Ponezumab Overview
Introduction of Ponezumab
Ponezumab (PF-04360365) is a humanized anti–amyloid beta (Aβ) monoclonal antibody designed for treatment of Alzheimer disease (AD). It is an immunoglobulin G2 (IgG2) antibody targeted against an epitope encompassing the C‐terminal amino acids of the Aβ1-40 peptide derived from the human amyloid precursor protein (APP). Mutations (A330S and P331S) in the IgG2Δa Fc region are intended to minimize the ability of ponezumab to activate complement or support antibody‐dependent cell‐mediated cytotoxicity. As Aβ1-40 is the predominant species present in blood vessel walls, ponezumab was developed to prevent or reverse β‐amyloid aggregation and deposition and thus prevent or reduce cerebral amyloid angiopathy (CAA) progression with minimal amyloid related imaging abnormalities (ARIA) risk. Ponezumab is originally developed by the small biotech company Rinat Neuroscience. The antibody drew widespread attention in 2006, when Pfizer acquired Rinat Neuroscience for a reported several hundred million dollars. Subsequent preclinical research reported a beneficial effect on reducing amyloid deposition in cerebral blood vessels along with improved vascular function, suggesting a possible application of ponezumab in CAA. Five phase 1 trials tested ponezumab's safety and pharmacokinetics in Western and Japanese patients with mild to moderate disease. Two phase 2 trials concluded in 2011. Between June 2013 and September 2015, Pfizer conducted a Phase 2 study of ponezumab in 36 patients with CAA. Three infusions of ponezumab or placebo over the course of 60 days were evaluated for changes in cerebrovascular reactivity, the drug showed drug-placebo differences, but did not meet the primary endpoint. In 2016, Pfizer dropped ponezumab from its pipeline.
Mechanism of Action of Ponezumab
The accumulation of amyloid β (Aβ) is thought to be integral to the pathogenesis of Alzheimer's disease (AD), contributing to the formation of neuritic plaques. Reducing amyloid deposits in brain may be warranted in some subpopulations of mild-to-moderate AD. However, amyloid is thought to begin accumulating long before the clinical symptoms of AD appear; therefore, removal of Aβ from brains of patients who have already progressed to dementia may have limited value. The brains of patients with AD also typically display cerebral amyloid angiopathy (CAA), a pathological condition caused by the progressive deposition of Aβ1–40 surrounding cerebral blood vessel walls. Although comorbidity of AD and CAA is almost universal, there are clear distinctions between them, such as the Aβ species being deposited (Aβ1–42 in AD vs. Aβ1–40 in CAA), the location of the Aβ deposits (brain parenchyma vs. brain vasculature), and the presence of cerebral microhemorrhages that are the signature of CAA. Ponezumab is a humanized IgG2Δa anti-Aβ monoclonal antibody that targets specific amino acids in the C-terminus of the Aβ sequence. It binds only to soluble Aβ and has a low propensity to induce immune responses. Ponezumab's primary mechanism of action is believed to be sequestration of Aβ in the blood and shifting the brain-blood equilibrium toward the periphery, thereby depleting central Aβ stores (the peripheral sink hypothesis). Studies of ponezumab in preclinical murine models of amyloid overexpression have reported depletion of insoluble brain Aβ deposits and reversal of cognitive defects.
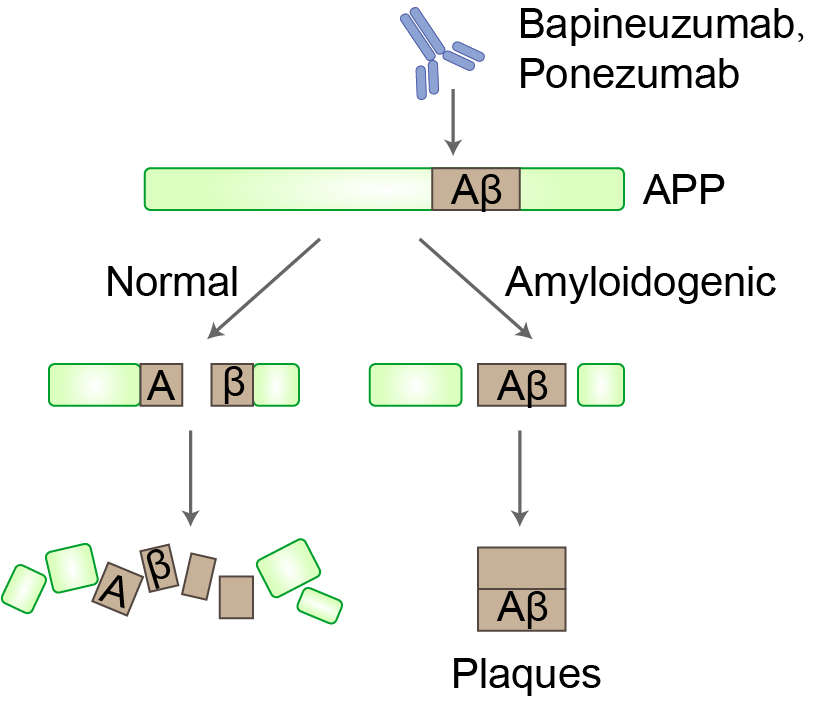 Fig 1. Mechanism of Action of Ponezumab
Fig 1. Mechanism of Action of Ponezumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

