Monalizumab Overview
Introduction of Monalizumab
Monalizumab (previously IPH2201) is a humanized Immunoglobulin G4 (IgG4) monoclonal antibody targeted at NK cell receptor NKG2A, which is expressed on tumor infiltrating cytotoxic NK and CD8+T lymphocytes. NIG2A is a member of C-type lectin receptors family, which are widespread, particularly in immune system cells, and are generally thought to recognize carbohydrate structures. The biological function of NIG2A is to serve as an immune checkpoint inhibitory of signal transduction, repressing the cytotoxic activity of the NK cells. The natural ligand of NIG2A is HLA class I histocompatibility antigen, alpha chain E (HLA-E), which is frequently overexpressed in tumor diseases, transplants and virus-infected. The drug has been used in trials studying the treatment of diseases including gynecologic cancer, chronic lymphocytic leukemia, and squamous cell carcinoma of the oral cavity.
Mechanism of Action of Monalizumab
Monalizumab is a monoclonal antibody developed to bind to NKG2A receptors expressed on T and NK cells, which could reestablish a broad anti-tumor response. NKG2A can be specifically recognized by HLA-E that is expressed on many solid and hematologic tumors. Binding of HLA-E to NKG2A leads to the inhibition of activation and cytotoxic activity of T and NK cells. Thus, tumor cells can protect themselves from lysis by expressing HLA-E. HLA-E has been found to highly express in many cancer tumors such as head and neck, ovary, endometrium, colon, cervix, lung, melanoma, esophageal and various types of leukemia and lymphoma cancers. Expression of HLA-E has become the important immune escape strategy of tumor cells. The antibody drug monalizumab is aimed at repressing the binding of NKG2A to HLA-E. By blocking the interactions between tumor-associated HLA-E and the NKG2A receptor, monalizumab could active T and NK cells and their cytotoxic cell responses, enhancing the immune function of the body and re-establishing the immune system to remove tumor cells.
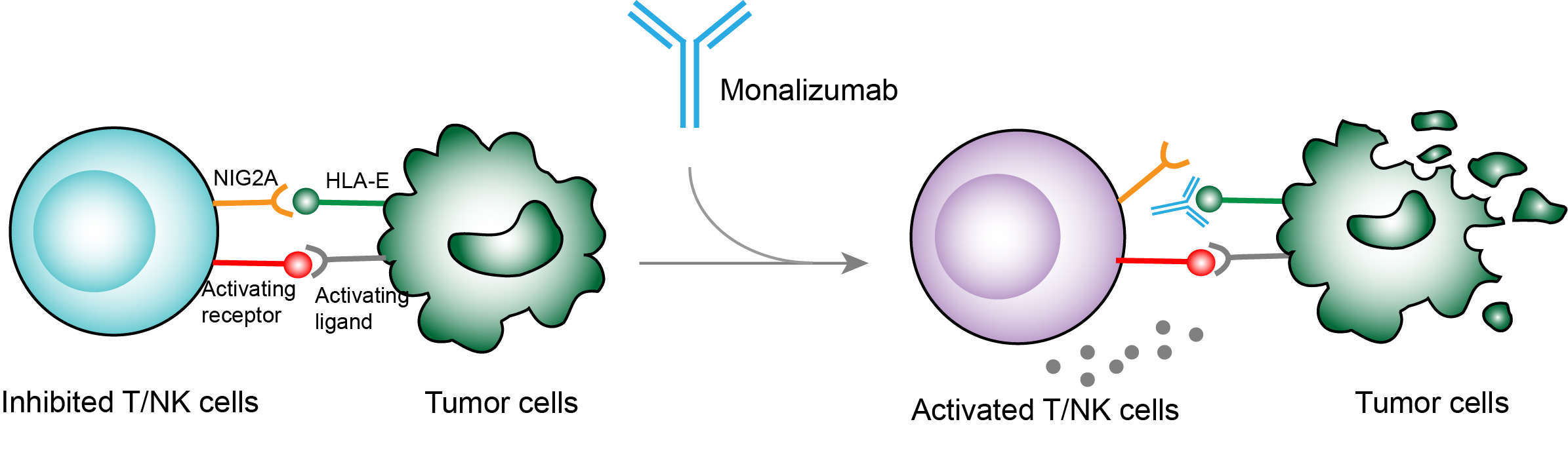 Fig.1 Mechanism of action of Monalizumab
Fig.1 Mechanism of action of Monalizumab
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

