Duligotumab Overview
Introduction of Duligotumab
Duligotumab, also known as MEHD7945A, duligotuzumab and RG7597, is a specially engineered monoclonal antibody targeting both ERBB3 and EGFR. ERBB3, also referred to as HER3, is a member of the ErbB family of receptor tyrosine kinases, which includes EGFR (HER1/ERBB1), HER2 (ERBB2), and HER4 (ERBB4). These receptors play a crucial role in regulating cell growth, survival, and differentiation. Dysregulation of these receptors is often associated with various cancers. Duligotumab not only blocks the signaling pathways mediated by ERBB3 and EGFR but also enhances ADCC, leveraging the body's immune effector functions. Importantly, this enhanced ADCC mechanism does not seem to significantly increase the risk of severe immune-related adverse events (AEs), making duligotumab a viable option for long-term therapy. Ongoing advancements in antibody engineering continue to refine the therapeutic potential of molecules like duligotumab. Current research is exploring the combination of duligotumab with immune checkpoint inhibitors such as anti-PD-1 or anti-CTLA-4 antibodies. This drug combination aims to further potentiate the immune response against tumors by both relieving immunosuppressive checkpoints and targeting ERBB3-expressing cells. Additionally, identifying predictive biomarkers for patient selection is anticipated to improve the clinical benefits of duligotumab. Advanced genomic and proteomic profiling will probably play a crucial role in enabling a more personalized approach to cancer therapy.
Biological and Chemical Properties of Human ERBB3
Protein Structure
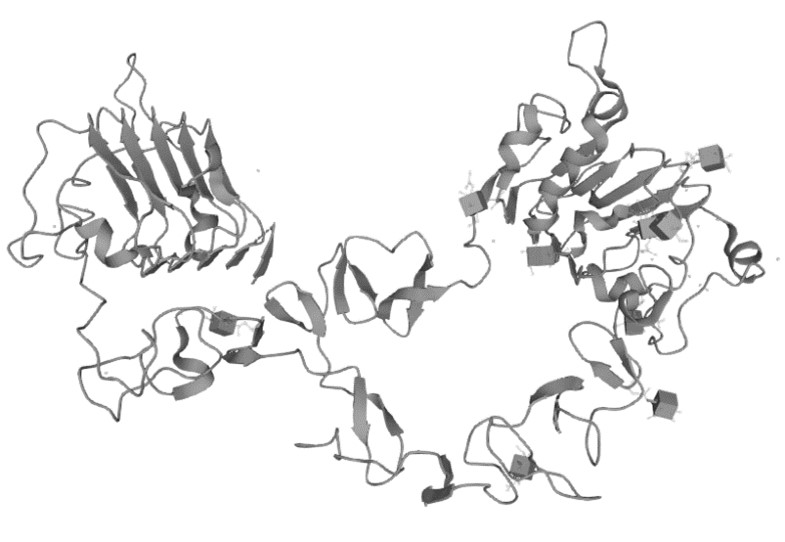 Figure 1. The Structure of Human ERBB3 (UniProt)1,2
Figure 1. The Structure of Human ERBB3 (UniProt)1,2
The Mechanism of Duligotumab Action
ERBB3 and EGFR Targeting and Inhibition of Downstream Signaling
ERBB3 and EGFR are members of the ErbB family of receptor tyrosine kinases. ERBB3 is unique because it lacks intrinsic kinase activity. It relies on dimerization with other family members like HER2, to activate downstream signaling pathways, including the PI3K/AKT, JAK/STAT, and MAPK pathways. These pathways are crucial for cell proliferation and survival. Duligotumab exerts its inhibition through binding to the extracellular domain of ERBB3 and EGFR, which turns to effectively prevent the formation of indispensable signaling complexes. This inhibition reduces tumor cell proliferation, survival, and metastatic potential.
Enhancement of Antibody-Dependent Cellular Cytotoxicity (ADCC)
ADCC is an immune mechanism in which antibody-coated target cells are destroyed by immune cells, such as Natural Killer (NK) cells. This process is mediated by the interaction between the Fc region of antibodies and Fc receptors (FcγRIII) on the surface of effector cells. Duligotumab is engineered to enhance this interaction by modifying its Fc region, leading to improved binding to FcγRIII receptors. As a result, NK cells are more effectively recruited to tumor cells, increasing tumor cell lysis and cell death. This enhancement of ADCC amplifies the immune system's ability to selectively target and eliminate cancerous cells, providing a dual mechanism of action alongside receptor blockade.
Modulation of the Tumor Microenvironment
Beyond its direct tumor-targeting actions, duligotumab may also influence the tumor microenvironment (TME). By depleting ERBB3-expressing cells, duligotumab can potentially reduce immunosuppressive signals within the TME, promoting a more immune-permissive environment. This modulation is particularly relevant in tumors where ERBB3 expression correlates with poor immune infiltration.
