Foralumab Overview
Introduction of Foralumab
Foralumab (TZLS‐0401; known formerly as NI‐0401) is a fully human monoclonal antibody that binds to the epsilon (ε) chain of the CD3/TCR complex present at the surface of all peripheral T cells. An amino acid sequence for both heavy and light chains has been identified from the translation of the nucleotide sequence to encode foralumab. It is composed of two heavy chains with an IgG1 constant region and two light chains with a kappa constant region. The heavy chain constant regions are mutated at two amino acid positions, 234 and 235. Foralumab is the only fully engineered anti-human CD3 antibody in clinical development to date. This phase 2 clinical compound has potential applications for a wide range of autoimmune and inflammatory diseases such as ulcerative colitis, multiple sclerosis, type 1 diabetes, inflammatory bowel disease, psoriasis and rheumatoid arthritis.
Mechanism of Action of Foralumab
Non-alcoholic fatty liver disease (NAFLD) affects 20–40% of the population. Its active form, Non-alcoholic steatohepatitis (NASH), is characterized by hepatocyte injury, liver inflammation and progression of fibrosis. NASH has emerged as an important cause of chronic liver disease and hepatocellular carcinoma (HCC) worldwide. The immune system plays an important role in the pathogenesis of NASH and underlies the hepatocyte injury and fibrosis progression in all disease stages.T cells recognize antigens through the T cell receptor (TCR), which is associated with the CD3 molecule. The CD3/TCR complex is present on the surfaces of all T cells and is involved in antigen recognition and signal transduction. Whereas TCR chains are subject to gene rearrangement and are variable, CD3 chains are invariable and are not antigen‐specific. The oral administration of anti‐CD3 monoclonal antibody (mAb) is a method developed for systemic immune modulation by the induction of regulatory T cells. Foralumab, a long half-life therapeutic mAb candidate, with high affinity and potency for CD3 epsilon, is a unique oral therapy that stimulates the gut immune system and provides an anti-inflammatory therapeutic effect in autoimmune diseases without immune suppression. Foralumab passes intact through the stomach and small bowel. The combination of the two points mutations introduced into the Fc portion of foralumab results in the abrogation of the binding to FcγRs and Clq and consequently eliminates T cell proliferation and the release of cytokines, including TNF‐α and IFN‐γ, in vitro. Foralumab decreased the proliferative responses of CD8 + cells, increased serum IL‐10 levels and decreased the release of TNF‐α, thus eliminating T cell proliferation and the release of proinflammatory cytokines.
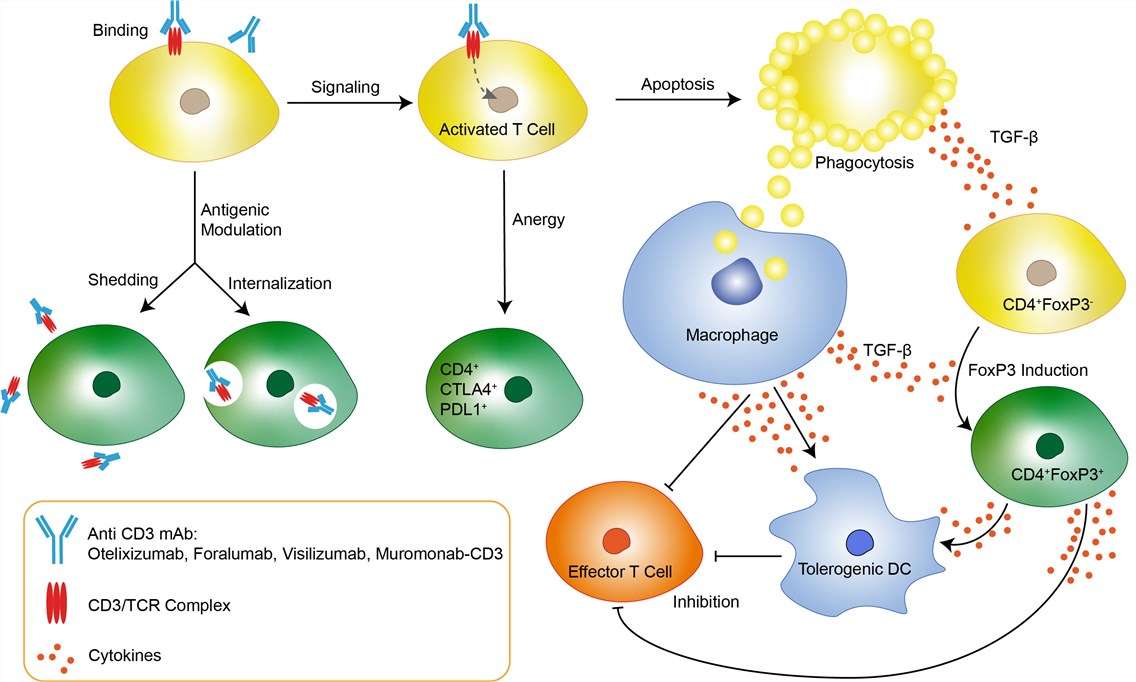 Fig 1. Mechanism of Action of Foralumab
Fig 1. Mechanism of Action of Foralumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

