Fezakinumab Overview
Introduction of Fezakinumab
Fezakinumab is a human IgG1-lambda type monoclonal antibody direct target at interleukin-22 (IL-22), which is a key factor of inflammatory disease. Fezakinumab was developed in an attempt to the treatment of psoriasis and rheumatoid arthritis. So far, fezakinumab has been used in the treatment of atopic dermatitis and rheumatoid arthritis. In 2018, a phase II trial was conducted to evaluate the efficacy and safety of fezakinumab in the treatment of moderate-to-severe atopic dermatitis. The results showed that fezakinumab had a well-tolerance and the clinical symptoms of the patients were improved. In addition, a phase II trial of the treatment for rheumatoid arthritis by fezakinumab combined with methotrexate has been completed, but the data have not yet been published.
Mechanism of Action of Fezakinumab
Interleukin-22 (IL-22), the target molecular of fezakinumab, is a member of the IL-10 cytokine family. L-22 can be secreted by helper T lymphocyte 17 (Th17) cells, helper T lymphocyte 22 (Th22) cells, natural killer cell 22 (NK22) and other immune cells, and mainly acts on non-hematopoietic endothelial cells and stromal cells. The most important function of IL-22 is to promote tissue proliferation and regeneration and regulate host mucosal barrier defense. However, IL-22 may also cause pathological inflammatory response. In recent years, many studies have shown that IL-22 plays a unique role in infection, autoimmune diseases and tumors. Its signaling mechanism involves the binding of IL-22 to interleukin-22 receptor 1 (IL-22R1) and the binding of this binary complex to the extracellular domain of interleukin-10 receptor 2 (IL-10R2). The therapeutic strategy for IL-22, a cytokine that is both an activator and an antagonist, may be an attractive therapeutic direction. Fezakinumab is a neutralizing antibody to IL-22, which treats some autoimmune diseases by blocking the binding of IL-22 to antibodies.
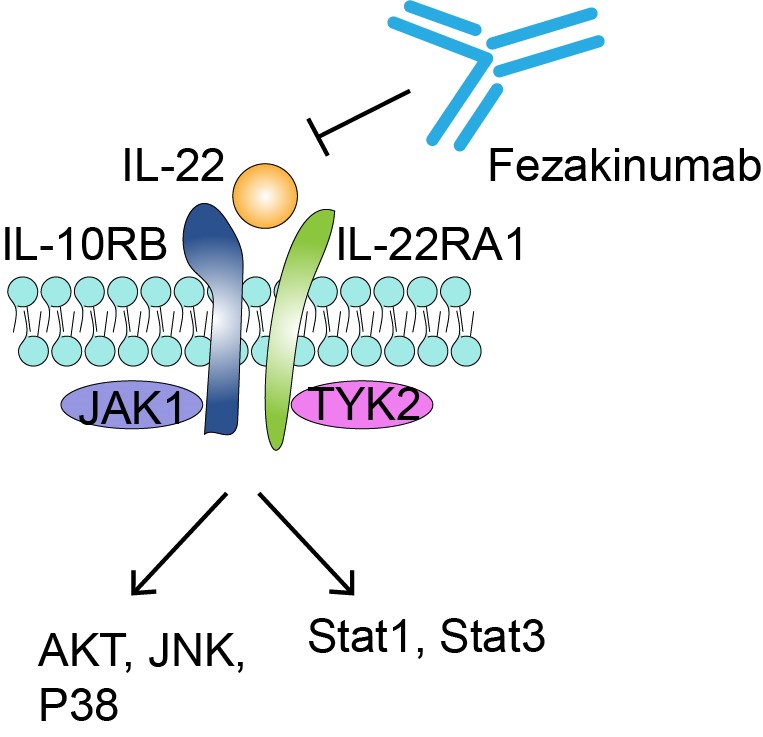 Fig.1 Mechanism of Action of Fezakinumab
Fig.1 Mechanism of Action of Fezakinumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

