Nebacumab Overview
Introduction of Nebacumab
Nebacumab, also known as HA-1A or Centoxin, is a human IgM monoclonal antibody that binds to the lipid A domain of endotoxin and is produced by the stable heteromyeloma cell line A6(H4C5), developed by Teng et al. This hybridoma was created by fusion of a murine-human heteromyeloma line with splenic B lymphocytes sensitized in vivo by immunization with killed E. coli J5 cells and subsequently transformed in vitro by Epstein-Barr virus. The clone produces only human IgM antibody and is free of Epstein-Barr viral genome and of detectable murine viruses. In experimental models, HA-1A protects animals against endotoxemia and development of the dermal Shwartzman reaction. HA-1A has been shown to bind N. meningitidis LPS. Clinical studies reported no severe side effects; antibodies to HA-1A were not detected in any patient. Licensed by Centocor in early 1986, HA-1A, renamed Centoxin, was one of a number of therapeutic mAb products the company was developing. Validations of the drug were given an extra boost when, in early 1991, the US army ordered a batch of Centoxin. In March 1991 the European Committee for Proprietary Medicinal Products recommended Centoxin for the treatment of Gram-negative sepsis. Based on this recommendation, Centoxin was subsequently approved in The Netherlands, Britain, Germany and France between March and December 1991. In September 1991, the FDA Vaccines and Related Biological Advisory Committee, although expressing some reservations about the validity of results showing Centoxin increased survival rates in septic shock, unanimously advised FDA approval with restrictive labeling for the drug. In late November 1991, the FDA was alerted to a trial undertaken in specially bred beagles used to assess Centoxin that had been undertaken by the US National Institutes of Health (NIH) Clinical Center's Department of Critical Care Medicine. The study showed the drug to be potentially lethal and unable to protect against sepsis. Finally, it has been withdrawn in 1993 because it failed to reduce mortality in clinical trials.
Mechanism of Action of Nebacumab
Lipopolysaccharide (LPS, endotoxin) is the main surface antigen (O-antigen) and important virulence factor of most of the Gram-negative bacteria pathogenic for humans and animals. LPS contributes greatly to the structural integrity of the bacterial cell wall and constitutes a “pathogen-associated molecular pattern” for host infection. Most of the known structures of LPS are smooth-type molecules built of O-specific polysaccharide, core oligosaccharide, and lipid A. Among all these defined regions, the glycolipid part of LPS called lipid A constitutes a center of biological activity of the endotoxin that stimulates different cells of the immune system. The hexa-acylated lipid A displays the highest immunostimulatory or endotoxic activity in the mammalian host and it was identified for the first time in Escherichia coli LPS. Interaction of such asymmetric, hexa-acylated lipid A region of LPS with mCD14/TLR4/MD-2 receptor complex on the surface of monocytes/macrophages constitutes a major mechanism responsible for innate immune response to Gram-negative infection. High levels of inflammatory mediators, as a response of the immune system to large amount of LPS released into a bloodstream, have profound effects on the cardiovascular system, kidneys, lungs, liver, and central nervous system and trigger the coagulation cascade. Excessive inflammatory response of the innate immune system finally leads to sepsis and septic shock. Nebacumab is a primarily human IgM monoclonal antibody that binds to the lipid A region of endotoxin and reduces circulating levels of endotoxin [lipopolysaccharide (LPS)] and tumor necrosis factor in vivo.
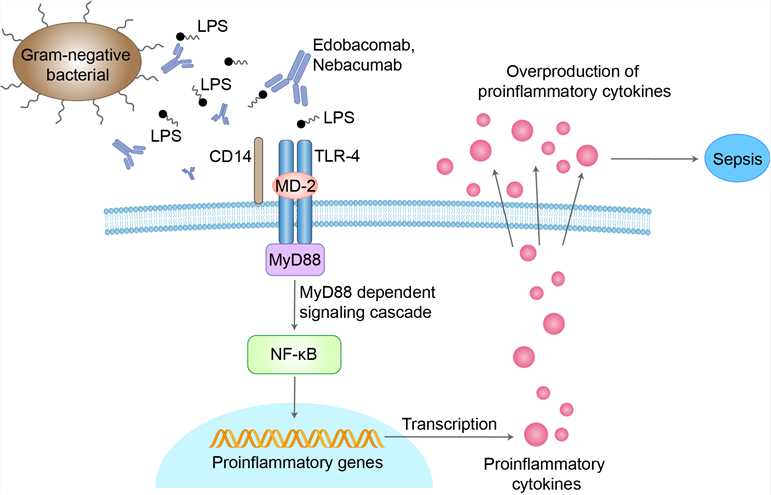 Fig 1. Mechanism of Action of Nebacumab
Fig 1. Mechanism of Action of Nebacumab
What We Provide
Therapeutic Antibody
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


