Carotuximab Overview
Introduction of Carotuximab
Carotuximab (TRC105) is a chimeric immunoglobulin G1 (IgG1) monoclonal antibody (mAb) targeting endoglin (CD105), a protein highly expressed on angiogenic and proliferative endothelial cells. Carotuximab received orphan drug designation for soft tissue sarcoma in the United States and the Europe Union. The Phase 3 TAPPAS (NCT02979899) is an open-label, randomized clinical trial evaluating TCR105 and pazopanib (a VEGF receptor tyrosine kinase inhibitor) versus pazopanib alone in patients with advanced angiosarcoma. Patients are randomly assigned to receive pazopanib 800 mg daily for adults (and 600 mg for 12-17 years old), and TCR105 (10 mg/kg weekly) or only pazopanib. The study has an adaptive design based on an interim analysis, which will determine the conditional power to meet the PFS endpoint. Based on the conditional power, the data monitoring committee will recommend continued accrual into 1 of 4 zones: 1) Favorable Zone: Continue study as planned (N = 124) to 95 events; 2) Promising Zone: Increase sample size to 200 patients with cutaneous and non-cutaneous disease and follow to 170 events ; 3) Enrichment Zone: Enroll patients with cutaneous disease only (N = 170) and follow to 110 events; and 4) Unfavorable Zone: Continue study as planned (N = 124) and follow to 95 events. The primary outcome measure is PFS; OS and ORR are secondary endpoints. The initial enrollment is 124 patients, but may expand up to a maximum of 200 patients. The estimated primary completion date of the study is September 2018. Carotuximab received US Food and Drug Administration (FDA)’s Fast Track designation for patients with advanced renal cell carcinoma in 2015, and it is undergoing evaluation in the Phase 2 TRAXAR study (NCT01806064) in combination with axitinib versus axitinib as monotherapy in this indication. The primary completion date for the study is December 2017.
Mechanism of Action of Carotuximab
CD105, a TGF-β co-receptor over-expressed on proliferating endothelial cells, plays a fundamental regulatory role in endothelial cell activation. It is overexpressed on endothelial cells of healing wounds, developing embryos, inflammatory tissues, and solid tumors, being a marker of activated endothelium, since its vascular expression is limited to proliferating cells. Dense CD105 expression on vessels is correlated with poor prognosis in many solid tumors including breast, lung, prostate, kidney, liver, and colon. Carotuximab binds human CD105 on proliferating endothelium with a KD of 1-2 ng/mL and induces antibody-dependent cell-mediated cytotoxicity (ADCC) of human umbilical vein endothelial cells. In addition, the binding of carotuximab to CD105 inhibits bone morphogenetic protein 9 (BMP9) binding and vascular endothelial growth factor (VEGF) and fibroblast growth factors (FGF)-induced angiogenesis.
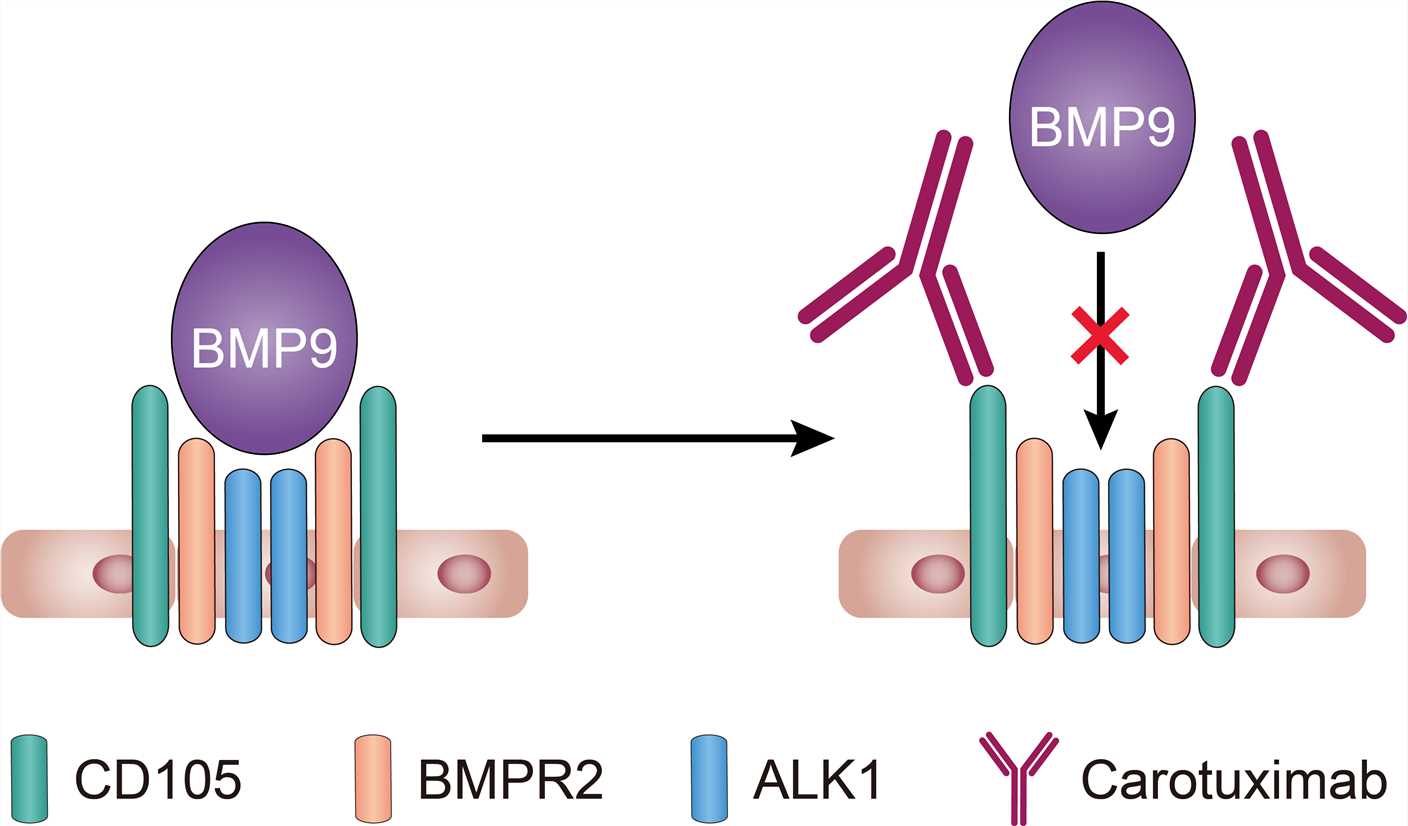
Fig.1 Mechanism of action of Carotuximab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

