Refanezumab Overview
Introduction of Refanezumab
Refanezumab is an anti-human MAG therapeutic antibody for the recovery of motor function after stroke. It is also known as GSK249320 and developed by GlaxoSmithKline. Refanezumab has been investigated in the treatment of some diseases such as cerebrovascular accident and ischaemic attack. The earlier study has assessed the safety, pharmacokinetics, and pharmacodynamics of escalating repeat doses of refanezumab in patients with stroke, showing it well-tolerated and no major safety issues. Another experiment explores its effect on healthy volunteers. In this trial, in addition to evaluating the general safety, pharmacokinetics and immunogenicity, the potential safety concerns of adverse effects on myelin of refanezumab was also included. The results showed that the most common adverse events were headache and upper respiratory tract infection, and there were no serious adverse events. The final trial validated the role of refanezumab as a monoclonal therapeutic antibody in patients with cerebral stroke. The results showed that the antibody was well tolerated and showed low immunogenicity, but the therapeutic effect on the patients was not improved.
Mechanism of Action of Refanezumab
Refanezumab is an IgG1‐type humanized monoclonal antibody against to myelin-associated glycoprotein (MAG) with disabled Fc region. The disabled Fc region can reduce the binding and complementarity of refanezumab to Fc- γ receptors so as to reduce unnecessary immune-related responses. Stroke is a disease with abnormal brain function, which is divided into ischemic stroke (caused by vascular blockage) and hemorrhagic stroke (caused by bleeding). At present, drugs for the treatment of stroke can be divided into anticoagulants (keep your blood from clotting easily), antiplatelet drugs (prevent blood clots), tissue plasminogen activators (breaks up a blood clot), statins (block HMG-CoA reductase to reduce plaques), and blood pressure drugs (contribute to chunks of plaque breaking off). The treatment of functional recovery after stroke is mainly focused on repair therapy. One way is to enhance brain repair by promoting axonal growth. After central nervous system injury, the three inhibitory molecules that inhibit axonal regrowth are myelin-associated glycoprotein (MAG), oligo-myelin glycoprotein, and nogo-A. Experimental studies in mouse models have found that MAG is the most highly overexpressed of these inhibitors in the stroke penumbra. Thus, inhibition of MAG could be a potential strategy for enhancing functional recovery poststroke. Refanezumab is designed as monoclonal antibody binding to MAG and blocking MAG-mediated inhibition of axonal regeneration. Interaction of MAG with receptors including nogo receptor-1, coreceptors TROY/p75, lingo-1 causes downstream signaling leading to rigidification of the axonal actin cytoskeleton and inhibition of neurite outgrowth. In preclinical studies, it was found that refanezumab could cross the blood-brain barrier (BBB) to neutralize MAG-mediated inhibition and promote axonal growth. In addition, it can also protect oligodendrocytes from oxidative stress death induced by glutamate. In theory, refanezumab can neutralize MAG released after injury, relieving MAG inhibition of neuronal regeneration, promoting neuronal regeneration and increasing neuronal survival rate. However, more experimental is needed to explore the potential risk.
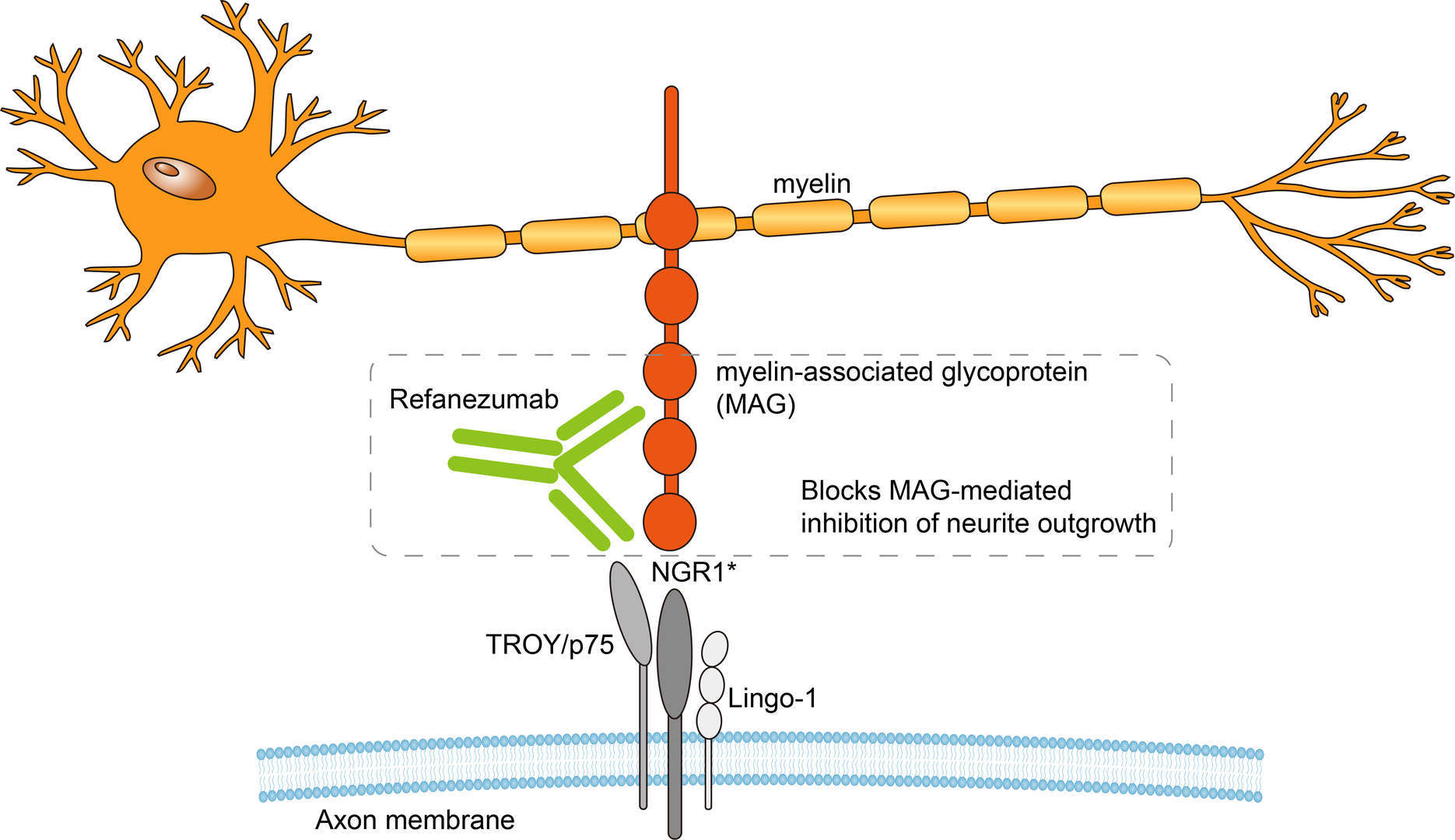 Fig.1 Mechanism of Action of Refanezumab
Fig.1 Mechanism of Action of Refanezumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

