mTOR Signaling Pathway
About mTOR Pathway
The mammalian target of rapamycin (mTOR), also known as the mechanistic target of rapamycin and FK506-binding protein 12-rapamycin-associated protein 1 (FRAP1), is a kinase that in humans is encoded by the MTOR gene. mTOR signaling pathway integrates both intracellular and extracellular signals and serves as a central regulator of cell metabolism, growth, proliferation and survival. Discoveries that have been made over the last decade show that the mTOR pathway is activated during various cellular processes (e.g. tumor formation and angiogenesis, insulin resistance, adipogenesis and T-lymphocyte activation) and is deregulated in human diseases such as cancer and type 2 diabetes. These observations have attracted broad scientific and clinical interest in mTOR. This is highlighted by the growing use of mTOR inhibitors in pathological settings, including the treatment of solid tumors, organ transplantation, coronary restenosis and rheumatoid arthritis. The mTOR protein belongs to the phospho-inositide 3-kinase (PI3K)-related kinase family and is conserved throughout evolution. mTOR nucleates at least two distinct multi-protein complexes, mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2). mTORC1 has five components: mTOR, Raptor, mLST8 (also known as GβL), PRAS40 and Deptor. mTORC2 comprises six different proteins, several of which are common to mTORC1, mTOR, Rictor, mSin1, Protor-1, mLST8 and Deptor. mTORC1 positively regulates cell growth and proliferation by promoting many anabolic processes, including biosynthesis of proteins, lipids and organelles, and by limiting catabolic processes such as autophagy. Much of the knowledge about mTORC1 function comes from the use of the bacterial macrolide rapamycin. Upon entering the cell, rapamycin binds to FKBP12 and interacts with the FKBP12-rapamycin binding domain (FRB) of mTOR, thus inhibiting mTORC1 functions. In contrast to its effect on mTORC1, FKBP12-rapamycin cannot physically interact with or acutely inhibit mTORC2. In contrast to mTORC1, for which many upstream signals and cellular functions have been defined, relatively little is known about mTORC2 biology. The early lethality caused by the deletion of mTORC2 components in mice, as well as the absence of mTORC2 inhibitors, have complicated the study of this protein complex. Nonetheless, many important discoveries have been made over the last few years. Using various genetic approaches, it has been demonstrated that mTORC2 plays key roles in various biological processes, including cell survival, metabolism, proliferation and cytoskeleton organization.
Several observations support the importance of mTOR pathway in cancer pathogenesis. Many components of the PI3K signaling pathway, which is upstream of both mTORC1 and mTORC2, are mutated in human cancers. Additionally, the loss of p53, a very common event in cancer, promotes mTORC1 activation. In addition, several familial cancer syndromes arise from mutations in genes encoding proteins that lie upstream of the mTOR complexes. Oncogenic activation of mTOR signaling induces several processes required for cancer cell growth, survival, and proliferation. Deregulation of protein synthesis downstream of mTORC1 at the level of 4E-BP1/eIF4E plays a central role in tumor formation. Loss of 4EBP1/2 and the concomitant activation of cap-dependent translation promotes cell cycle progression and cell proliferation in culture. 4E-BP1/eIF4E also mediates the effects of oncogenic Akt signaling on mRNA translation, cell growth, and tumor progression. It is thought that eiF4E affects cell proliferation and tumorigenesis by promoting the translation of specific mRNAs coding for pro-oncogenic proteins regulating cell survival, cell cycle progression, angiogenesis, energy metabolism, and metastasis. SREBP1 also drives the expression of components of the oxidative branch of the pentose phosphate pathway, which controls the production of the reducing equivalents and ribose-5-phosphate needed for lipogenesis and nucleotide biosynthesis, respectively. PI3K signaling promotes the activation of the pro-lipogenic factor SREBP1 and mTORC1 is required to relay oncogenic/growth factor signaling to SREBP1. Many gliomas overexpress the mTORC2 subunit Rictor and its forced overexpression promotes mTORC2 assembly and activity and endows cancer cells with increased proliferative and invasion potential. In mice, the development of prostate cancer induced by the loss of the tumor suppressor PTEN requires mTORC2 function. These results support an important role of mTORC2 in promoting tumorigenesis and suggest that strategies aimed to reduce the activity of this complex could have roles as anti-cancer therapies.
