Non-small Cell Lung Cancer Overview
About Non-small Cell Lung Cancer
Non-small cell lung cancer (NSCLC) is a disease in which malignant (cancer) cells form in lung tissue, accounting for about 80% to 85% of lung cancer. It includes any type of epithelial lung cancer other than small cell lung carcinoma (SCLC), and the main subtypes of NSCLC are adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Other types like pleomorphic, carcinoid tumor, salivary gland carcinoma, and unclassified carcinoma are less common types. Non-small cell cancer has no special symptoms of disease, and the symptoms may be similar to or coincide with the symptoms of other diseases, which makes diagnosis more difficult. Some symptoms of early progression of the disease are feeling weak, being very tired, having trouble swallowing, swelling in the face or neck, and continuous or recurring infections like bronchitis or pneumonia. And some symptoms of less advanced cancer include chronic cough, coughing up blood, chest pain, hoarseness, shortness of breath, wheezing, chest pain, weight loss, and loss of appetite. Air pollution and smoking are the main risk factors for non-small cell lung cancer. The earlier you smoke, the higher the frequency of smoking, and the longer you smoke, the greater your risk of lung cancer. Others such as family genetic history and radioactive environments also are induced factors of non-small cell lung cancer. Early detection of non-small cell lung cancer is mainly by X-ray. If the degree of skepticism is high, then further biopsy and imaging tests are needed to determine and staging.
Overview of Non-small Cell Lung Cancer pathways
Main Signaling Pathways in Non-small Cell Lung Cancer
Diagnosis of Non-small Cell Lung Cancer
The diagnosis usually begins with the appearance of suspicious symptoms, so the results of physical exam and medical history can be helpful for the diagnosis of non-small cell lung cancer. Imaging and biopsy can be used to further locate and confirm the suspected cases. More than 75% of patients with non-small cell lung cancer have clinical manifestations such as cough, chest pain, haemoptysis, weight loss, dyspnoea, and other symptoms may also include venous congestion in the upper chest, shoulder pain together with the ulnar aspect of the arm and forearm. When the patient has the above suspected symptoms, further imaging examination including chest x-rays and CT scan are needed. Clinical data show that the detection rate of CT scan is better than that of x-ray, so patients with suspected lung tumors must undergo a contrast-enhanced CT scanning of the chest and upper abdomen. In addition, microscopic examination of sputum samples, that is sputum cytology, is also one of the means of detection and diagnosis of lung cancer. Biopsy is usually an effective method for the diagnosis of lung cancer. The methods of biopsy include fine-needle aspiration (FNA) biopsy, bronchoscopy, thoracoscopy, mediastinoscopy, anterior mediastinoscopy and lymph node biopsy. The advantage of microscopic examination is that tissue specimens can be obtained at the same time of biopsy. Research on markers of non-small cell lung cancer (NSCLC) has been under way. The 2011 Lancet Seminar on non-small-cell lung cancer mentioned that thyroid transcription factor 1, cytokeratin 7, and mucin staining are recommended markers for adenocarcinoma, and P63 and cytokeratin 5/6 for squamous-cell carcinoma. This conclusion is supported by clinical data and is suitable for the detection of cytological samples in tissue biopsies.
