Colorectal Cancer Overview
About Colorectal Cancer
Colorectal cancer (CRC) is a common malignancy with an increasing incidence in many developed countries. It is the fourth most common cancer diagnosed in the United States, and is the second most deadly malignancy, after lung cancer. Symptoms can include a change in bowel habits, rectal bleeding or blood in stool and persistent abdominal discomfort. Many lifestyle-related factors have been linked to colorectal cancer. In fact, the links between diet, weight, and exercise and colorectal cancer risk are some of the strongest for any type of cancer. Dietary factors that increase the risk include red meat, processed meat, and alcohol. In addition, risk factors include colorectal polyps, inflammatory bowel disease, type 2 diabetes and night shift work. CRC arises from the colorectal epithelium as a result of the accumulation of genetic alterations in defined oncogenes and tumor suppressor genes (TSG). Adenocarcinomas make up about 96% of colorectal cancers. These cancers start in cells that make mucus to lubricate the inside of the colon and rectum. Other, much less common types of tumors can also start in the colon and rectum, such as carcinoid tumors, gastrointestinal stromal tumors (GISTs), lymphomas and sarcomas. Diagnosis of colorectal cancer is often based on sigmoidoscopy or colonoscopy. This is then followed by medical imaging to determine if the disease has spread. Screening is effective for preventing and decreasing deaths from colorectal cancer. Treatments used for colorectal cancer may include some combination of surgery, radiation therapy, chemotherapy and targeted therapy.
Main Signaling Pathways in Colorectal Cancer
Diagnosis of Colorectal Cancer
The early diagnosis of colorectal cancer is critical. If patients with CRC were diagnosed in the early stage, the 5-year survival rate could have been up to 90%. Unfortunately, more than 60% of CRC cases had already developed to an advanced stage by the time of detection, resulting in a survival rate around 8-9%. Currently, noninvasive monitoring tests, e.g. fecal occult blood test (FOBT) and tumor markers, including carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9), have been commonly used in clinical settings. However, unsatisfactory sensitivity and specificity have limited the clinical application in CRC diagnosis, prognosis and survival significantly. Although, the pre-operative endoscopic and radiological imaging has been used for CRC diagnosis, these invasive techniques suffer from poor patient compliance. Colorectal cancer diagnosis is performed by sampling of areas of the colon suspicious for possible tumor development, typically during colonoscopy or sigmoidoscopy, depending on the location of the lesion. Presence of metastases is determined by a CT scan of the chest, abdomen and pelvis. Other potential imaging tests such as PET and MRI may be used in certain cases. The microscopic cellular characteristics of the tumor are reported from the analysis of tissue taken from a biopsy or surgery. A pathology report contains a description of the microscopical characteristics of the tumor tissue, including both tumor cells and how the tumor invades into healthy tissues and finally if the tumor appears to be completely removed.
Targeted Therapy for Colorectal Cancer
In recent decades, significant improvements have been seen in the survival of patients with metastatic colorectal cancer (mCRC), which has been driven to a large extent by the approval of new drugs. Vascular endothelial growth factor (VEGF) is a protein that helps tumors form new blood vessels (a process known as angiogenesis) to get nutrients they need to grow. These Drugs include Bevacizumab, Ramucirumab and Ziv-aflibercept. When combined with chemo, these drugs can often help people with advanced colon or rectal cancers live longer. Epidermal growth factor receptor (EGFR) is a protein that helps cancer cells grow. There's often a lot of it on the surface of cancer cells. Drugs that target EGFR can be used to treat some advanced colon or rectal cancers. These include Cetuximab and Panitumumab. However, these drugs don't work in colorectal cancers that have mutations (defects) in the KRAS, NRAS or BRAF gene. Bevacizumab has shown clinical benefit with both irinotecan-based and oxaliplatin-based regimens. Moreover, the continuation of bevacizumab after failure of first-line bevacizumab-containing chemotherapy was found to contribute to prolonging the survival of patients with mCRC. Anti-EGFR antibody agents, cetuximab and panitumumab, demonstrated a survival benefit in mCRC patients.
