Brentuximab Vedotin Overview
Introduction of Brentuximab Vedotin
Brentuximab vedotin is an antibody-drug conjugate (ADC) medication consisting of the monoclonal antibody cAC10 and the cytotoxic agent MMAE used to treat relapsed or refractory Hodgkin lymphoma (HL) and systemic anaplastic large cell lymphoma (ALCL). cAC10 (SGN-30) is a chimeric anti-CD30 monoclonal antibody that is derived from the fusion of the variable heavy and light region of the murine anti-CD30 antibody AC10, with the constant γ1-heavy and κ-light region of the human immunoglobulin. MMAE is a synthetic derivative of dolastatin 10, a cytostatic pseudopeptide isolated from the marine shell-less mollusk Dorabella auricularia. MMAE has the effect of cytostasis, tubulin-dependent GTP hydrolysis, and polymerization. Brentuximab vedotin was initially approved in 2011. In January 2012, the drug label was revised with a boxed warning of a condition known as progressive multifocal leukoencephalopathy and death due to opportunistic JC virus infection post treatment.
Mechanism of Action of Brentuximab Vedotin
Hodgkin Lymphoma (HL) accounts for10% of all lymphomas and 0.6% of all cancers in the developed world. HL is histologically defined by rare mononuclear Hodgkin cells and multinucleated Reed-Stemberg (HRS) cells residing in a sea of inflammatory cells. HRS cells are derived from germinal center B cells, but phenotypically lose the expression of typical B cell markers. Despite an excellent cure rate with modern frontline HL therapy, approximately 15% to 20% of patients are not cured with first line or second line therapy and will require additional treatments. HRS cells are characterized by the expression of CD30 receptors. CD30 is a transmembrane glycoprotein that belongs to the tumor necrosis factor (TNF) receptor superfamily. In normal tissue, CD30 expression is restricted to activated T and activated B cells which makes it an ideal therapeutic target for monoclonal antibodies. Brentuximab vedotin is an antibody-drug conjugate (ADC) that was developed by conjugating the tubulin toxin monomethyl auristatin E (MMAE) to the chimeric monoclonal anti-CD30 antibody cAC10. On average, four molecules of MMAE are conjugated to one cAC10. After binding to CD30, brentuximab vedotin in internalized and processed into lysosomal vesicles leading to the release of MMAE from the antibody by reduction or acid hydrolysis within the lysosomes. Subsequently, MMAE is released into cytoplasm and inhibits microtubule polymerization leading to cell cycle arrest followed by cell death. As HRS cells die, a small amount of MMAE is released into the tumor microenvironment which can kill neighboring cells by a CD30-independent manner.
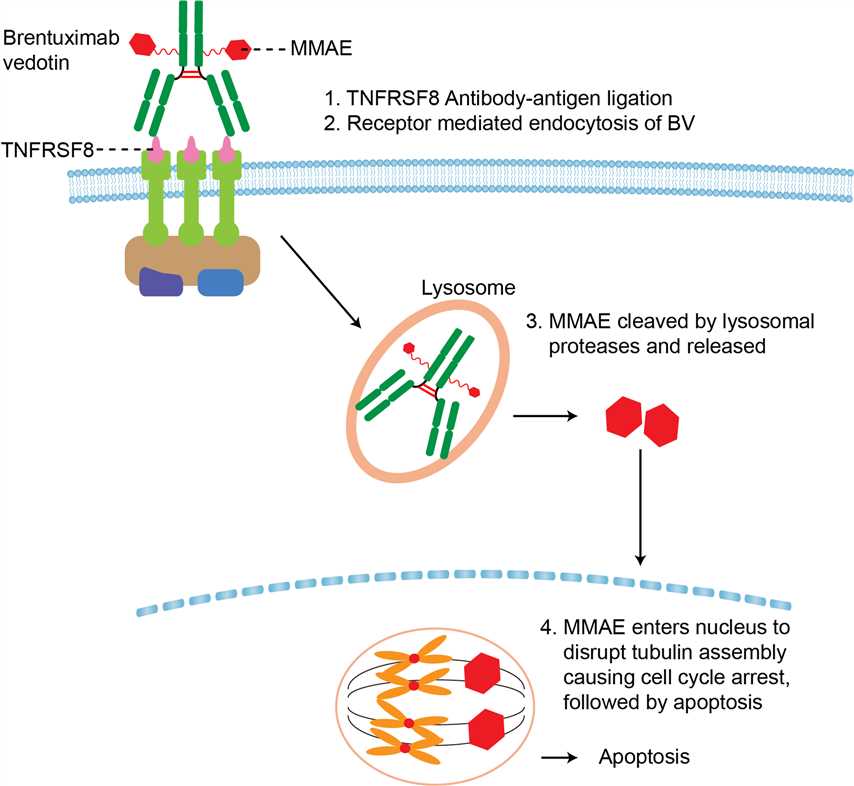
Fig 1. Mechanism of Action of Brentuximab vedotin
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

