Tesidolumab Overview
Introduction of Tesidolumab
Tesidolumab, as known as LFG-316, is a human immunoglobulin G1 (IgG1) monoclonal antibody directed against the complement pathway protein C5, with complement pathway inhibitory activity and potential immunomodulating activity. The drug has been used in trials studying the treatment of Geographic Atrophy, Non-infectious Panuveitis, Exudative Macular Degeneration, Non-infectious Posterior Uveitis, and Age-related Macular Degeneration, among others. In addition, it has reached Phase II clinical trial as a potential therapy for the ocular conditions, geographic atrophy, wet age-related macular degeneration and non-infectious uveitis.
Mechanism of Action of Tesidolumab
Geographic atrophy (GA), also known as atrophic age-related macular degeneration (AMD) or advanced dry AMD, is an advanced form of age-related macular degeneration that can result in the progressive and irreversible loss of retina (photoreceptors, retinal pigment epithelium, choriocappillaris) which can lead to a loss of visual function over time. It is estimated that GA affects >5 million people worldwide and approximately 1 million patients in the US, which is similar to the prevalence of neovascular (wet) AMD, the other advanced form of the disease. The incidence of advanced AMD, both geographic atrophy and neovascular AMD, increases exponentially with age and while there are therapies for wet AMD, GA currently has no approved treatment options. The aim of most current clinical trials is to reduce the progression of GA lesion enlargement. Complement component 5 is the fifth component of complement, which plays an important role in inflammatory and cell killing processes. This protein is composed of alpha and beta polypeptide chains that are linked by a disulfide bridge. An activation peptide, C5a, which is an anaphylatoxin that possesses potent spasmogenic and chemotactic activity, is derived from the alpha polypeptide via cleavage with a C5-convertase. The C5b macromolecular cleavage product can form a complex with the C6 complement component, and this complex is the basis for formation of the membrane attack complex, which includes additional complement components. C5 plays a key role in the activation of the complement cascade. Tesidolumab is a human immunoglobulin G1 (IgG1) monoclonal antibody directed against the complement pathway protein C5. Upon administration, tesidolumab targets and binds to C5, thereby preventing both C5 convertase-mediated cleavage of C5 and the formation of C5a and C5b. This inhibits C5-mediated signal transduction, the formation of the membrane attack complex (MAC) and the activation of the terminal complement pathway, and results in the prevention and/or inhibition of both complement-mediated inflammation and cell destruction.
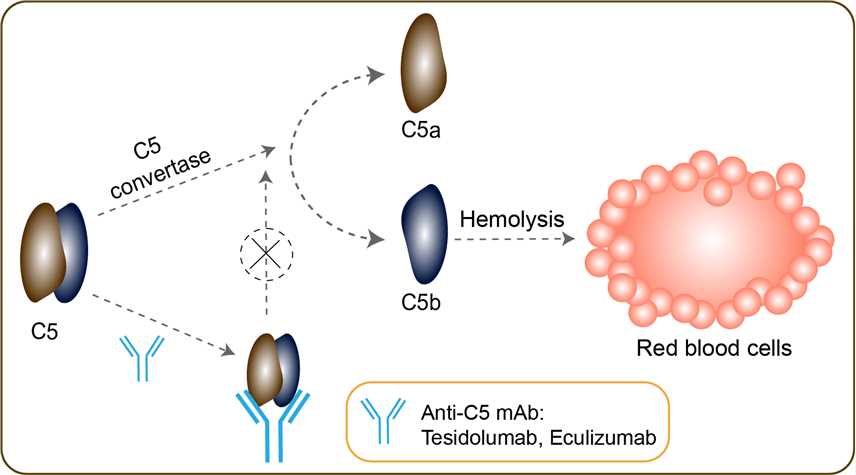
Fig 1. Mechanism of Action of Tesidolumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