Associated Diseases
Related Pathways
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


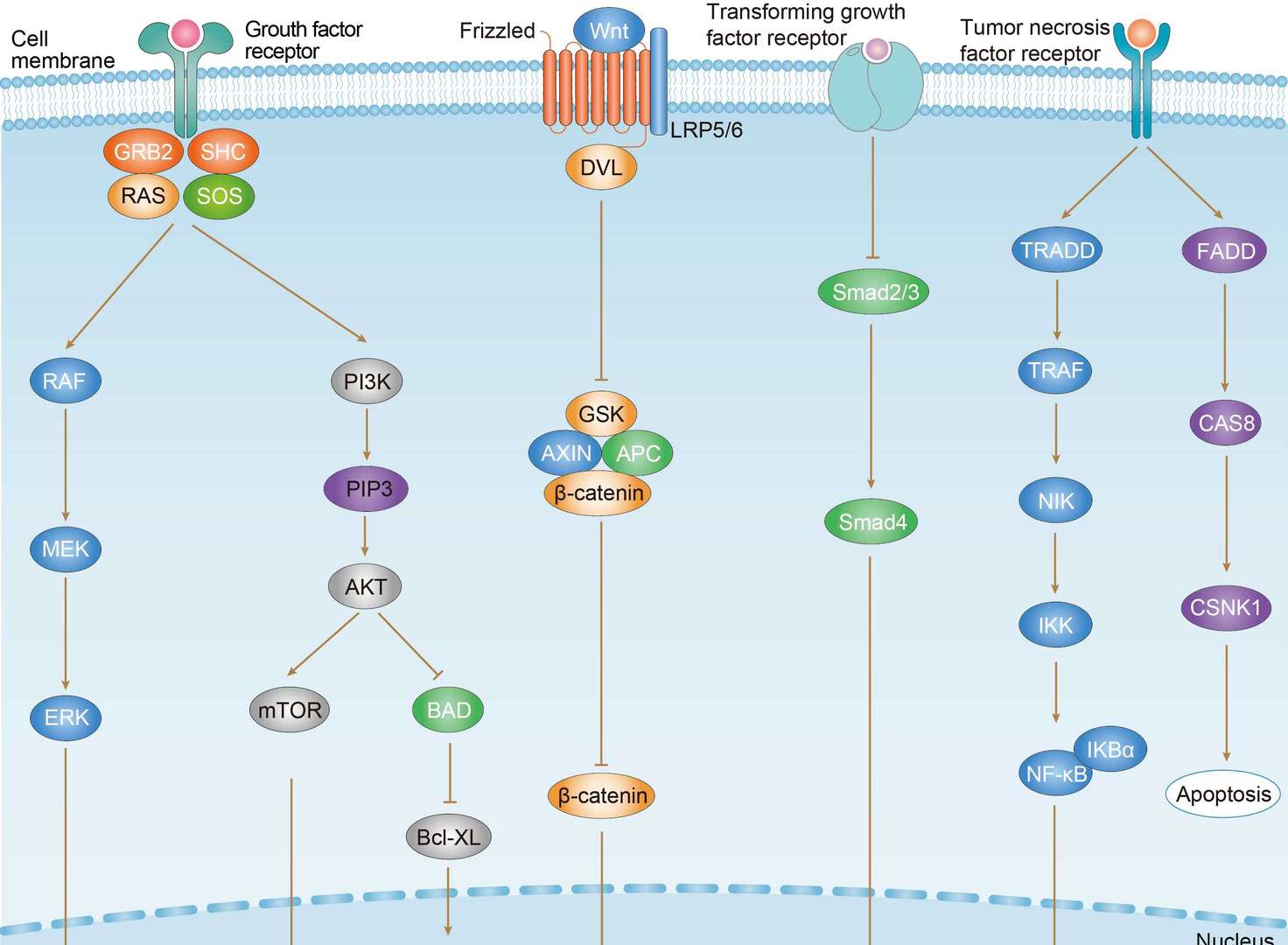 Colorectal Cancer
Colorectal Cancer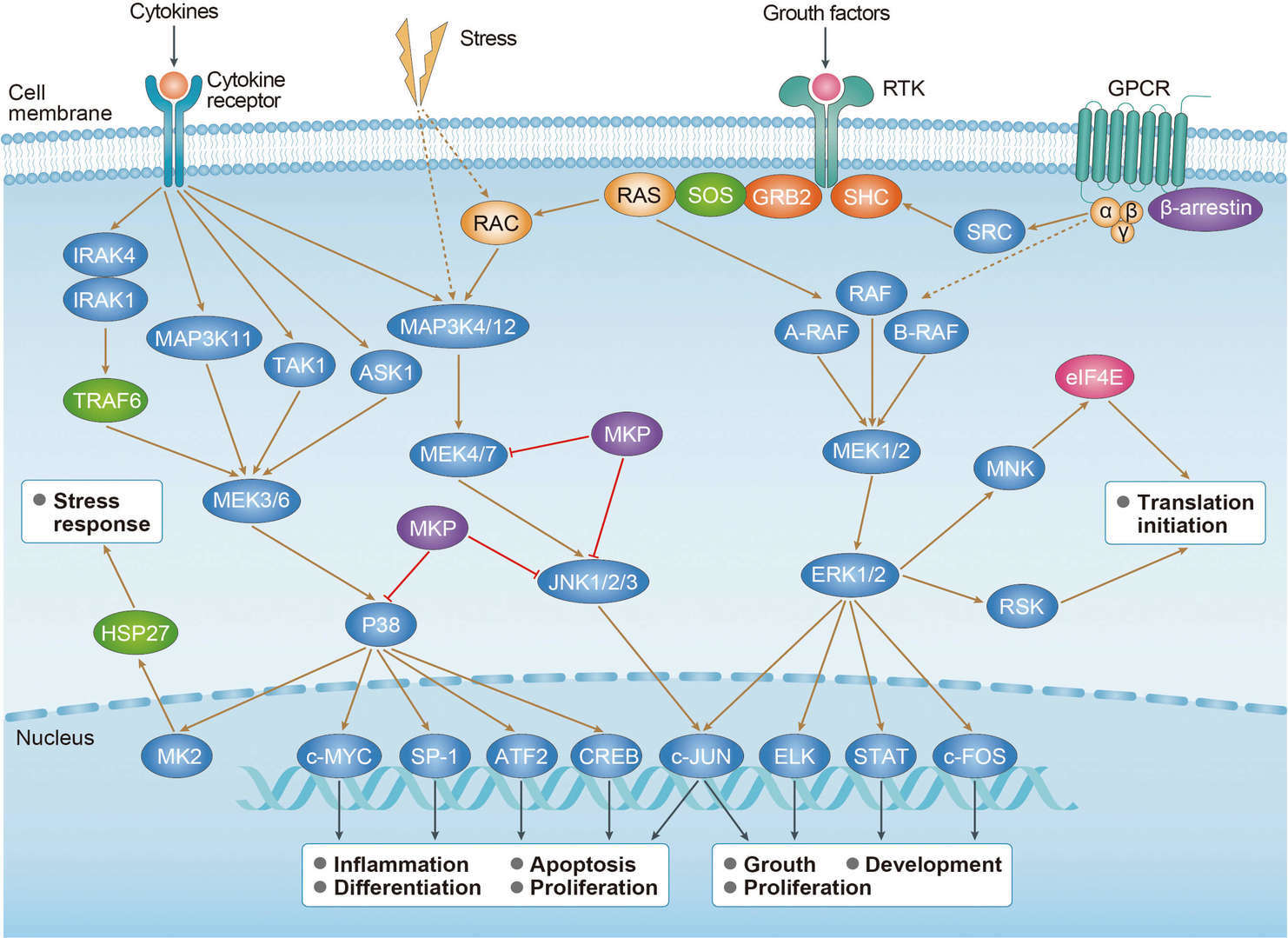 