Therapeutic Antibody Overview
Introduction
It has been more than one hundred years from antibody's first discovery in blood serum to the current therapeutic antibody development. Antibody structure and antibody function is well known, so it won’t be repeated in this overview.
What Are Therapeutic Antibodies?
Therapeutic antibodies are biopharmaceuticals widely used in the treatment of cancer, autoimmunity, and inflammatory diseases or for drug delivery to target antigen, most of which are monoclonal antibodies. Therapeutic antibody recognizes and binds to the antigen receptor to activate or inhibit a series of biological process for blocking cancer cell growth or triggering immune system. The mechanism of action (MOA) between therapeutic antibody and small molecule pharmaceutical is dramatically different, even though both of them target the same antigen site. Compared to small molecule pharmaceutical, therapeutic antibody possesses characteristic properties of high affinity, high specificity, and low immunogenicity, as it is essentially a monoclonal antibody that can recognize and bind to a unique epitope. Early monoclonal antibody (mab) was generated by fusing myeloma cell and spleen cells to obtain hybridoma secreting mab. Early therapeutic antibody was usually produced in murine host, leading to serious human anti-mouse antibody expression called HAMA reaction, which in turn interfered with the therapeutic effects. While, the disadvantages of short half-life, xenogenicity and limited activity were gradually improved with the development of gene engineering, in vitro cell culture technology and the deeper understanding of each antibody fragment function. In the past four decades, therapeutic antibodies have made great improvement in antibody engineering. Below is a timeline of important technologies for engineering therapeutic antibodies and representative approved biopharmaceuticals (Fig. 1).
Development Process of Therapeutic Antibodies
Benefiting from the advances of antibody engineering technologies, immunogenicity of therapeutic antibody is rapidly reduced. According to different immunogenicity, therapeutic antibody can be divided into four stages, consisting of murine antibody, chimeric antibody, humanized antibody and fully human antibody. Fig. 2 shows these four types of therapeutic antibodies, among which chimeric antibody has the highest immunogenicity.
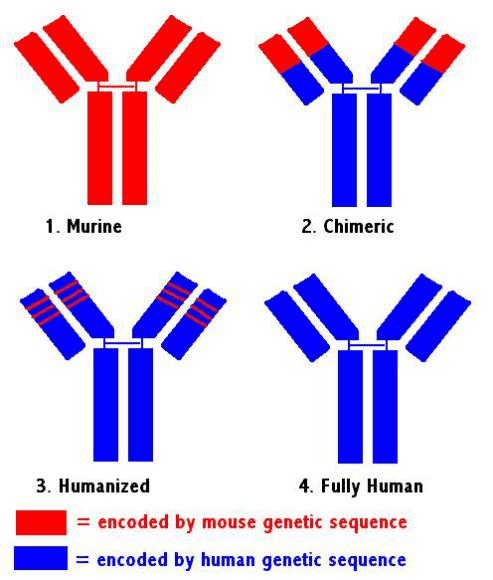 Fig.1 Therapeutic antibody formats.
Fig.1 Therapeutic antibody formats.
The era of therapeutic antibody emerged from hybridoma technology, generating monoclonal antibody targeting a unique epitope. In 1975, Kohler and Milstein introduced an in vitro cultured hybridoma secreting anti-sheep red blood cell monoclonal antibody by fusing mouse myeloma cell and mouse spleen cells. This hyriboma overcame the problem of B cells in vitro culture, which helped Kohler and Milstein win the Nobel Prize of Physiology or Medicine in 1984. 11 years after the appearance of hybridoma technology, a murine anti-CD3 monoclonal antibody, was approved as the first therapeutic antibody. Unexpectedly, with the increasing number of patients treated with murine monoclonal antibody, terrible side-effect appeared. Human immune system recognized murine monoclonal antibody as a foreign antigen that induced HAMA reaction, which shortened the half-life of murine monoclonal antibody in human serum. Moreover, murine monoclonal antibody Fc fragment did not interact with complementary system or effector cells to activate CDC or ADCC effector function. In the following years, considerable efforts were devoted to genetically engineering monoclonal antibodies in order to make them less immunogenicity.
