Immunocytochemistry Protocol & Troubleshooting
Immunocytochemistry (ICC) is a technique for assessing the presence of specific proteins or antigens in cells through the use of labeled antibodies. It is the product of combining immunofluorescence techniques with morphological techniques. We describe the optimized protocol for ICC and the procedure for the detection of antigens by antibodies in the cellular environment.
Creative Biolabs provides our customers with specific ICC protocols and troubleshooting, which is applied to study the presence of proteins and subcellular localization.
Solutions and Reagents
| Stages | Solutions and Reagents |
| Sample Preparation | Phosphate buffer (PBS), paraformaldehyde, permeabilization buffer |
| Antibody Labeling | Blocking buffer, primary antibody, antibody dilution buffer, PBS, secondary antibody |
| Dyeing | Streptavidin peroxidase conjugated, hematoxylin/acetic acid, ultra-pure sterile water, coverslip solution |
Immunocytochemistry Procedure
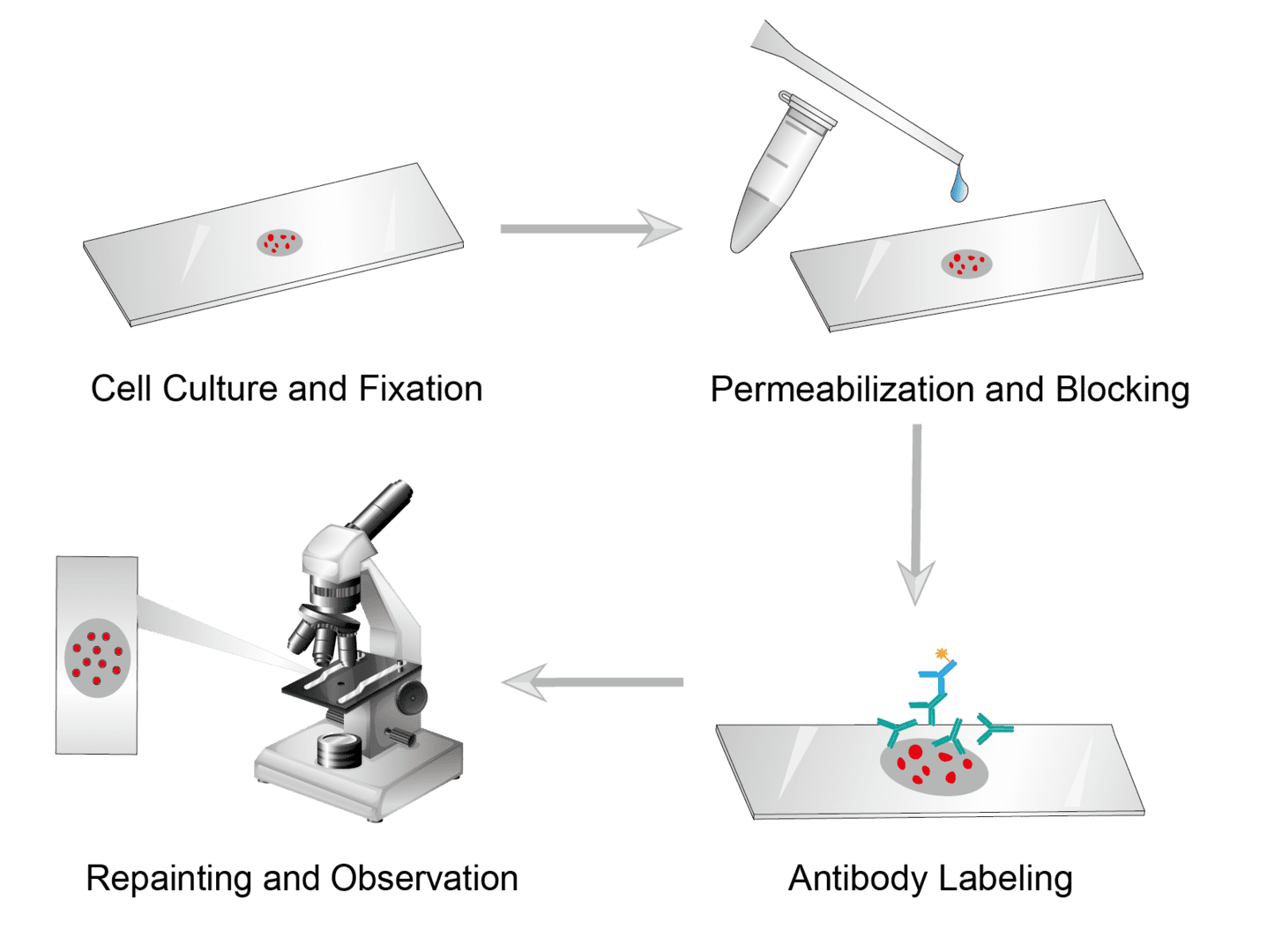
Seed cells on sterile glass coverslips or tissue culture plates, and add fresh medium. Allow cells to grow to the desired density before labeling. Then fix cells with appropriate fixation methods, wash them, and set them aside.
Incubate the slides with a permeation medium for 10-15 minutes to permeate the membrane. Wash slide when finished to remove the permeate. Then Block the sample using the blocking buffer and wash it for use.
Dilute the primary antibody to the recommended concentration with blocking buffers. Then add the primary antibody to the slide and incubate at room temperature, wash in PBS when finished.
Prepare appropriately diluted fluorescent dye-coupled secondary antibodies. Then add secondary antibodies to slide and incubate at room temperature in a dark environment. And wash gently in PBS upon completion.
Blocking can prevent non-specific binding of the antibody to the membrane. Place the membrane in the blotting vessel, add enough blocking solution to cover the entire membrane surface, and shake to incubate. This can help to reduce background noise while enhancing the target protein signal.
Troubleshooting
You can browse our ICC troubleshooting tips and advice below, which focuses on the causes and possible solutions to common problems in ICC assays.
No staining or faint staining
- Cell causes. First, target proteins may not be present or may be present at very low levels in the cell sample. Second, cells may also be detached from the coverslip or over-fixed. Cells can be made to adhere by reducing the intensity of the wash, using another fixation solution, or lengthening/reducing the fixation time. It is also possible that the cells are not fully permeabilized and you can use other permeabilizing solutions to improve. Cell drying will not show a fluorescent signal, so it is important to keep the sample in liquid throughout the procedure.
- Antibody causes. You may not be using enough primary or secondary antibodies. So, you can adjust the antibody dilution to determine the optimal antibody concentration or increase antibody incubation time. Antibodies may not be compatible. You should ensure that secondary antibodies are produced against the animal that produced the primary antibody in order to achieve synergy. If using fluorescent assays, you should ensure that the coupled fluorescent-stained antibodies are not exposed to light.