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


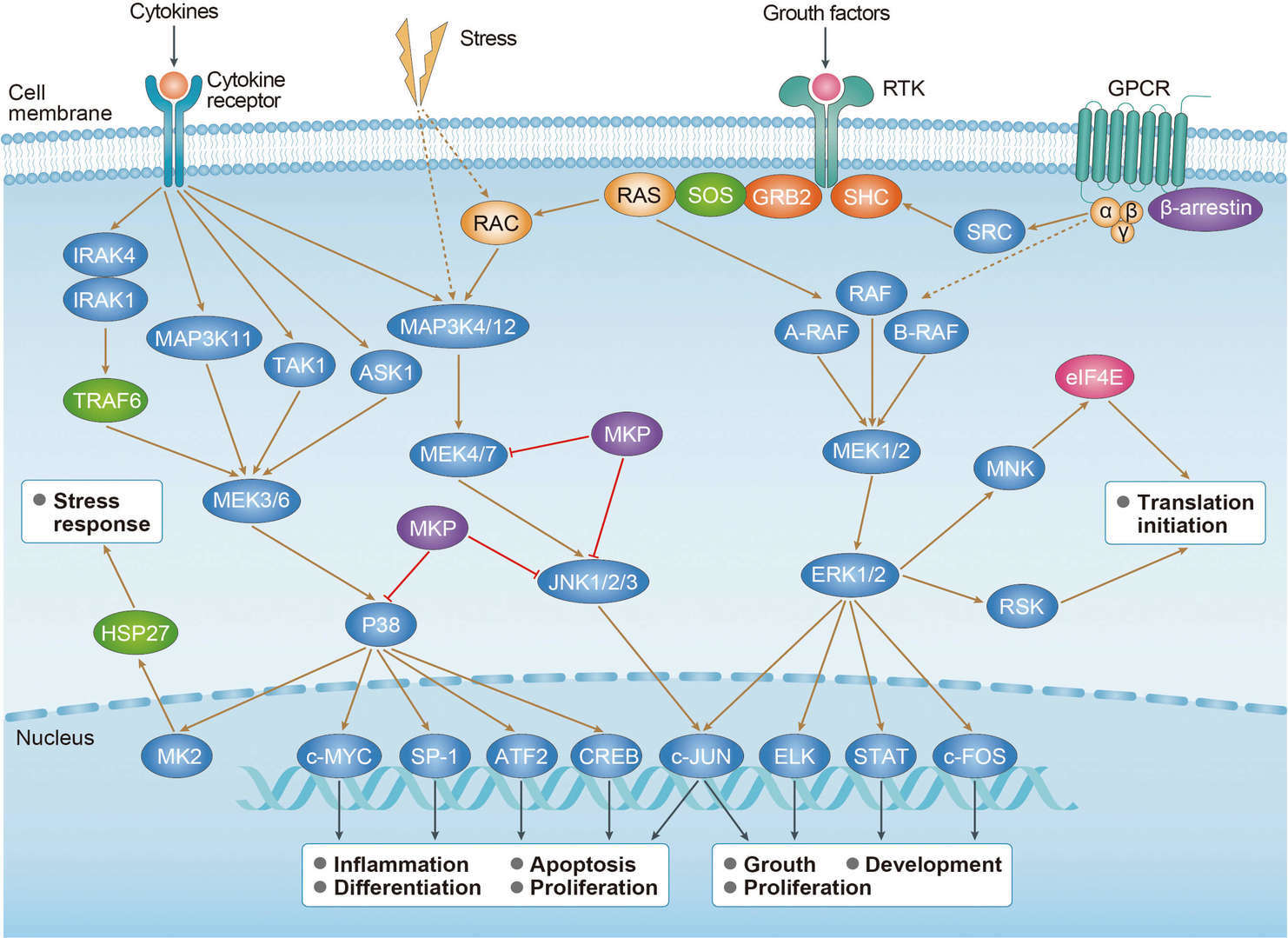