Cantuzumab Ravtansine Overview
Introduction of Cantuzumab Ravtansine
Cantuzumab ravtansine, also known as IMGN242 or huC242-DM4, is an innovative antibody-drug conjugate (ADC) that targets the overexpressed MUC1 antigen. MUC1 is a high-molecular-weight transmembrane mucin found in various malignancies, making it a viable target for cancer treatment. Cantuzumab ravtansine represents a significant advancement in targeted cancer therapeutics, consisting of a humanized monoclonal antibody (huC242) covalently linked to the cytotoxic drug DM4 (a potent derivative of maytansinoid) via a disulfide bond. This linkage ensures the precise delivery of DM4 to cancer cells expressing the MUC1 antigen.
Cantuzumab ravtansine has undergone clinical trials to evaluate its safety, pharmacokinetics, and therapeutic efficacy across various cancer types. These trials have specifically focused on its potential as a treatment for MUC1-positive malignancies. The clinical trials involve dosage-escalation studies to identify the maximum tolerable dose and to detect early signs of anticancer activity, although specific details of these trials are not provided here. Like other antibody-drug conjugates (ADCs), developing has presented challenge s, including minimizing off-target toxicities, ensuring stable linker technology to maintain ADC integrity until it reaches the tumor site, and optimizing the drug-to-antibody ratio (DAR). Furthermore, choosing appropriate patients is essential, as a treatment response can only be achieved when the MUC1 antigen is present.
In oncology, cantuzumab ravtansine exhibits promising therapeutic promise. Its unique approach harnesses the precise targeting capability of monoclonal antibodies to deliver a potent cytotoxic payload straight to cancer cells. While the clinical development of this ADC is ongoing, it offers a potentially more precise and effective treatment option for patients with MUC1-expressing malignancies. Continued research into the safety, efficacy, and optimal dosing regimen of cantuzumab ravtansine will be crucial for advancing this therapeutic approach.
Biological and Chemical Properties of Cantuzumab Ravtansine
Protein Structure
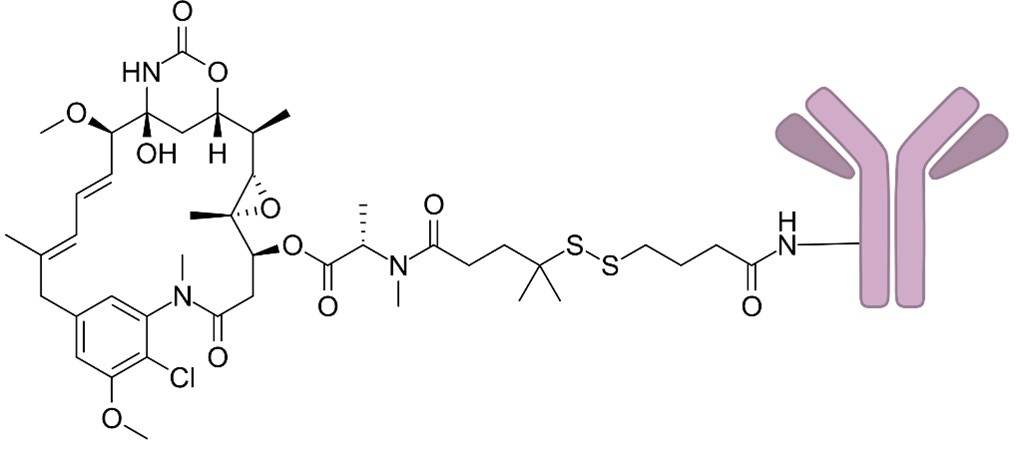 Figure 1. The Structure of Cantuzumab Ravtansine (Wikipedia)1
Figure 1. The Structure of Cantuzumab Ravtansine (Wikipedia)1
Protein Chemical Formula
C6570H10130N1726O2018S44(C42H59ClN3O11S2)n
The Mechanism of Cantuzumab Ravtansine Action
Cantuzumab ravtansine consists of cantuzumab, a monoclonal antibody, and ravtansine (DM4), a potent cytotoxic agent. The mechanism of action begins with the huC242 antibody specifically binding to the MUC1 antigen on the surface of cancer cells. This binding triggers the internalization of the ADC-antigen complex into the cell via endocytosis. Once inside the cell, the disulfide bond is cleaved, releasing the DM4 payload. As a potent inhibitor of microtubule assembly, DM4 disrupts the microtubule dynamics required for mitotic spindle formation, leading to cell cycle arrest and cell death in the targeted cancer cells. This mechanism allows cantuzumab ravtansine to specifically target cancer cells and deliver a highly toxic agent directly to them.
The absorption of cantuzumab ravtansine occurs through receptor-mediated endocytosis following its binding to CanAg, a new glycoform of MUC1. After entering the target cell, the conjugate proceeds to the lysosome, an organelle responsible for degradation. The acidic environment within the lysosome facilitates the cleavage of the linker connecting the antibody and the cytotoxic chemical, releasing DM4. DM4 then binds to tubulin, disrupting microtubule dynamics and causing cytotoxicity. This disruption leads to cell cycle arrest at the G2/M phase, ultimately resulting in apoptosis. The targeted delivery of DM4 by the cantuzumab component minimizes off-tissue toxicity, increasing the agent's therapeutic index. Numerous preclinical models and clinical trials have evaluated the specificity and efficacy of cantuzumab ravtansine, showing significant anti-tumor activity while maintaining a tolerable safety profile. The ongoing clinical development of cantuzumab ravtansine indicates its potential as a valuable therapeutic tool, particularly for malignancies with high CanAg antigen expression. Further research and development may expand its applicability to a broader range of neoplastic diseases.