- Chromogen and substrate causes. Improper use of chromogen and substrate can cause this. You can choose another suitable chromogen and substrate and repeat the test. It is also possible that the re-staining agent is over-stained and the re-staining time can be reduced.
- Microscope causes. Check and adjust the microscope settings, increase the exposure time of the camera and observe if the situation can be improved.
Messy staining results
- Antibody causes. The antibody concentration is too high so you may switch the antibody to a lower concentration in the test. And primary antibodies should be from a different species than the cells. Please check if you are using recombinant antibody solutions that may form aggregates. We recommend centrifuging all antibody preparations prior to use.
- Cell causes. Disrupted cell morphology or the creation of air bubbles can lead to signal clutter. Keep the cells at high humidity to avoid them drying out as much as possible.
High background
- Antibody causes. If you are experiencing a high background signal, it is possible that the antibody is binding to a non-specific site. There are steps you can take to reduce the high background signal. You can titrate the antibody to determine the optimal concentration to promote the reaction of primary and secondary antibodies, increase the concentration of the blocking buffer, or switch to a monoclonal antibody to reduce the high background.
- Fixation causes. Too long a fixation time can affect epitopes and lead to high background levels. Try using shorter exposure times, lower temperatures, lower concentrations of fixative, or reducing the length of fixation.
- Blocking agent causes. Insufficient closure leads to high background. You can increase the closure latency and consider replacing the blocking agent.
- Incubation causes. Incubate for too long or at too high an incubation temperature.
- Washing causes. It is critical that each step needs to be washed correctly and adequately between steps. When a high background occurs, you can increase the amount of wash, gently agitate the wash, or increase the concentration of Tween in PBS.
- Contamination causes. You need to make sure that the slides are clean and free of dust. The buffer should be kept fresh to prevent microbial contamination.
If you are having difficulty with a specific ICC operation, please first review the common problems listed above. If your question is not answered, please fill out the form in as much detail as possible and contact us. Our technical support will contact you shortly to take your comments seriously and help resolve the problem.
ICC is a relatively simple and straightforward experimental approach. For each ICC study, we can help you identify and optimize variables and troubleshoot various factors. For further services and assistance, please contact us.
Products with Tested Data
At Creative Biolabs, we are dedicated to providing high-quality antibodies for various research applications. Each product in our extensive range has been rigorously tested to ensure superior reliability and efficacy. To showcase the performance of our antibodies, we have conducted numerous experiments using Immunocytochemistry (ICC). Below, you will find a table listing a selection of our antibody products along with images from these experiments, demonstrating their proven reliability.
| Product Name | Catalog Number | Target | Image | Description |
|---|---|---|---|---|
| Rabbit Anti-H2AC4 Recombinant Antibody (clone 3H5) | H2AC4 | H2AC4 |