 MAPK Pathway
MAPK Pathway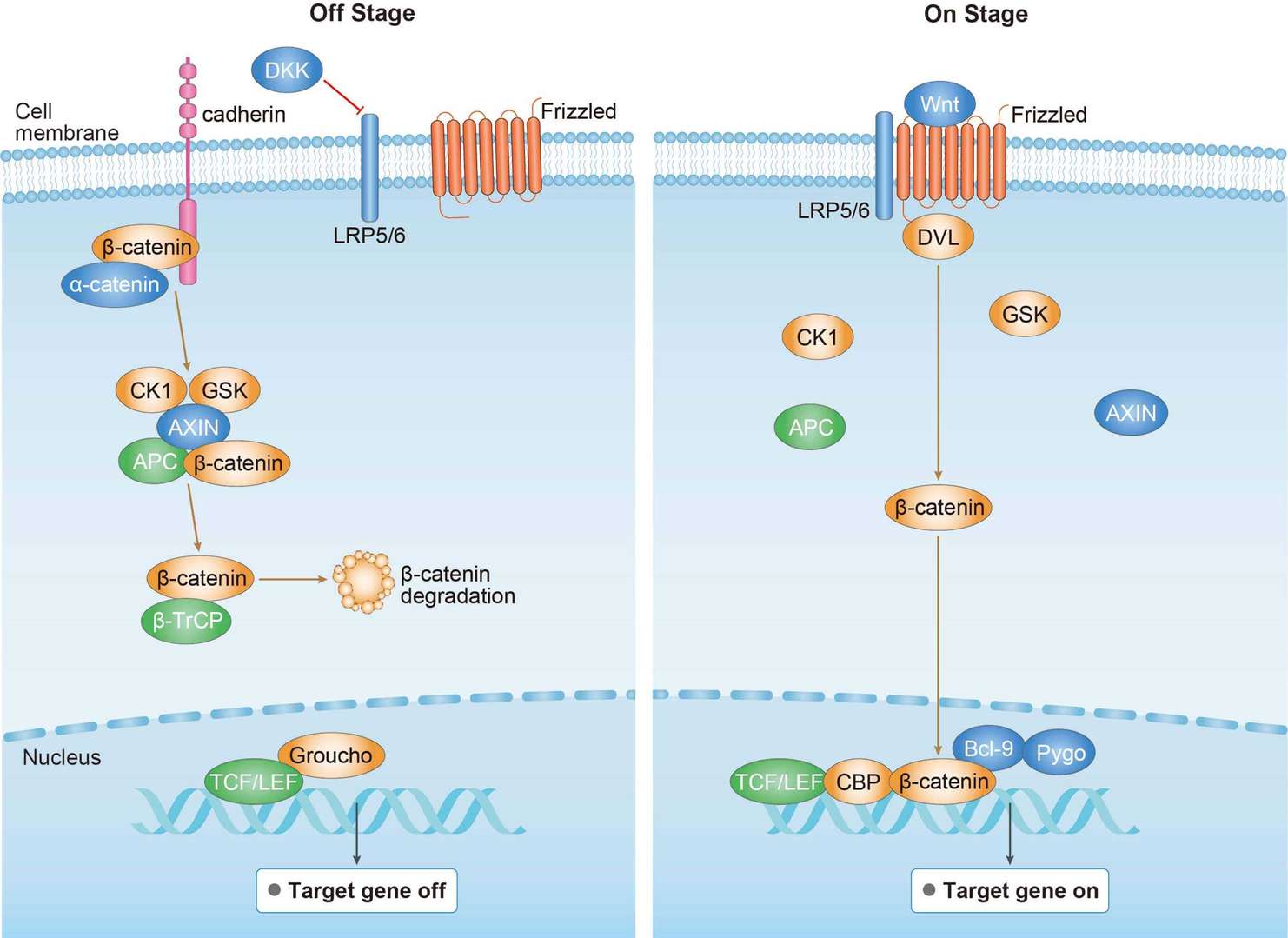 Wnt Pathway