MAPK Pathway
MAPK Pathway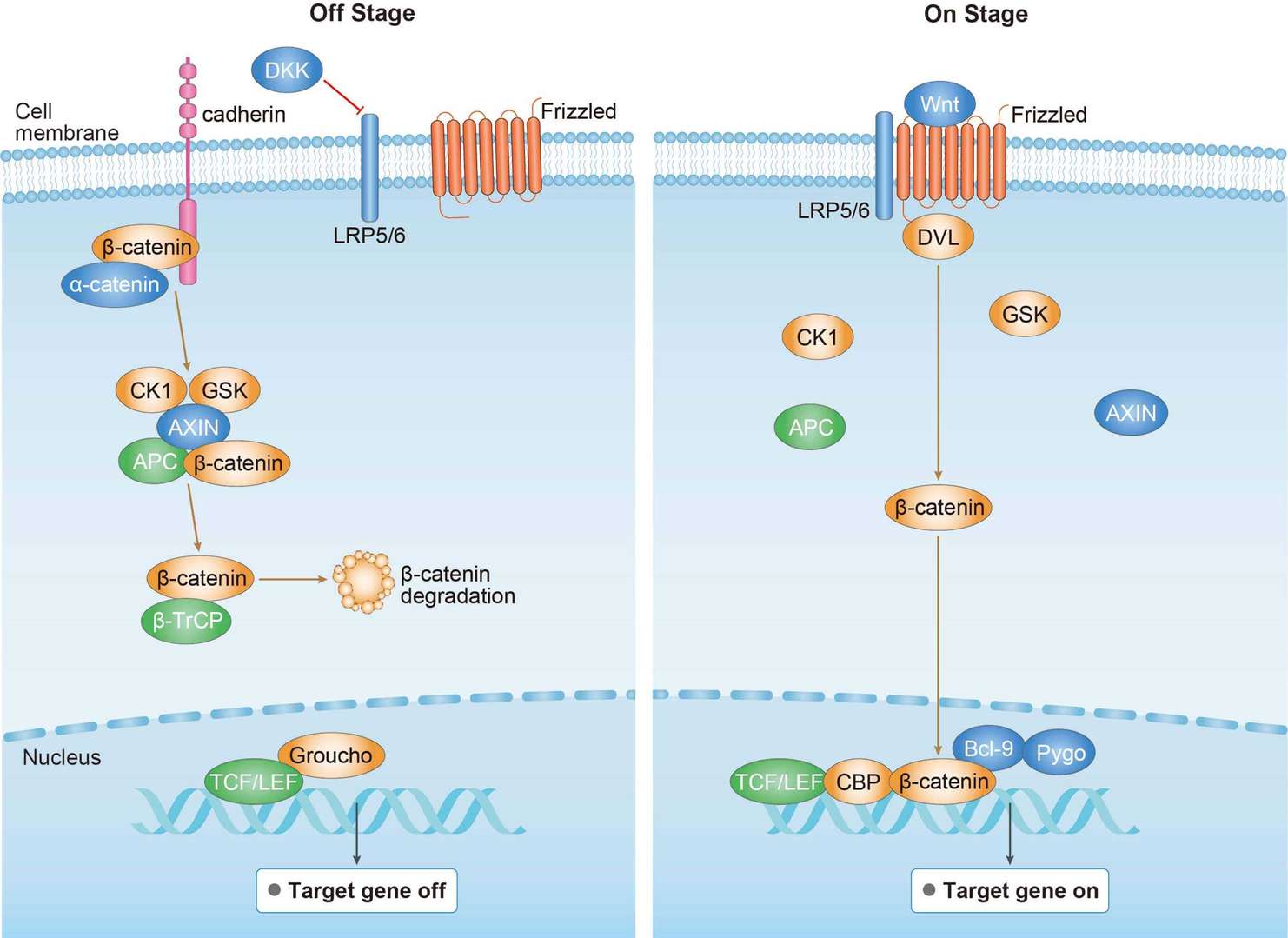 Wnt Pathway
Wnt Pathway