|
Immunocytochemistry analysis of this product diluted at 1:100 and staining in Hela cells performed on a Leica BondTM system. After dewaxing and hydration, antigen retrieval was mediated by high pressure in a citrate buffer (pH 6.0). Section was blocked with 10% normal goat serum 30min at RT. Then primary antibody (1% BSA) was incubated at 4°C overnight. The primary is detected by a biotinylated secondary antibody and visualized using an HRP conjugated SP system. |
| Recombinant Rabbit Anti-PRKCB Antibody (clone R05-3F4) | PRKCB | PRKCB |

|
Immunocytochemical analysis of PKC beta 1 (green) in K562 using PKC beta 1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-NCK1 Antibody (clone R09-7F6) | NCK1 | NCK1 |

|
Immunocytochemical analysis of NCK1 (green) in HeLa using NCK1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-NDUFB4 Antibody (clone R05-5C8) | NDUFB4 | NDUFB4 |

|
Immunocytochemical analysis of NDUFB4 (green) in HeLa using NDUFB4 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-PAK1 Antibody (clone R08-5I5) | PAK1 | PAK1 |

|
Immunocytochemical analysis of PAK1 (green) in Jurkat using PAK1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SP1 Antibody (clone R01-9G6) | SP1 | SP1 |

|
Immunocytochemical analysis of SP1 (green) in HeLa using SP1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-STK4 Antibody (clone R09-3C1) | STK4 | STK4 |

|
Immunocytochemical analysis of MSP (green) in K562 using an MSP antibody and DAPI (blue). |
| Recombinant Rabbit Anti-NTHL1 Antibody (clone R08-8E9) | NTHL1 | NTHL1 |

|
Immunocytochemical analysis of NTH1 (green) in Hela using NTH1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-HIF1AN Antibody (clone R03-2B3) | HIF1AN | HIF1AN |

|
Immunocytochemical analysis of HIF1AN (green) in HT-1080 using HIF1AN antibody and DAPI (blue). |
| Recombinant Rabbit Anti-VAV2 Antibody (clone R06-8F5) | VAV2 | VAV2 |

|
Immunocytochemical analysis of VAV2 (green) in HeLa using VAV2 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-LYZ Antibody (clone R09-9D6) | LYZ | LYZ |