Wnt Pathway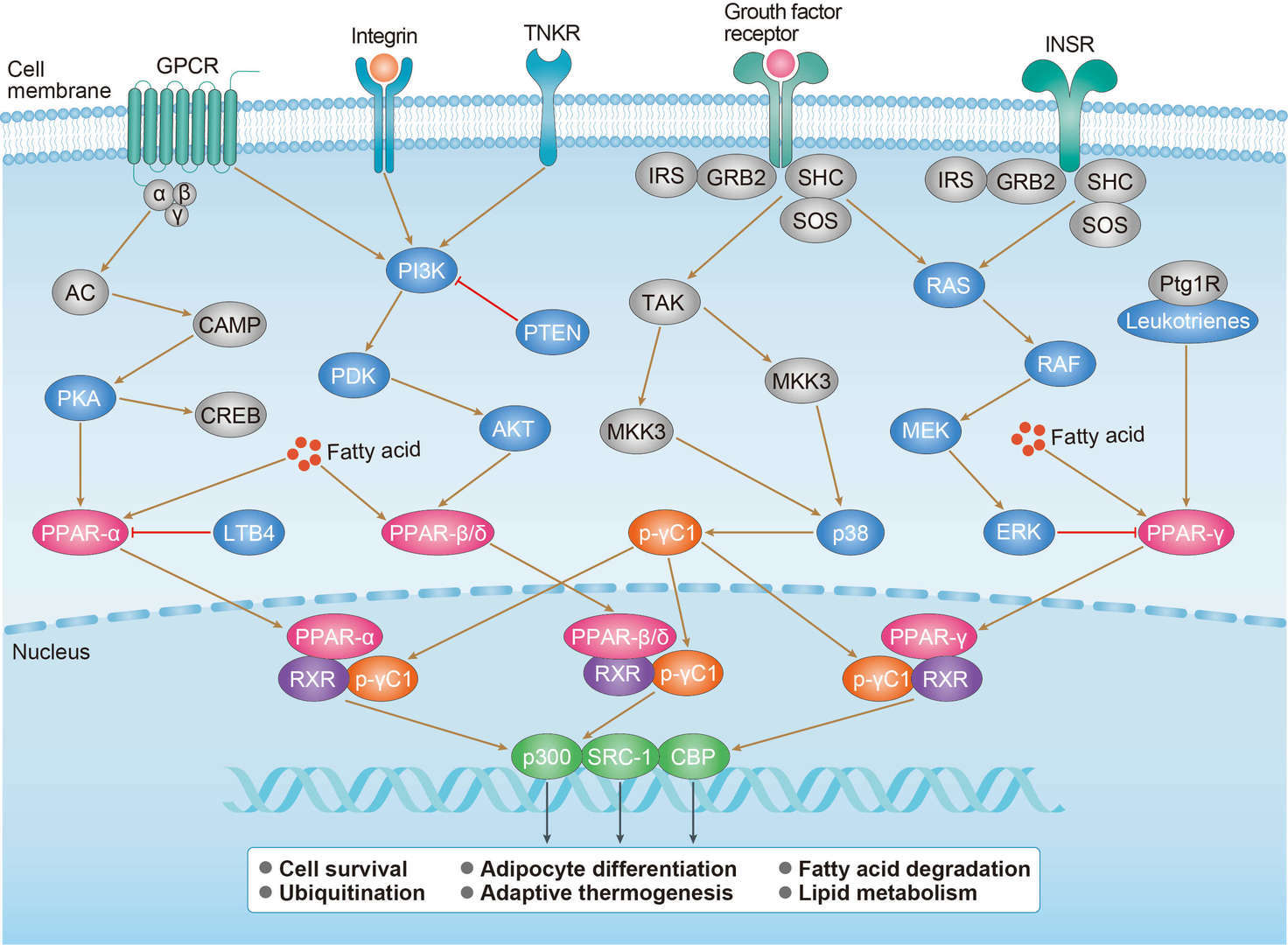 PPAR Pathway
PPAR Pathway