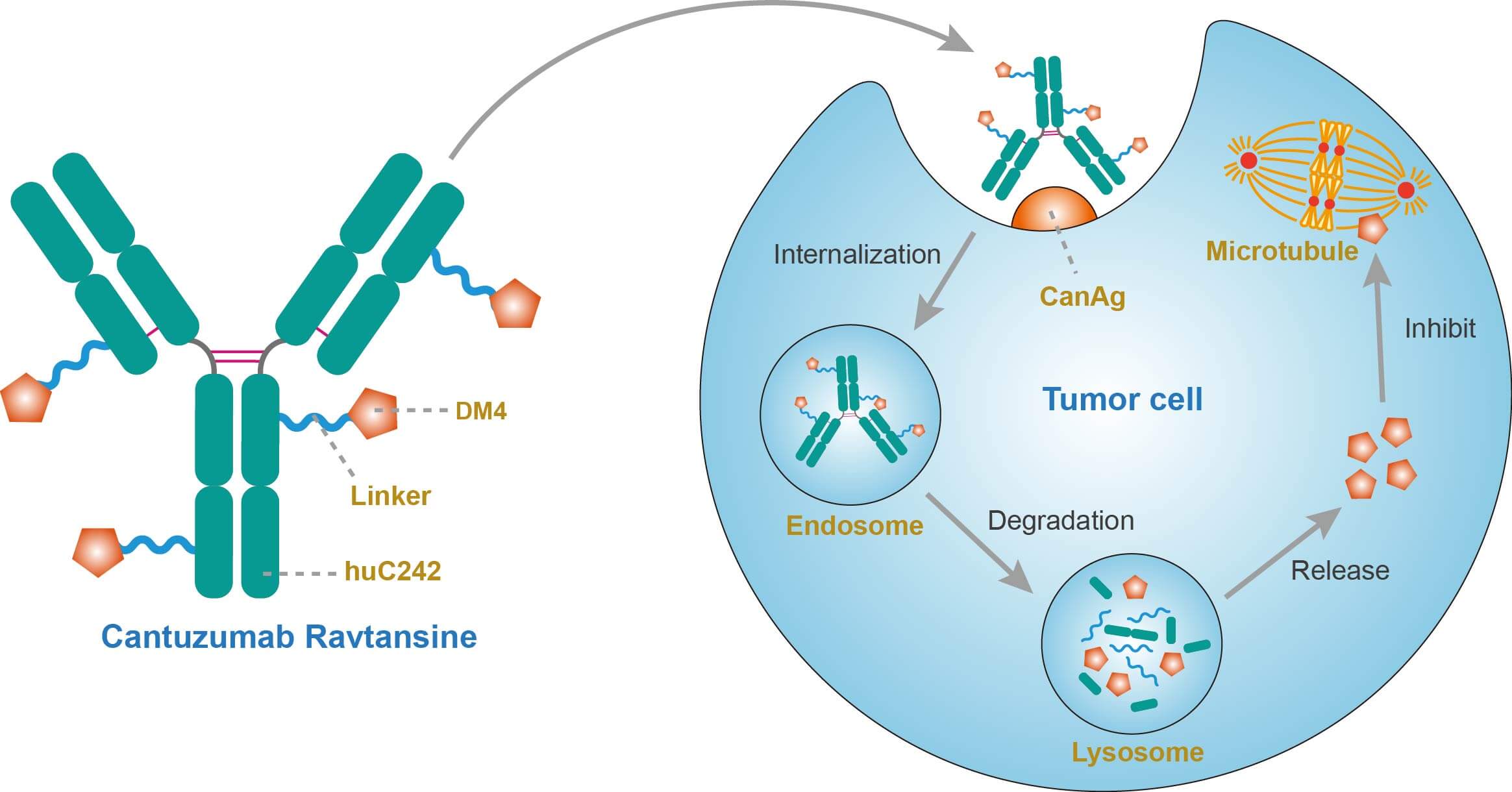 Figure 2. The Mechanism of Cantuzumab Ravtansine Action (Creative Biolabs Original)
Figure 2. The Mechanism of Cantuzumab Ravtansine Action (Creative Biolabs Original)
Clinical Projects of Cantuzumab Ravtansine*
| NCT ID | Study Title | Study Status | Conditions | Sponsor | Start Date |
| NCT00352131 | Maytansinoid DM4-Conjugated Humanized Monoclonal Antibody huC242 in Treating Patients With Solid Tumors | COMPLETED |
Non-colorectal Cancer Pancreatic Cancer |
ImmunoGen, Inc. | 2005-02 |
* The table is excerpted from the following website: https://clinicaltrials.gov/search?cond=Cantuzumab%20Ravtansine
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

