Lerdelimumab Overview
Introduction of Lerdelimumab
Lerdediumab, also known as CAT-152, is a human monoclonal antibody targeting transforming growth factor (TGF β2). It is an immunosuppressive agent developed for the treatment of ocular diseases as an auxiliary material for glaucoma drainage surgery. It has been found that it can inhibit fibrotic effects induced by TGF β2 and inhibit posterior capsule opacification. In addition, it also shows some potential activity in the treatment of fibrosis, cataract, retinopathy and connective tissue diseases. Lerdiumab has been granted European orphan drug status as a potential treatment to prevent scar formation after glaucoma surgery.
Mechanism of Action of Lerdelimumab
TGF β2 is a cytokine belonging to a newly discovered TGF-β superfamily that regulates cell growth and differentiation. TGF-β plays an important role in inflammation, tissue repair and embryonic development, cell growth, differentiation and immune function. Generally speaking, TGF-β stimulates cells derived from mesenchymal cells and inhibits cells derived from epithelium or neuroectoderm. TGF β2 plays an important role in ocular diseases. Under normal circumstances, TGF β2 exists in the form of no activity, and TGF β2 can not play the corresponding biological function until activated. It has been found that TGF β2 is a bifunctional cell division regulator, which can inhibit the proliferation of retinal pigment epithelial cells. However, it can promote the division and proliferation of collagen fibers and fibroblasts. The excessive activity of TGF β2 is considered to be the main cause of intraocular and peripheral scar formation after glaucoma surgery. The use of monoclonal antibody lerdiumab, targeting TGF β2 can balance the active state of TGF β2 and reduce scar formation.
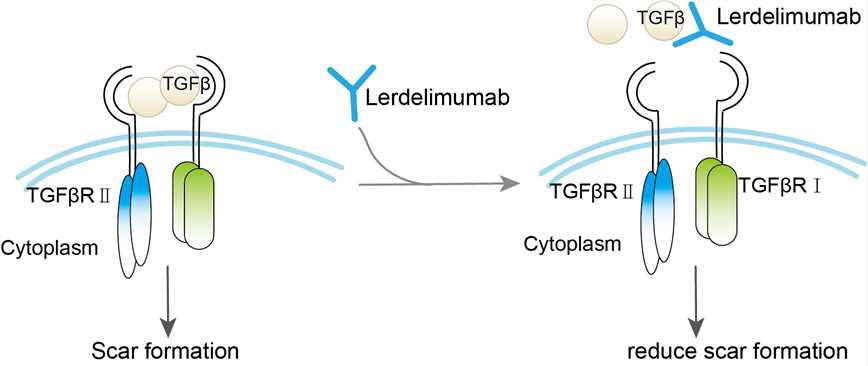
Fig.1 Mechanism of action of Lerdelimumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

