Satumomab Pendetide Overview
Introduction of Satumomab Pendetide
Satumomab Pendetide, also known as Indium (111In) satumomab pendetide, is a mouse monoclonal antibody targeted at tumor associated glycoprotein (TAG) 72 (TAG-72). It was developed as an imaging agent, and was the first monoclonal antibody approved by the US Food and Drug Administration (FDA) for tumor imaging. Indium (111In) Satumomab Pendetide is a complex that consist in a murine monoclonal antibody target at TAG-72 and a radionuclide indium-111. The drug was used for the detection of colon and ovarian cancer. And it is commonly used to determine the extent and location of extrahepatic malignant diseases in patients with known colorectal or ovarian cancer. However, this diagnostic reagent is no longer commercially available.
Mechanism of Action of Satumomab Pendetide
Indium (111In) satumomab pendetide is a monoclonal antibody labeled with radionuclides for tumor imaging and detection. The antibody part of the drug binds to TAG-72 antigens on the surface of tumor cells, and the radioactive element Indium emits low-energy gamma photons for planar imaging. TAG-72 is a surface glycoprotein found in a variety of tumor cells, and often used as a marker for tumor diagnosis in ovarian cancer, breast cancer, colon cancer, lung cancer and pancreatic cancer. The indium-111 is a radioactive isotope of indium (In), and its half-life is close to 3 days, which makes it suitable for total antibody coupling. Pendetide, also known as GYK-DTPA, is a chelating agent consisting of pentetic acid (DTPA) linked to the tripeptide glycine (G)-L-tyrosine (Y)-L-lysine (K), and is used to chelate radioactive elements to antibodies. Indium (111In) satumomab pendetide) is used in tumor imaging of most colorectal and ovarian cancers and other cancers. Colon and ovarian cancers are both cancers that cause a high number of deaths. Tumor imaging detection reagents can detect carcinogenesis as early as possible, which is helpful to the diagnosis of diseases.
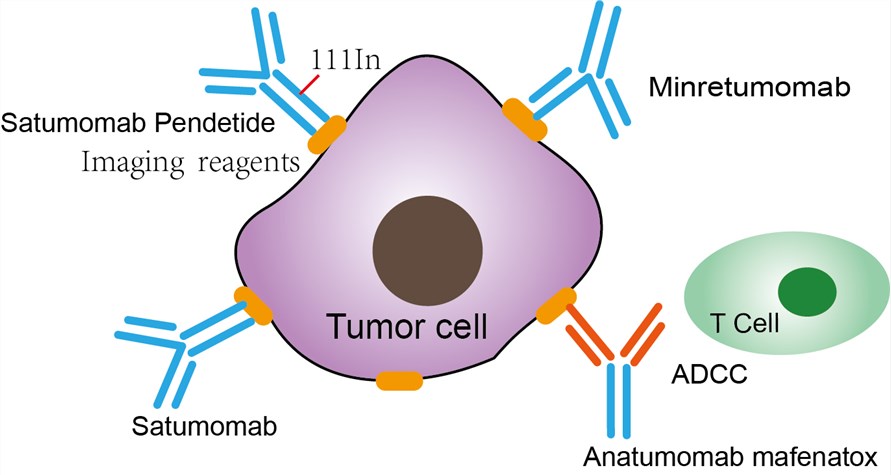
Fig.1 Mechanism of action of Satumomab Pendetide
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