Targeted Therapy for Non-small Cell Lung Cancer
The main targeted therapies for non-small cell lung cancer are tyrosine kinase inhibitors and monoclonal antibodies. Gefitinib and Erlotinib belong to the first representative of epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI). Erlotinib has been approved for the use of as a first-line treatment in patients with allergic EGFR mutations. It has been found that compared with conventional chemotherapy. It has been found that compared with conventional chemotherapy, Erlotinib can prolong the progression-free survival time of EGFR mutation positive patients with advanced NSCLC and is well tolerated. In 2004, Erlotinib was approved for the treatment of locally advanced or metastatic NSCLC patients after progress on at least one previous chemotherapy regimen. As the second generation irreversible EGFR, human epidermal growth factor receptor 2 (HER2) and HER4 inhibitor, Afatinib is mainly used to treat NSCLC patients with EGFR gene exon 19 deletion or exon 21 (L858R) mutation. And Afatinib was approved for first-line treatment of metastatic NSCLC patients with sensitive EGFR mutations. The third generation of EGFR-TKI, AZD9291 has been proved to be able to overcome the T790 resistant mutation. In 2015, AZD9291 was officially approved to be listed on the market. Another class of drugs for the treatment of non-small cell lung cancer is ALK/ROS1 inhibitors. Currently, ALK inhibitors approved for clinical use include Crizotinib, Ceritinib and Alectinib. As a competitive inhibitor of ALK, Crizotinib can inhibit tumor cell proliferation and accelerate apoptosis by selectively competing adenosine triphosphate (ATP) to block kinase protein. It has been approved for use