Keliximab Overview
Introduction of Keliximab
Keliximab, from Macaca irus and Homo sapiens, is a recombinant chimeric immunoglobulin (Ig)G1 λ monoclonal antibody (mAb) that binds specifically to human CD4 antigen. This drug was designed for the treatment of severe chronic asthma. A randomized double-blind, placebo-controlled trial has been performed, evaluating the efficacy of a single infusion of keliximab in severe chronic corticosteroid dependent asthmatics and significant increases in morning and evening peak expiratory flow rates (PEFR) were observed in the highest dose cohort. Keliximab studies have provided evidence of the therapeutic potential of a non-depleting CD4 monoclonal antibody (mAb) in the treatment of rheumatoid arthritis (RA) and showed promise in the treatment of RA in phase II/III clinical trials.
Mechanism of Action of Keliximab
CD4 is a surface antigen found on several immune cell types, primarily on the helper/initiator subset of T lymphocytes. On T helper cells, CD4 acts in conjunction with the T cell receptor as an accessory protein by binding MHC Class II antigens (primarily on antigen presenting cells) and subtending protein kinase p56Ick signal transduction. Because of the central role of T helper cells as regulators of immune function, they are believed to play an important role in the pathogenesis of several immunologically mediated diseases, e.g., asthma and rheumatoid arthritis (RA), and anti-CD4 monoclonal antibodies are being explored as a therapeutic modality in these diseases. Keliximab is a primatized monoclonal antibody with human λ and γ1 constant regions directed toward domain 1 of human CD4. Keliximab was generated by immunizing a cynomolgus monkey with a truncated soluble form of human CD4 and then the cynomolgus immunoglobulin heavy and light chain variable regions were grafted onto human IgGl constant regions. This antibody binds with high affinity to human and chimpanzee CD4, induces CD4 receptor down modulation, and is a potent inhibitor of in vitro T cell responses. Keliximab is specific for human and chimpanzee CD4 and does not bind CD4 from other primates or rodents. The epitope recognized by keliximab is shared with that recognized by Leu 3a. It was hypothesized that keliximab's clinical effect related to the: 1) degree of coating of CD4; 2) reduced expression of CD4 antigen on the T-cell; 3) circulating concentrations of the antibody; 4) changes in activation markers; and 5) alteration of the T-helper cell Th1/Th2 cytokine profile. In addition, the in vitro effects of the antibody on the allergen-specific proliferation of peripheral blood mononuclear cells (PBMC) was evaluated from house-dust mite sensitive asthmatics. It was hypothesized that these would be decreased in a dose-dependent manner.
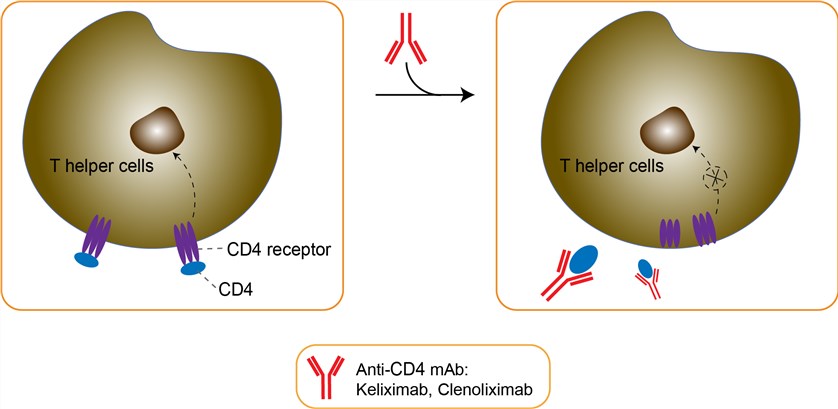 Fig 1. Mechanism of Action of Keliximab
Fig 1. Mechanism of Action of Keliximab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

