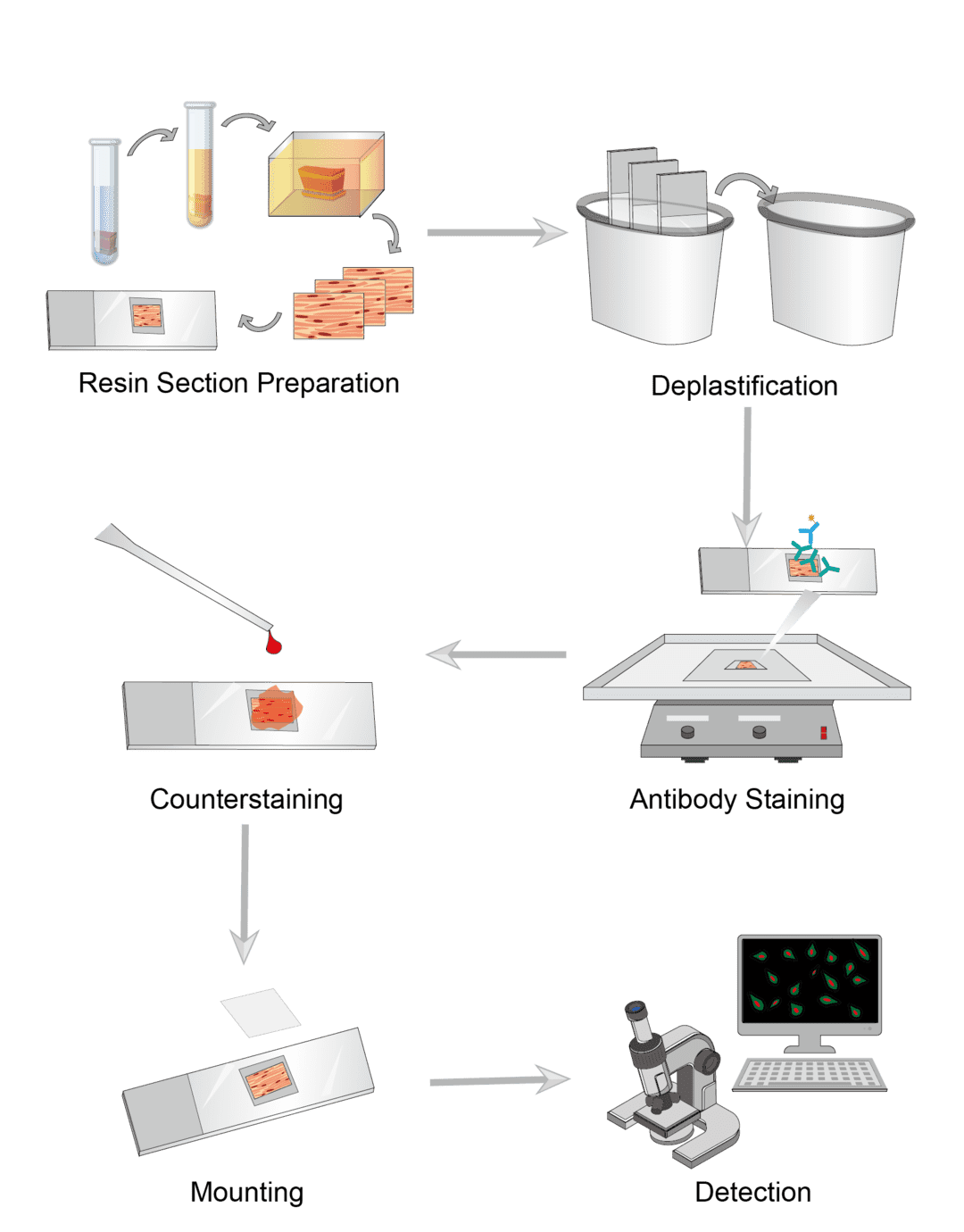
Prepare freshly dissected tissues and wash and cut for use. Soak the tissue with fixative for fixation. The recommended fixative for resin embedding is glutaraldehyde. Wash the tissues with buffer at the end of fixation. Dehydrate the tissues by soaking them several times with gradient concentrations of ethanol. Then use the resin instead of alcohol for infiltration. The resin solution must be prepared immediately prior to use. Pour the resin into the mold and then transfer the tissue quickly to the mold for polymerization at 60°C. Trim all excess resin around the sample, section with a sharper special blade. And transfer the section to a slide and dry at room temperature until ready to use.