|
Immunocytochemical analysis of lysozyme (green) in HEPG2 using lysozyme antibody and DAPI (blue). |
| Recombinant Rabbit Anti-CBL Antibody (clone R01-9G1) | CBL | CBL |

|
Immunocytochemical analysis of CBL (green) in K562 using CBL antibody and DAPI (blue). |
| Recombinant Rabbit Anti-POU2F2 Antibody (clone R02-5E7) | POU2F2 | POU2F2 |

|
Immunocytochemical analysis of NUP155 (green) in HeLa using NUP155 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SAFB Antibody (clone R03-7A1) | SAFB | SAFB |

|
Immunocytochemical analysis of SAFB1 (green) in 293 using SAFB1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-RAD51 Antibody (clone R04-3C6) | RAD51 | RAD51 |

|
Immunocytochemical analysis of Rad51 (green) in Jurkat using Rad51 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-S100A6 Antibody (clone R01-8A4) | S100A6 | S100A6 |

|
Immunocytochemical analysis of S100 alpha 6/PRA (green) in hela using S100 alpha 6/PRA Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SOD1 Antibody (clone R08-5A5) | SOD1 | SOD1 |

|
Immunocytochemical analysis of superoxide dismutase 1 (green) in HCT116 using superoxide dismutase 1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-Bsg Antibody (clone R03-7C3) | Bsg | Bsg |

|
Immunocytochemical analysis of CD147 (green) in 3T3 using CD147 antibody. |
| Recombinant Rabbit Anti-UBE3A Antibody (clone R02-1A0) | UBE3A | UBE3A |

|
Immunocytochemical analysis of Ubiquitin Ligase E3A (green) in Hela using Ubiquitin Ligase E3A Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-ACTA1 Antibody (clone R05-4F6) | ACTA1 | ACTA1 |

|
Immunocytochemical analysis of α-skeletal muscle actin (green) in HeLa using α-skeletal muscle actin antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SUPT5H Antibody (clone R01-5C2) | SUPT5H | SUPT5H |

|
Immunocytochemical analysis of SPT5 (green) in Hela using SPT5 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-PRDX1 Antibody (clone R04-0H5) | PRDX1 | PRDX1 |

|
Immunocytochemical analysis of peroxidase 1 (green) in Hela using peroxidase 1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-PSEN1 Antibody (clone R05-4D4) | PSEN1 | PSEN1 |

|
Immunocytochemical analysis of Presenilin 1 (green) in SKOV-3 using Presenilin 1 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SMYD3 Antibody (clone R03-2I7) | SMYD3 | SMYD3 |

|
Immunocytochemical analysis of SMYD3 (green) in K562 using SMYD3 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-LCK Antibody (clone R01-8A5) | LCK | LCK |

|
Immunocytochemical analysis of Lck (green) in Jurkat using Lck antibody and DAPI (blue). |
| Recombinant Rabbit Anti-LEF1 Antibody (clone R02-3A4) | LEF1 | LEF1 |

|
Immunocytochemical analysis of LEF1 (green) in Jurkat using LEF1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SIRPA Antibody (clone R08-1G4) | SIRPA | SIRPA |

|
Immunocytochemical analysis of SIRP α (green) in hela using SIRP α antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SKP2 Antibody (clone R08-0A2) | SKP2 | SKP2 |

|
Immunocytochemical analysis of SKP2 (green) in hela using SKP2 Rabbit mAb and DAPI (blue). |
| Recombinant Rabbit Anti-CHEK2 Antibody (clone R04-8E5) | CHEK2 | CHEK2 |

|
Immunocytochemical analysis of Chk2 (green) in HCT116 using Chk2 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-MAP2K1 Antibody (clone R01-3C2) | MAP2K1 | MAP2K1 |

|
Immunocytochemical analysis of MEK1 (green) in HeLa using a MEK1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-NDUFS1 Antibody (clone R02-3I2) | NDUFS1 | NDUFS1 |

|
Immunocytochemical analysis of NDUFS1 (green) in HeLa using NDUFS1 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SYP Antibody (clone R06-2A2) | SYP | SYP |