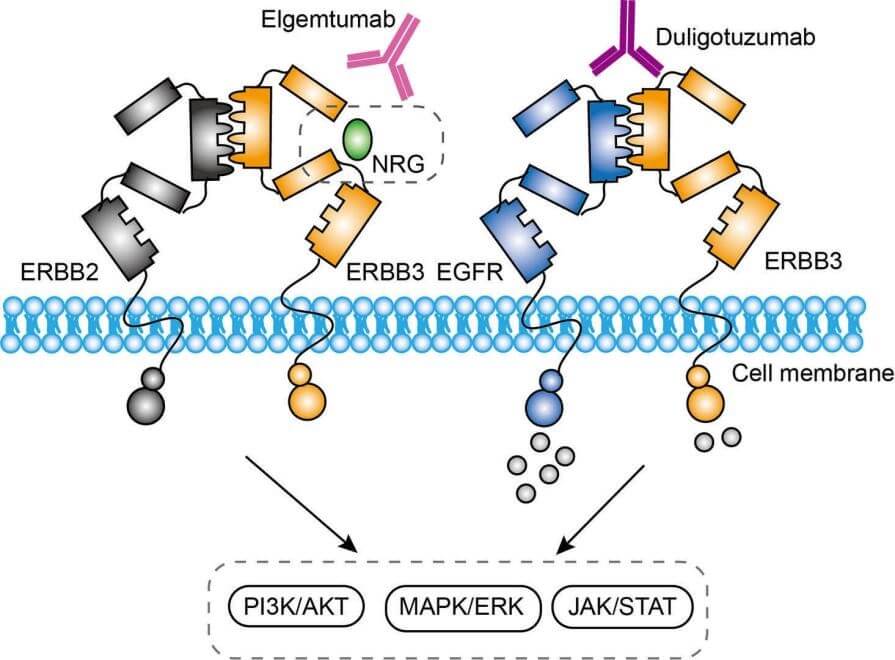 Figure 2. The Mechanism of Duligotumab Action (Creative Biolabs Original)
Figure 2. The Mechanism of Duligotumab Action (Creative Biolabs Original)
Clinical Applications of Duligotumab
The primary clinical application of duligotumab lies in oncology, where ERBB3 is often overexpressed or dysregulated. Results from current studies demonstrate promising improvements in progression-free survival (PFS) and overall survival (OS), especially in patients with high ERBB3 expression. Several cancer types, including breast cancer, lung cancer, and head and neck squamous cell carcinoma, exhibit upregulation of ERBB3. Recent studies have explored the efficacy of duligotumab in combination with other therapeutic agents, such as EGFR inhibitors and chemotherapeutics. The combined therapy applied in HER2-positive breast cancer is a good example. The addition of duligotumab to existing treatment regimens has shown synergistic effects that blocking both HER2 and ERBB3 disrupts critical growth and survival pathways, which eventually enhances antitumor activity. Beyond well-established oncological indications, there is growing interest in exploring duligotumab's potential in other malignancies expressing ERBB3. Early-phase clinical studies are investigating its efficacy in colorectal, ovarian, and pancreatic cancers. Moreover, preclinical research is ongoing to understand the role of ERBB3 in hematological malignancies and the potential for extending duligotumab therapy to these cancers.
Clinical Projects of Duligotumab*
| NCT ID | Study Title | Study Status | Conditions | Sponsor | Start Date |
| NCT01986166 | A Study of MEHD7945A and Cobimetinib in Patients With Locally Advanced or Metastatic Cancers With Mutant KRAS | COMPLETED | Neoplasms | Genentech, Inc. | 2013-11 |
| NCT01207323 | A Study of the Safety and Pharmacokinetics (PK) of MEHD7945A in Participants With Locally Advanced or Metastatic Epithelial Tumors | COMPLETED | Epithelial Tumors, Malignant | Genentech, Inc. | 2010-11-09 |
| NCT01652482 | Safety and Efficacy Study of MEHD7945A + FOLFIRI Versus Cetuximab + FOLFIRI as Second Line Therapy in Participants With KRAS Wild-Type Metastatic Colorectal Cancer (mCRC) | COMPLETED | Colorectal Cancer | Genentech, Inc. | 2012-10 |
* The table was excerpted from the following website: https://clinicaltrials.gov/search?cond=%20MEHD7945A
- Uniprot Database (https://www.uniprot.org/uniprotkb/P21860/entry#sequences)
- The image was retrieved from UniProt Database and used under [CC BY 4.0]. It was not modified and the titles was " The Structure of Human ERBB3 (UniProt)".
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