Before performing immunohistochemical labeling, we need to desplast the resin-embedded sections. For each stage of deplastification, place the slide in a glass chamber containing different solutions, depending on the resin composition. Soak several times to remove and then wash.
Select the ideal blocking reagent and incubate the sample with the blocking buffer. Incubation time and temperature are set on a case-by-case basis. When finished, wash the slide and remove the blocking buffer. For direct labeling, dilute the primary antibody appropriately and transfer the slide to incubation buffer containing the primary antibody for incubation. Then wash the slides. For indirect assays, transfer the sections to incubation buffer containing secondary antibody and continue incubation. Wash the slide after completion to remove excess solution. Add the appropriate dilution of chromogenic agent to the slides and incubate away from light.
The counterstaining step is optional. Counterstaining provides contrast with antibody staining to better distinguish the target signal. Select the ideal counterstains, incubate the sections, and rinse upon completion.
After all staining, use the appropriate sealing solution to mount coverslips and seal the samples. Mounted slides can be stored permanently at room temperature. Observe the color of the antibody stain in the tissue section under the microscope.
Troubleshooting
A variety of problems often arise during experiments, but experience allows us to quickly identify and resolve them. Below is a summary of common problems that may arise in resin sections for immunohistochemical analysis, for our customers to refer to in order to solve the problems.
Tissue section problem
- Uncured resin problem. This is caused by the resin and curing agent not being well catalyzed. This means that the two parts are either not mixed sufficiently or the mixing ratio is not correct. To avoid uncured resin, you should use fresh resin and curing agent and mix equal amounts of resin and hardener thoroughly with exact measurements.
- Resin bubble problem. Please make sure to use fresh resin and hardener as much as possible, or use a high-quality resin product that contains a defoamer.
- Uneven resin problem. The resin is naturally leveled. When an uneven resin surface occurs, you should make sure that you are working on a level table and when using enough resin.
No or inadequate staining
- Antigen causes. If the test slide is inadequately stained or unstained, while the positive control slide is adequately stained, the antigen may be the problem. The antigen is not present in the test tissue or is present but at a level below the limit of detection. Consider using an amplification procedure or increasing the primary antibody concentration, incubation time or temperature.
- Antibody causes. If the positive control is weak or unstained and the test slide is also weak or unstained, the antibody may be the cause. Check the antibody, including conditions such as dilution concentration, incubation time and temperature. You need to be careful that the secondary antibody and primary antibody are compatible. Because the test requires the use of a secondary antibody that will interact with the primary antibody.
- Reagent causes. Check and make sure that the buffer solution is not contaminated, and that the wash time is too long can also cause a reduced signal. It is necessary to check the relevant reagents such as buffers, chromogenic agents. Need to pay attention to include expiration dates, storage parameters, pH and order of use.
- Microscope causes. Check microscope parameter settings and increase the exposure time of the camera.
High background
- Section causes. Consider first if the tissue section is too thick and thinner sections can be prepared. Then check if sections are adequately fixed or if necrosis and autolysis are present. If any of these conditions are present, avoid sampling necrotic areas and ensure that the tissue is properly fixed. As well, inappropriate or too thick section adhesives can affect the background. Throughout the process, check and ensure that tissue sections are not dry.
- Deplastification causes. You should repeat the deplastification step several times to completely remove the resin from the sample.
- Blocking causes. Possible reasons for this are inadequate blocking of endogenous enzyme activity, biotin or protein during the blocking phase. You can increase the concentration of blocking agent or use a different blocking agent. In addition, you need to be aware that the blocking buffer should be made from a blocking serum of the same species.
- Primary antibody causes. Improper antibody concentration causes high background. You can re-titrate primary antibody and select appropriate concentration. Check the primary antibody incubation time, too long time can also cause this problem. You can shorten the incubation time. As well as ensure that sections are cleaned and no antibody solution remains.
- Secondary antibody causes. As with primary antibody, check secondary antibody and label concentration and incubation time. It is important to note that the secondary antibody chosen needs to be unbound to tissue immunoglobulins. As well as ensure that sections are cleaned and no antibody solution remains.
- Chromogen causes. If the chromogen concentration is too high and the reaction time is too long, you need to reduce the chromogen concentration and shorten the incubation time. It is also possible that the counterstain is masking the IHC reaction and we suggest you to replace a different counterstain. As well as ensure that the sections are cleaned and no excess stain remains.
Want to learn more about resin sectioning? Contact us for tips and tricks for all your immunohistology experiments.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