in locally advanced or metastatic NSCLC patients with positive ALK gene rearrangement. As the second generation ALK inhibitor, Ceritinib has a certain efficacy in the treatment of patients with ALK fusion gene positive and Crizotinib acquired drug resistance. Alectinib is a highly selective ALK inhibitor, which is effective in the treatment of NSCLC patients with Crizotinib resistance and central nervous system metastasis. In addition, monoclonal antibodies as vascular endothelial growth factor (VEGF) inhibitor (Bevacizumab, Ramucirumab) and Epidermal growth factor receptor (EGFR) inhibitor (Cetuximab, Necitumumab) also used in the treatment of non-small cell lung canc
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


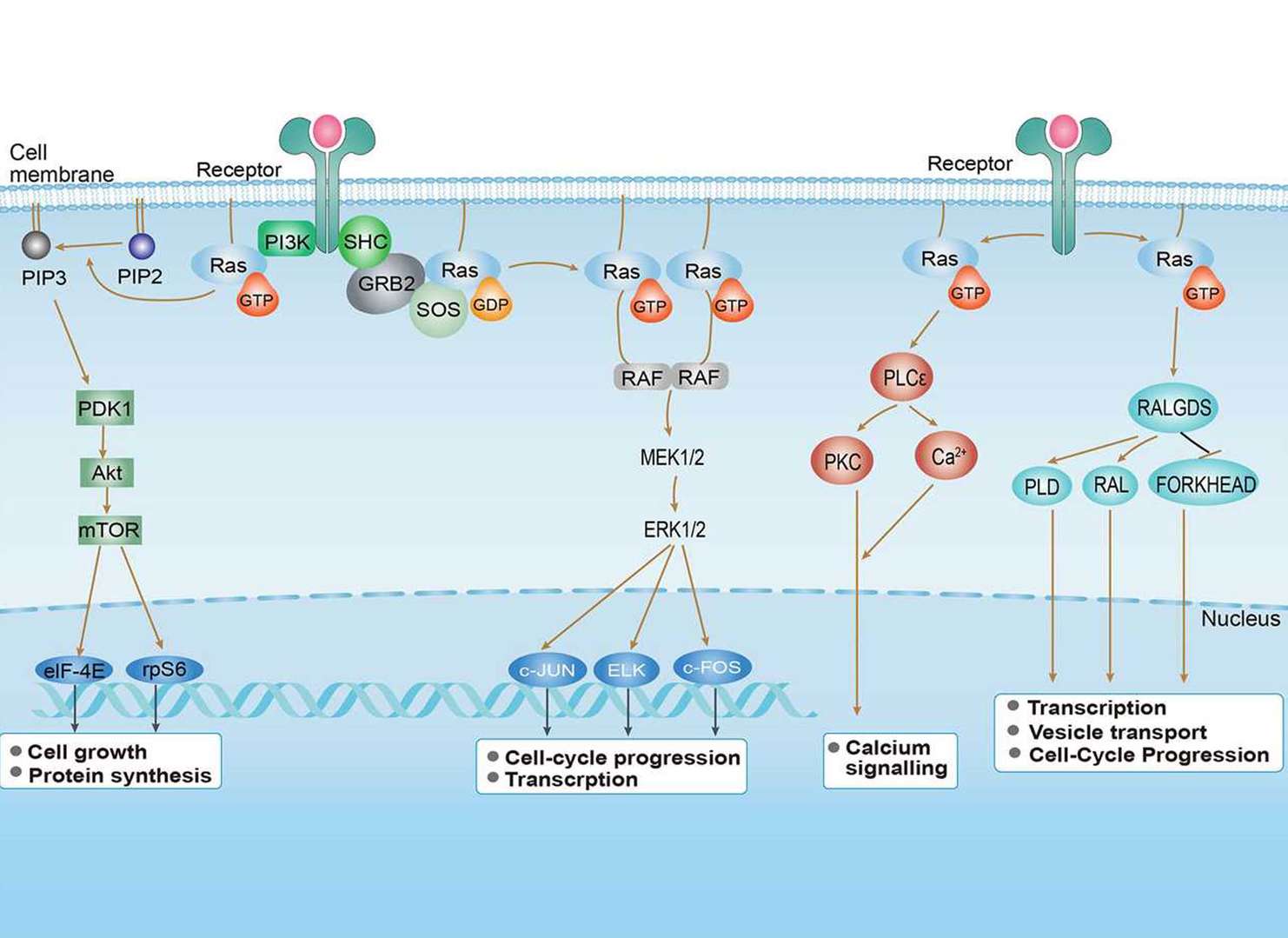 Ras Signaling Pathway
Ras Signaling Pathway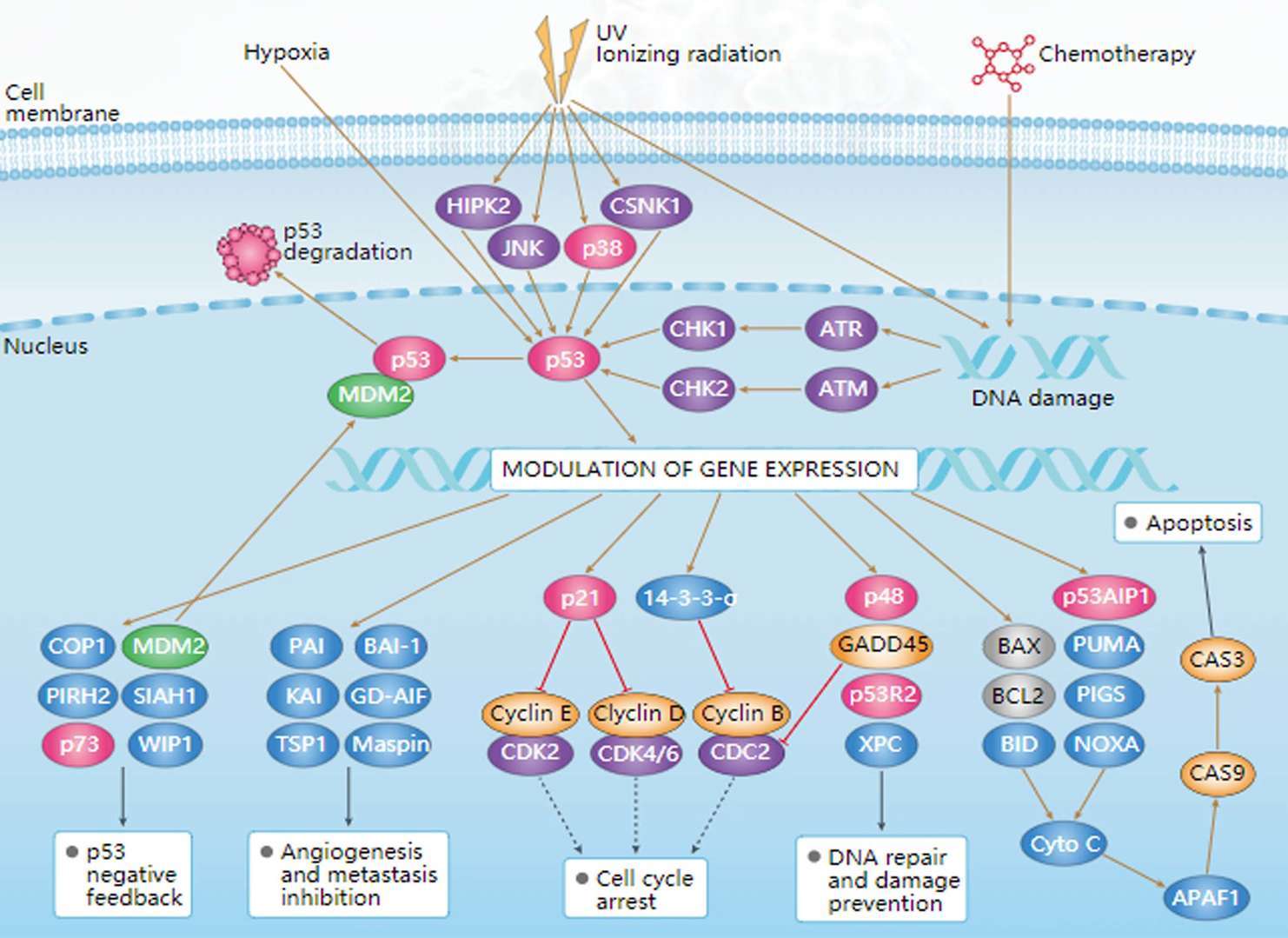 p53 Signaling Pathway
p53 Signaling Pathway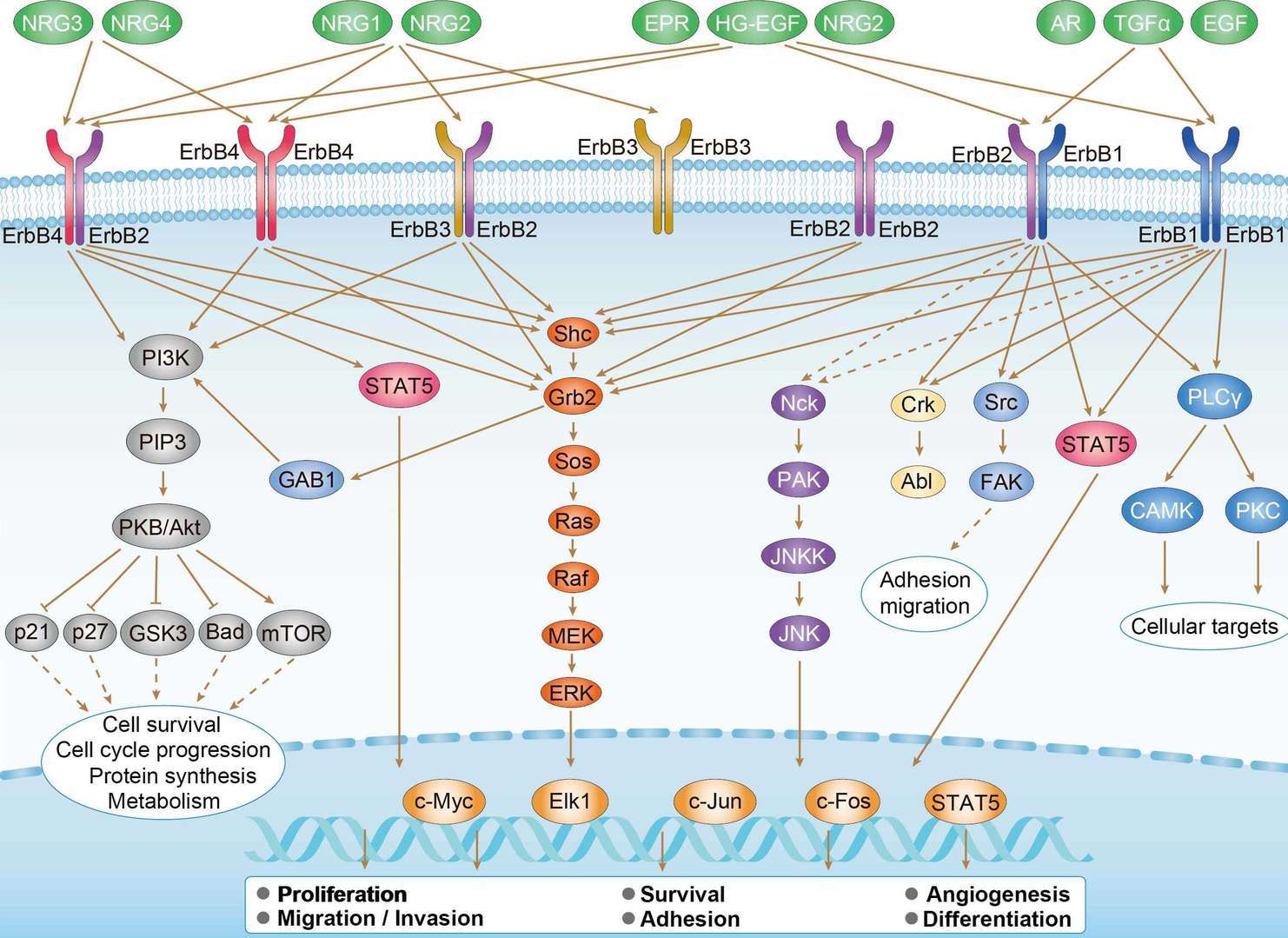 ErbB Signaling Pathway
ErbB Signaling Pathway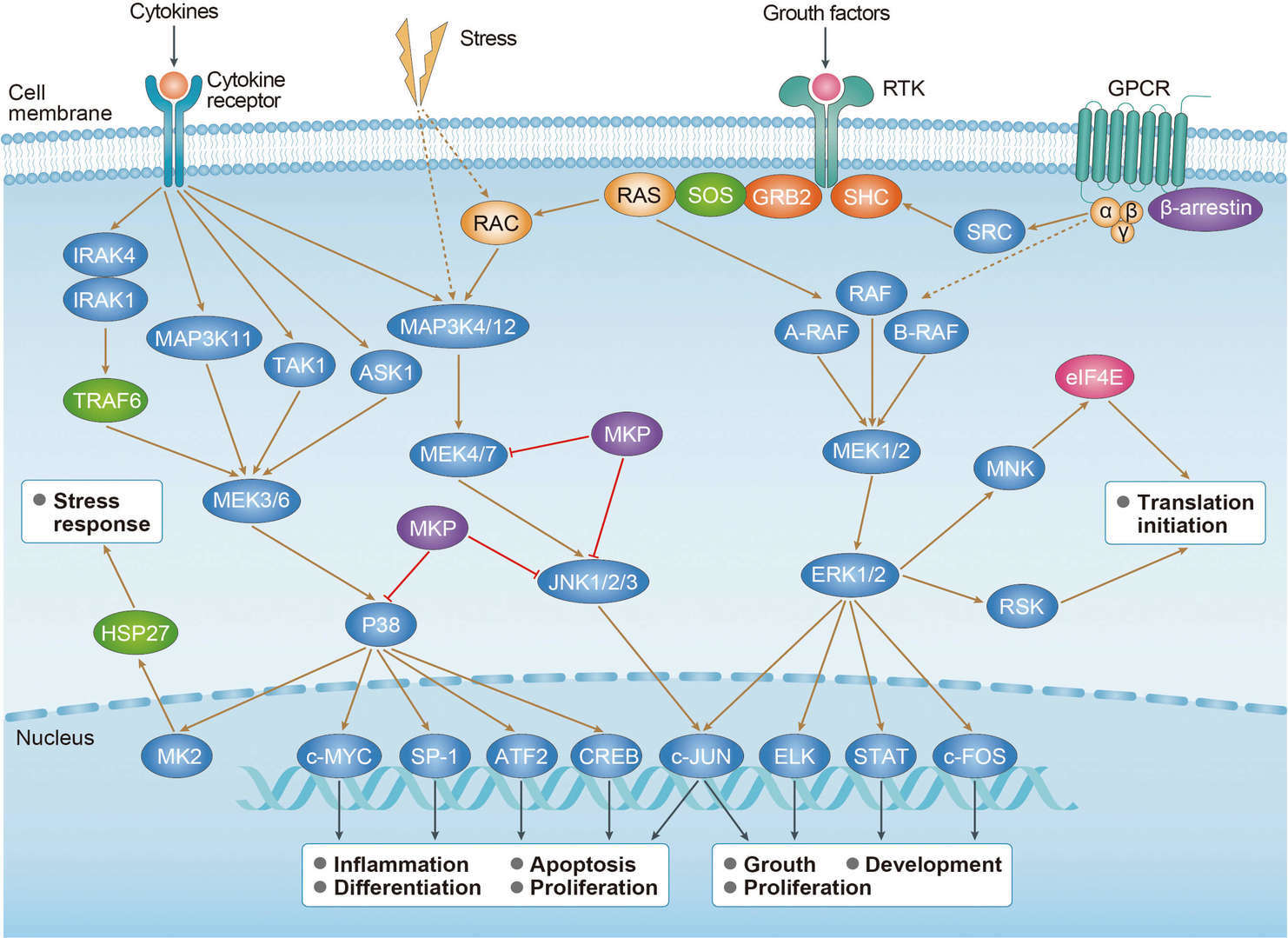 MAPK Signaling Pathway
MAPK Signaling Pathway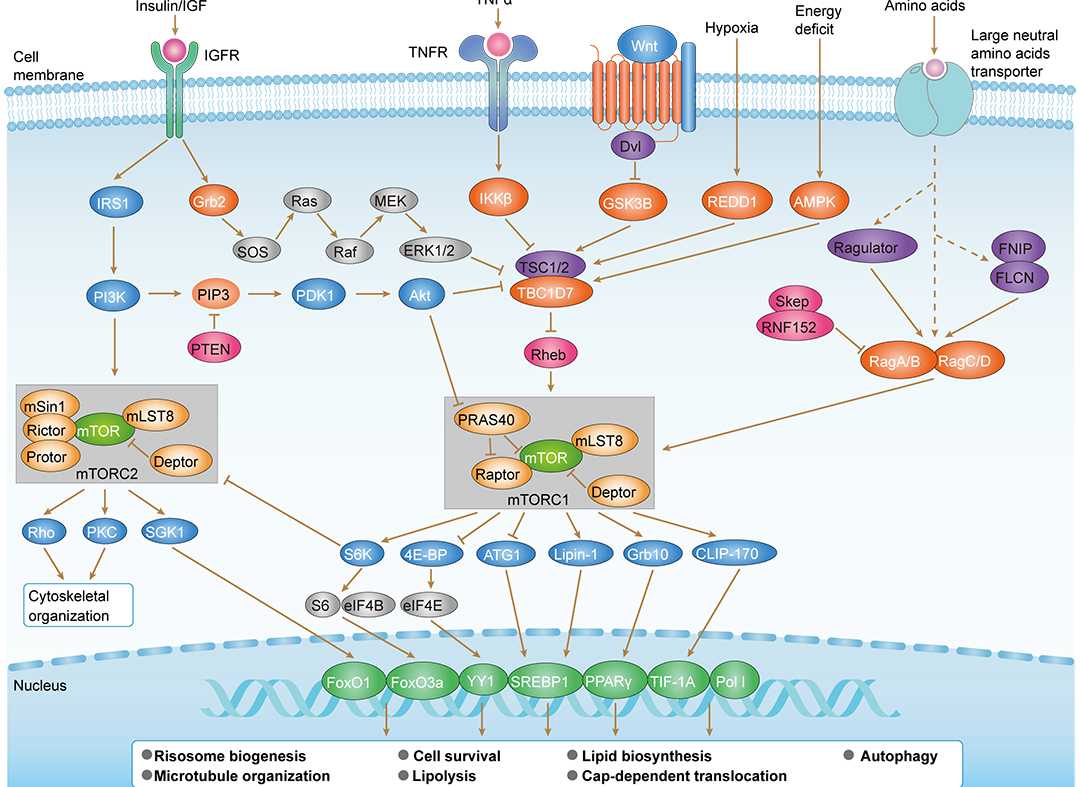 mTOR Signaling Pathway
mTOR Signaling Pathway