Palivizumab Overview
Introduction of Palivizumab
Palivizumab is a humanized IgG monoclonal antibody (mAb) manufactured with recombinant DNA technology. It is used in the prevention of respiratory syncytial virus (RSV) infections. It is recommended for infants that are high-risk because of prematurity or other medical problems such as congenital heart disease. Palivizumab directed against an epitope in the A antigenic site of the F protein of RSV. Palivizumab is a composite of human (95%) and murine (5%) antibody sequences. The human heavy chain sequence was derived from the constant domains of human IgG1 and the variable framework regions of the VH genes Cor and Cess. The human light chain sequence was derived from the constant domain of Cκ and the variable framework regions of the VL gene K104 with Jκ -4. The murine sequences were derived from a murine monoclonal antibody, Mab 1129, in a process that involved the grafting of the murine complementarity determining regions into the human antibody frameworks. Palivizumab is composed of two heavy chains and two light chains and has a molecular weight of approximately 148,000 Daltons. Palivizumab was approved for medical use in 1998 but the indications for prophylaxis vary worldwide. Palivizumab is primarily indicated for the following conditions: prematurity (gestational age ≤ 35 weeks), bronchopulmonary dysplasia/chronic lung disease, hemodynamically significant congenital heart disease, and other serious medical disorders on an ad hoc basis. Infants with these medical conditions are at increased risk of more severe disease and more serious subsequent sequelae.
Mechanism of Action of Palivizumab
RSV is an enveloped, non-segmented, single-stranded, negative-sense RNA virus belonging to the Paramyxoviridae family. Its envelope glycoproteins (Env) G and F are responsible for virus attachment and fusion with the target cell membrane. RSV F is a class I fusion glycoprotein that is synthesized as an inactive precursor (F0) that is processed by a furin-like protease at two sites to generate three polypeptides: the N-terminal fragment (F2), a 27-amino-acid glycopeptide (pep27) and the C-terminal fragment (F1). The mature, active protein exists as a trimer of F2-F1 heterodimers folded into a compact prefusion conformation. The process of membrane fusion begins once prefusion RSV F is triggered by an unknown mechanism to initiate a dramatic conformational change. This refolding can also occur spontaneously on virions and cell surfaces, and it can be stimulated with heat, indicating that prefusion RSV F is metastable. During the refolding, the hydrophobic fusion peptide at the N terminus of the F1 subunit pulls out from the central cavity of the prefusion trimer and inserts into the host-cell membrane. The resulting pre-hairpin intermediate then further refolds as the heptad repeats adjacent to the fusion peptide (HRA) associate with the heptad repeats adjacent to the viral transmembrane region (HRB), located near the C terminus of the F1 subunit. This association leads to the formation of an extremely stable six-helix bundle, which is characteristic of the RSV F postfusion conformation and brings the viral and host-cell membranes together. Inhibition of any of these steps during the fusion process may prevent entry and infection of the host cells and could thus serve as a target for therapeutic intervention. Palivizumab acts by binding the RSV F protein on the surface of the virus and blocking a critical step in the membrane fusion process.
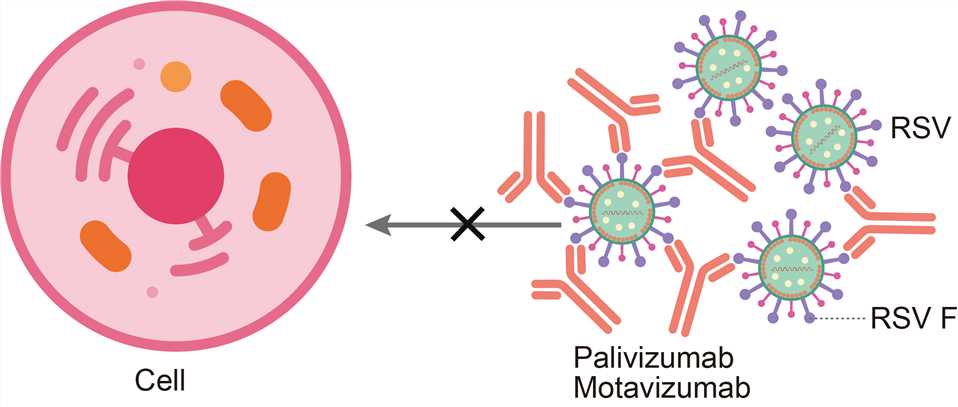
Fig.1 Mechanism of action of palivizumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

