Vβ 8 & M2 BsAb Overview
Introduction of Vβ 8 & M2 BsAb
Vβ 8 & M2 BsAb, also called recombinant anti-TCR (Vβ 8) & anti-M2 bispecific antibody, is a bispecific fusion protein with four binding sites, two sites of which bind to TCR (Vβ 8) antigens and the other two bind to influenza virus M2 protein. Bispecific antibody, also known as bifunctional antibody or bivalent antibody, can specifically bind two different antigens at the same time. Because of its specificity and bifunctionality, bispecific antibody has become a research hotspot in the field of antibody engineering. Both chains of recombinant anti-TCR (Vβ 8) & anti-M2 bispecific antibody come from mouse. This BsAb can retarget of T cells to virus-infected cells. It was designed for the treatment of influenza virus infection.
Mechanism of Action of Vβ 8 & M2 BsAb
Vβ 8 & M2 BsAb was studied as early as 1996 to develop a defense to influenza virus infection. One chain of Vβ 8-M2 BsAb can bind to T cell surface receptor (TCR), and the other chain can target influenza virus protein. TCR is a heterodimer composed of two different peptide chains, α and β peptide chains. Each peptide chain can be divided into variable region (V region), constant region (C region), transmembrane region and cytoplasmic region. TCR Vβ 8 is a variant of the TCR β chain which is heterodimerized with TCR α chain and expressed on a subset of T cells. The cross-linking of TCR Vβ 8 with staphylococcal enterotoxin B (SEB) can lead to the activation and proliferation of a large number of T cells and the release of a large number of cytokines. The mechanism of Vβ 8 & M2 BsAb is that it binds to influenza virus protein (hemagglutinin M2) and T cell receptor, and kills influenza virus infected cells and inhibits virus replication in vitro by T cells activated by staphylococcal enterotoxin B (staphylococcal enterotoxin B). Although the replication of influenza viruses is significantly inhibited, the influenza virus system does not appear to be suitable for bispecific antibody research. As a result, subsequent studies have rarely been updated.
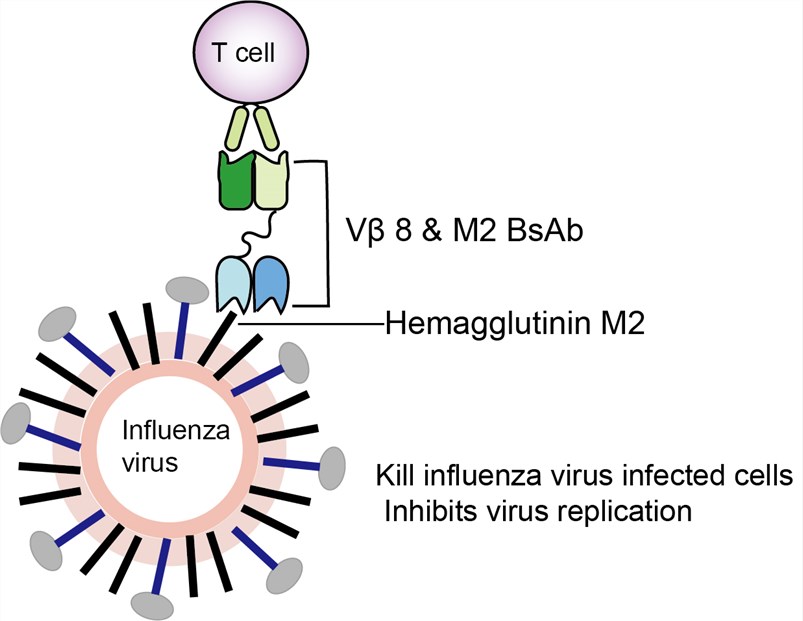
Fig.1 Mechanism of action of Vβ 8 & M2 BsAb
What We Provide
Therapeutic Antibody
Vβ 8 & M2 BsAb
We provide high-quality Vβ 8 & M2 BsAb for use in WB, FC, IP, ELISA, Neut, FuncS, IF and most other immunological methods. For lab research use only, not for diagnostic, therapeutic or any in vivo human use.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


