Icrucumab Overview
Introduction of Icrucumab
Icrucumab (IMC-18F1) is a fully human immunoglobulin G1 (IgG1) monoclonal antibody (mAb) developed. for the treatment of solid tumors. Icrucumab targets and binds human vascular endothelial growth factor receptor-1 (VEGFR-1, also known as Flt-1) with high specificity and affinity. In preclinical studies, icrucumab monotherapy suppressed the growth of human breast-tumor xenografts in mice. In these tumor growth studies, icrucumab inhibited cellular activities associated with tumor survival by reducing mitogen-activated protein kinase (MAPK), reducing Akt activation, reducing tumor cell proliferation, and increasing tumor cell apoptosis. Because icrucumab does not bind to murine targets, a murine antibody (MF-1) was added to assure an effect on nontumor targets expressing VEGFR-1 (e.g., VEGFR-1-positive bone-marrow derived progenitor cells), and enhanced activity against tumor growth was observed when compared with icrucumab alone, suggesting that blockade of VEGFR-1 inhibits tumor growth through direct tumor targeting and hostderived supporting mechanisms. The tumor suppression properties of icrucumab were enhanced when icrucumab was administered in combination with chemotherapeutic agents used in breast cancer xenograft models.
Mechanism of Action of Icrucumab
Angiogenesis is a crucial process in the development of a closed circulatory system in vertebrates. Among the angiogenic factors described so far, vascular endothelial growth factor (VEGF)-A is the major player not only in physiological angiogenesis but also in pathological angiogenesis. VEGF-A binds and activates two tyrosine kinase receptors, VEGF receptor 1 (VEGFR-1) and VEGFR-2. VEGFR-1 is a kinase-impaired receptor tyrosine kinases (RTK) and belongs to the VEGFR family. It binds VEGF-A, placenta growth factor (PlGF), and VEGF-B. An important feature of VEGFR-1 is that, unlike other VEGFR genes, it expresses two types of mRNA, one for a full-length receptor and another for a soluble short protein known as soluble VEGFR-1 (sFlt-1). The binding-affinity of VEGFR-1 for VEGF-A is one order of magnitude higher than that of VEGFR-2, whereas the kinase activity of VEGFR-1 is about 10-fold weaker than that of VEGFR-2. Through its ligand-binding region and by trapping ligands, VEGFR-1 plays a negative role in angiogenesis at embryogenesis. In adulthood, however, VEGFR-1 is expressed not only on endothelial cells but also on macrophages, and promotes the function of macrophages, inflammatory diseases, cancer metastasis, and atherosclerosis via its kinase activity. Soluble VEGFR-1 is abnormally overexpressed in the placenta of preeclamptic patients, and suggested to cause the major pathological symptoms on the maternal side such as hypertension and renal dysfunction, most likely by blocking the physiological VEGF-A. The downstream signaling of VEGFR-1 is not fully understood mainly due to the mild biological activity of this receptor in culture, although PI3-kinase is one of the candidates for the activation and signaling. Icrucumab targets and binds human VEGFR-1, which blocks the VEGF/VEGFR-1 interaction, inhibiting the cellular activities associated with tumor survival.
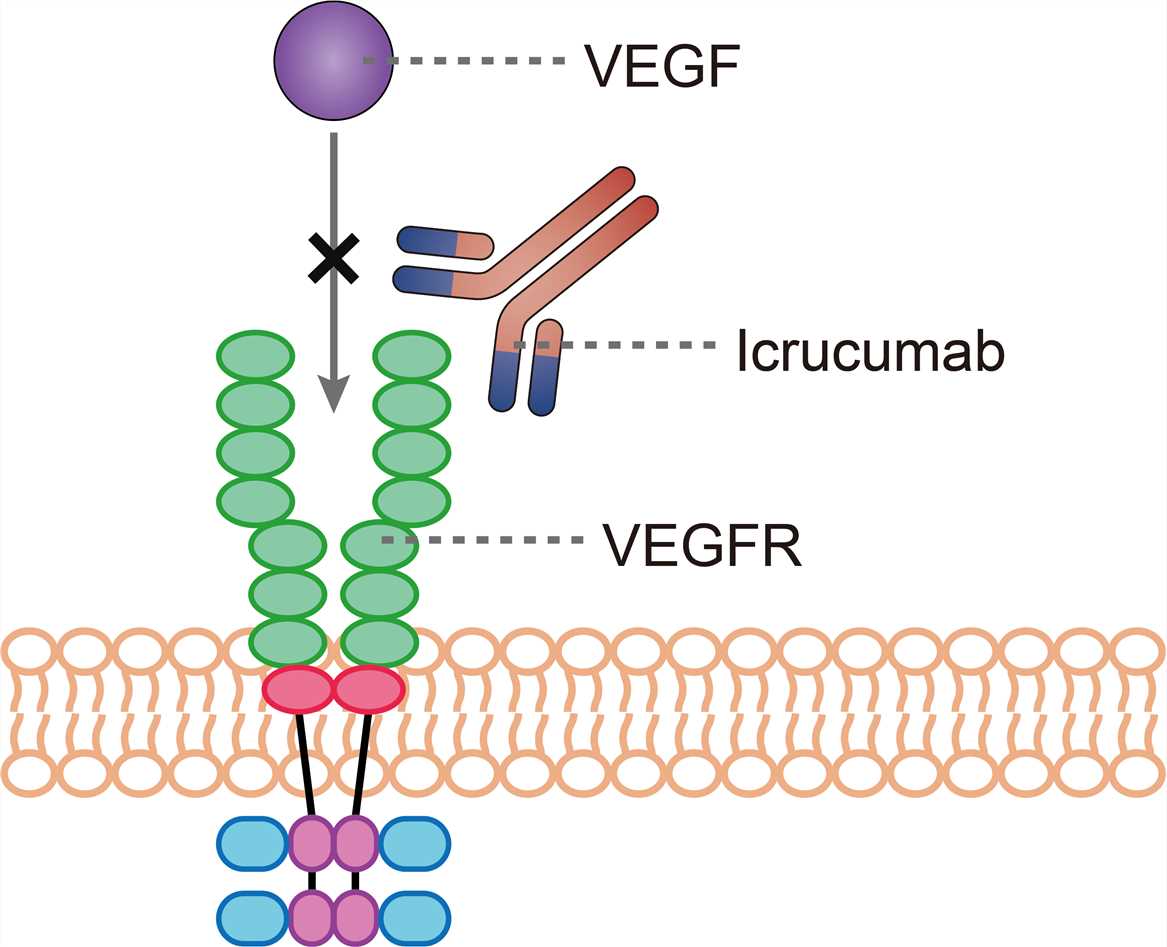
Fig.1 Mechanism of action of Icrucumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

