Zalutumumab Overview
Introduction of Zalutumumab
Zalutumumab is a high affinity, completely human IgG1 monoclonal antibody (mAb) directed towards the epidermal growth factor receptor (EGFR, also known as ErbB1/HER1). It is designed for the treatment of squamous cell carcinoma of the head and neck (SCCHN), a type of cancer. Zalutumumab is derived from transgenic mice immunized with A431-derived EGFR. In 2009, zalutumumab treatment was approved for Fast Track status by the U.S. Food and Drug Administration (FDA) for patients suffering from SCCHN who have failed standard therapies and have no other options. The drug has undergone pre-clinical and Phase I and II studies and is also in Phases I and II for SCCHN front-line with chemo-radiation and SCCHN with radiation. Additionally, a Phase II is under way for SCCHN and Phase III studies are also being performed for SCCHN and SCCHN front-line with radio therapy. In 2014, a study of zalutumumab as addition to chemoradiation for SCCHN showed no benefit, and 94% developed a skin rash (11% severe enough to discontinue).
Mechanism of Action of Zalutumumab
EGFR is a 170 kDa transmembrane glycoprotein composed of a single polypeptide chain. It belongs to the ErbB receptor family, also known as the EGF receptor family. The binding of EGF at the cell surface induces the dimerization of EGFR, which results in the activation of EGFR tyrosine kinase activity and receptor trans-autophosphorylation. Sites of tyrosine autophosphorylation in the activated EGFR interact with downstream signaling proteins to form large signaling complexes. The receptor-signaling protein complexes then initiate the activation of various signaling pathways, and eventually stimulate cell proliferation and survival. Zalutumumab acts via two distinct mechanisms. First, as an inverse agonist, it specifically binds to the EGFr domain III on the cell surface and competitively inhibits the ligand binding of EGF and TGF-a to EGFR. It then blocks receptor activation by restricting the molecule’s conformational flexibility: In the inactive conformation, the intracellular tyrosine kinase domains of EGFR molecules are spatially separated to an extent that prevents receptor dimerization and phosphorylation. The EGFR cascade cannot be started. This mechanism was found to be effective in established xenograft models at doses giving full receptor occupancy. Second, in contrast to panitumumab, which is an IgG2 antibody, Zalutumumab has been shown to arrest tumor growth and induce tumour regression in animal disease models even at very low, subsaturation antibody concentration. This antitumor effect is mediated by antibody-dependent cellular cytotoxicity (ADCC): The fragment antigen binding (Fab) region of the antibody binds to the antigen on EGFR-expressing tumor cells. Through an immunological response, the body’s NK lymphocytes recognize and bind to the Fc portion on the antibody through an Fc receptor, CD16. The NK cell is then activated through cross-linking of Fc receptors which sends a signal to induce apoptosis. A recent study showed that this mechanism of action might be important in preventing tumour outgrowth or metastasis in vivo, even in cancers insensitive to EGFR signaling inhibition.
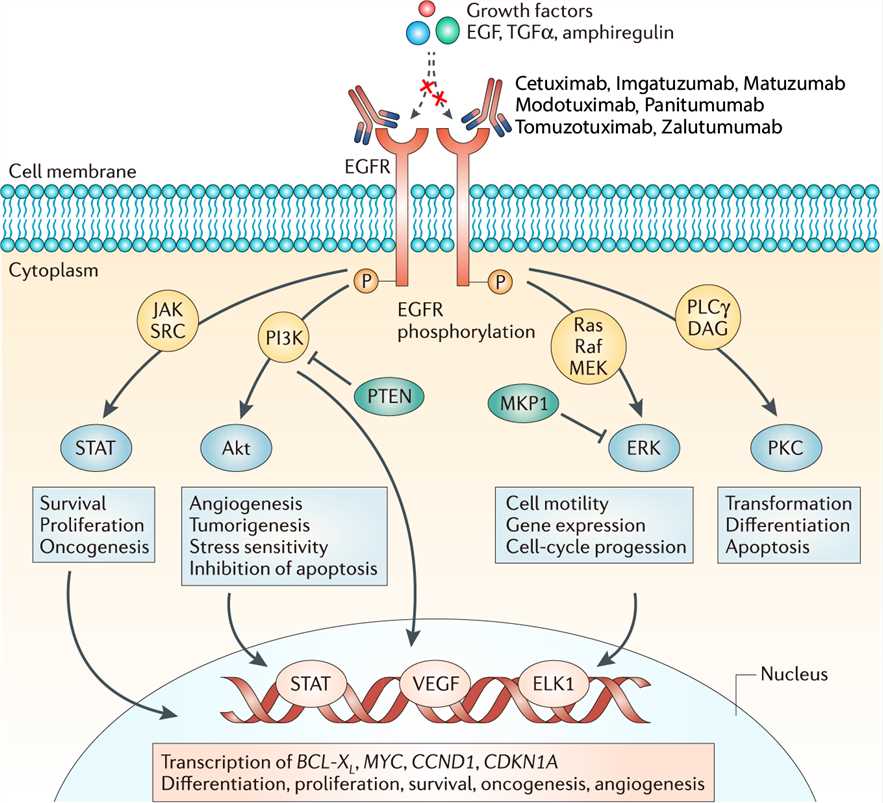
Fig.1 Mechanism of action of Zalutumumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