The second generation of therapeutic antibody is chimeric antibody. In 1984, Morrison firstly introduced chimeric antibody using recombinant DNA technology, joining mouse antibody variable region gene to human antibody constant region gene. The constructed chimeric vector was transfected into mouse myeloma cells and the cells expressed chimeric antibodies. Combining the mouse variable region with human constant region improved the monoclonal antibody humanization proportion to 70%, which reduced HAMA. In 1994, the first chimeric therapeutic antibody, Fab formats, was approved for inhibiting platelet aggregation by binding platelet glycoprotein Ⅱb/Ⅲa receptor. In 1997, the first full-length IgG chimeric therapeutic antibody was approved for treatment of non-hodgkin’s lymphoma and rheumatoid arthritis. Chimeric formats therapeutic antibody achieved greatersuccess than murine therapeutic antibody because of its lower immunogenicity and effective Fc effector function, which encouraged people to fight against cancer and autoimmune diseases by focusing on therapeutic antibody development.
In order to further improve humanization proportion, humanized antibody was developed by researchers. In 1986, Jones provided a method for reducing immunogenicity brought by humanized antibody's framework region through grafting complementarity-determine region (CDR) from mouse antibody into corresponding region of human antibody. This method was widely used with a modification in later humanized therapeutic antibody research and development. Humanized therapeutic antibody improved humanization proportion to 85%-90%. The first approved humanized therapeutic antibody is anti-CD25 used in the treatment of adults with relapsing forms of multiple sclerosis. Nowadays, chimeric antibody and humanized antibody are main formats of human therapeutic antibody and play an important role in cancer therapy.
4. Fully Human Antibody
Another stage of therapeutic antibody is fully human antibody which benefits from the development of two technologies. The first one is in vitro screening technology–phage display. In 1990, Greg Winter`s laboratory introduced this new technology by amplifying immunoglobulin variable region from hybridomas or B cells and cloning into expression vector. Phages carrying complete variable region gene displayed protein expressed by antibody variable region on their surface, then they could be selected directly by specific antigen. Once expressed variable region protein binding the specific antigen was selected, its gene is immediately available, allowing sequencing of phage clone and further manipulation, which made it possible to rapidly and effectively select fully human antibody. Another technology is transgenic mouse replacing the whole mouse IgG repertoire with a human one. The transgenic mouse is immunized with specific antigen, then high affinity antibody can be generated through hybridoma technology. Compared to phage display, transgenic mouse has obvious advantages. They do not need additional affinity maturation procedure. Moreover, a full-length IgG is directly generated, which is often the preferred format of therapeutic antibody. However, if the antigen is toxic, the phage display may be a better choice.
Therapeutic Monoclonal Antibodies Approval and Revenue
In pharmaceutical industry, therapeutic antibodies are now being uses as important drug modalities. Recently, significant work is being carried out by scientists to revolutionize the antibodies production technique and to develop complex monoclonal antibodies based therapies (bispecifics, brain penetrant monoclonal antibodies and monoclonal antibodies mixture) in order to address noteworthy medical needs. The development of therapeutic monoclonal antibodies is the fastest in the field of drug discovery and development, and therapeutic monoclonal antibodies have brought new hope for the treatment of diseases such as cancer, immune disease. As of January 2017, 68 therapeutic monoclonal antibodies has been approved for the treatment of different indications. Therapeutic monoclonal antibodies have shown an unprecedented pace of development. Only three therapeutic monoclonal antibodies were approved in 2013, and four in 2014. While, 10 therapeutic monoclonal antibodies were successively approved in 2015 and 2016, respectively. It is notable that market revenue of therapeutic monoclonal antibodies is increasing year by year. Therapeutic monoclonal antibody occupies 5 of the top 10 best-selling biological agents.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

