Dezamizumab Overview
Introduction of Dezamizumab
Dezamizumab is a fully humanized recombinant monoclonal IgG1 anti-serum amyloid P component (SAP) antibody, with potential anti-amyloid activity. It was developed for the treatment of AL amyloidosis (immunoglobulin light chain amyloidosis). The small-molecule drug, miridesap, depletes circulating SAP but leaves some SAP in amyloid deposits. This residual SAP is a specific target for dezamizumab, a fully humanized monoclonal IgG1 anti-SAP antibody that triggers immunotherapeutic clearance of amyloid. Studies have reported safety and clinical proof-of-concept results for miridesap followed by the fully humanized monoclonal anti-human SAP antibody, dezamizumab, in patients with different forms of systemic amyloidosis. Infusion of anti-SAP antibody triggered transient early inflammatory cytokine production and an acute phase response, followed by substantial plasma C3 depletion, but there was no new or increased renal or other organ dysfunction. Amyloid clearance from the liver was associated with improved liver function tests. However, maximal removal of amyloid from all organs and tissues, which will be required for optimal clinical efficacy, was not achieved with just a single dose of anti-SAP antibody.
Mechanism of Action of Dezamizumab
The extracellular accumulation of amyloid fibrils disrupts the architecture and function of affected tissues and organs in systemic amyloidosis, causing fatal disease. Systemic amyloid deposits usually do not elicit inflammation or the physiological phagocytic mechanisms that normally clear extracellular protein and cellular debris. Current management of systemic amyloidosis comprises support for damaged amyloidotic organs, coupled with attempts to reduce production of amyloidogenic proteins. Even if fibril precursor abundance is sufficiently reduced, thereby arresting new amyloid accumulation, existing amyloid is cleared slowly and variably. Efforts to develop targeted immunotherapeutic clearance of amyloid are therefore underway. Human serum amyloid P component (SAP) from the plasma is present in all human amyloid deposits due to its avid but reversible binding to amyloid fibrils of all types. The small-molecule drug, miridesap, previously known as CPHPC, swiftly depletes circulating SAP but leaves some SAP in amyloid deposits. The residual SAP acts as a specific antigen target for therapeutic anti-SAP antibodies that can bind to it and thereby trigger amyloid removal. Plasma SAP depletion by miridesap is essential before anti-SAP antibody administration to avoid formation of potentially proinflammatory circulating immune complexes. In humanized murine models, binding of anti-SAP antibodies to amyloid activates the classical complement pathway and opsonizes the deposits with fixed complement C3, attracting and engaging macrophages that fuse into multinucleated giant cells uniquely able to surround, engulf, and destroy large complement opsonized objects.
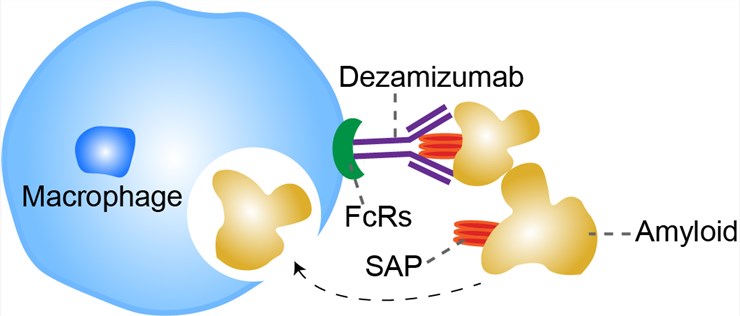
Fig 1. Mechanism of Action of Dezamizumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