|
Immunocytochemical analysis of synaptophysin (green) in 3T3 using synaptophysin antibody and DAPI (blue). |
| Recombinant Rabbit Anti-TARDBP Antibody (clone R02-2A8) | TARDBP | TARDBP |

|
Immunocytochemical analysis of TDP 43 (green) in 293t using TDP 43 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-FGFR1OP Antibody (clone R02-1H4) | FGFR1OP | FGFR1OP |

|
Immunocytochemical analysis of FGFR1 oncogene chaperone (green) in K562 using FGFR1 oncogene chaperone antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SNTA1 Antibody (clone R03-4E7) | SNTA1 | SNTA1 |

|
Immunocytochemical analysis of Syntrophin alpha 1 (green) in U87-MG using Syntrophin alpha 1 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-NCK2 Antibody (clone R01-3C6) | NCK2 | NCK2 |

|
Immunocytochemical analysis of Nck beta (green) in HeLa using Nck beta antibody and DAPI (blue). |
| Recombinant Rabbit Anti-HIPK2 Antibody (clone R06-5G2) | HIPK2 | HIPK2 |

|
Immunocytochemical analysis of HIPK2 (green) in hela using HIPK2 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-CHAT Antibody (clone R07-1G8) | CHAT | CHAT |

|
Immunocytochemical analysis of choline acetyltransferase (green) using choline acetyltransferase antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SNAP29 Antibody (clone R01-8C8) | SNAP29 | SNAP29 |

|
Immunocytochemical analysis of SNAP29 (green) in K562 using SNAP29 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-SMAD4 Antibody (clone R05-1I7) | SMAD4 | SMAD4 |

|
Immunocytochemical analysis of Smad4 (green) in HeLa using Smad4 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-VAMP3 Antibody (clone R01-8D3) | VAMP3 | VAMP3 |

|
Immunocytochemical analysis of Cellubrevin (green) in hela using Cellubrevin Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-RRM2 Antibody (clone R09-4G8) | RRM2 | RRM2 |

|
Immunocytochemical analysis of RRM2 (green) in Jurkat using RRM2 Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-CLPX Antibody (clone R05-2A6) | CLPX | CLPX |

|
Immunocytochemical analysis of CLPX (green) in HT-1080 using CLPX antibody and DAPI (blue). |
| Recombinant Rabbit Anti-ACTR2 Antibody (clone R04-3H3) | ACTR2 | ACTR2 |

|
Immunocytochemical analysis of actin-associated protein 2 (green) in HeLa using ACT2 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-ALCAM Antibody (clone R03-2E5) | ALCAM | ALCAM |

|
Immunocytochemical analysis of CD166 (green) in SKOV-3 using CD166 antibody and DAPI (blue). |
| Recombinant Rabbit Anti-HCK Antibody (clone R02-5C1) | HCK | HCK |

|
Immunocytochemical analysis of Hck (green) in hela using Hck antibody and DAPI (blue). |
| Recombinant Rabbit Anti-CSNK2B Antibody (clone R01-5D5) | CSNK2B | CSNK2B |

|
Immunocytochemical analysis of casein kinase 2 beta (green) in Hela using casein kinase 2 beta antibody and DAPI (blue). |
| Recombinant Rabbit Anti-APC Antibody (clone R06-2E4) | APC | APC |

|
Immunocytochemical analysis of APC (green) in LNcap using APC antibody and DAPI (blue). |
| Recombinant Rabbit Anti-ARHGDIA Antibody (clone R07-1D8) | ARHGDIA | ARHGDIA |

|
Immunocytochemical analysis of RhoGDI (green) in Hela using RhoGDI Antibody and DAPI (blue). |
| Recombinant Rabbit Anti-CYCS Antibody (clone R02-9A7) | CYCS | CYCS |

|
Immunocytochemical analysis of cytochrome C (green) in K562 using cytochrome C antibody and DAPI (blue). |
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

