TGF-β Signaling Pathway
About TGF-β Pathway
Transforming growth factor beta (TGF-β) is a cytokine that participates in both physiological and pathological processes. During tumor progression, TGF-β signaling regulates immune/inflammatory response and tumor microenvironment. The TGF-β superfamily consists of at least 40 structurally and functionally related cytokines. TGF-β family proteins are classified into several subtypes, including TGF-βs, activins/inhibins, and bone morphogenetic proteins (BMPs)/growth differentiation factors according to structural characteristics. TGF-β signals through TGF-β receptors (TGF-βRs, TβRs) I and II to activate downstream signaling pathways. The TGF-βRs are single-pass transmembrane proteins with serine/threonine kinase activity. In the absence of ligand, TGF-βRI and TGF-βRII exist as monomers, homodimers, or heterodimers on the cell surface. Ligand binding promotes formation of a tetrameric complex between TGF-βRII dimers and two TGF-βRIs. TGF-βRIs and TGF-βRIIs have N-terminal extracellular ligand binding domains, transmembrane segments, and C-terminal cytosolicserine/threonine kinase domains. TGF-β binds specifically to the constitutively active TGF-βRII, which activates TGF-βRI by phosphorylating the glycine/serine-rich domain. Activated TGF-βRI then phosphorylates downstream effectors to induce signal transduction. TGF-βR activity is regulated by beta glycan, a type III TGF-βR, and endoglin.
Canonical TGF-β signaling is dependent upon Smad family proteins. Active TGF-βI at the cell surface phosphorylates receptor-activated Smads (R-Smads). There are two sub-classes of R-Smads in which Smad2 and Smad3 mediate the TGF-β/activin pathway. Smad4 acts as a co-factor that binds to activated R-Smads to form a complex that translocates to the nucleus and regulates transcription. TβRs can also activate non-Smad-dependent signaling pathways, including the mitogen-activated protein kinase (MAPK) pathway mediated by p38, c-Jun amino terminal kinase (JNK), extracellular signal-regulated kinases (ERK), nuclear factor-κB (NF-κB), Rho, and phosphatidylinositol 3-kinase (PI3K)-Akt. TGF-β pathway is a very complex signaling network, and its role in cellular homeostasis varies with different genetic profiles of cancer cells. On the one hand, TGF-β signaling can induce cell differentiation and act as a tumor suppressor in non-malignant tumor, on the other hand, it is also capable of promoting angiogenesis and EMT in cancer cells. Once signal transduction is initiated, TGF-β is involved in cell differentiation and can also act as a tumor suppressor in cell cycle control. Furthermore, TGF-β induces p53-independent expression of p21 and inhibits oncogene expression, while TGF-β receptors also cross-talk with oncogenic signaling pathways. TGF-β pathway has dual anti- and pro-tumoral roles at the cancer cell level, depending on tumor stage and genetic alteration background, with mechanistic differences between cancer models.
Associated Diseases
Related Pathways
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


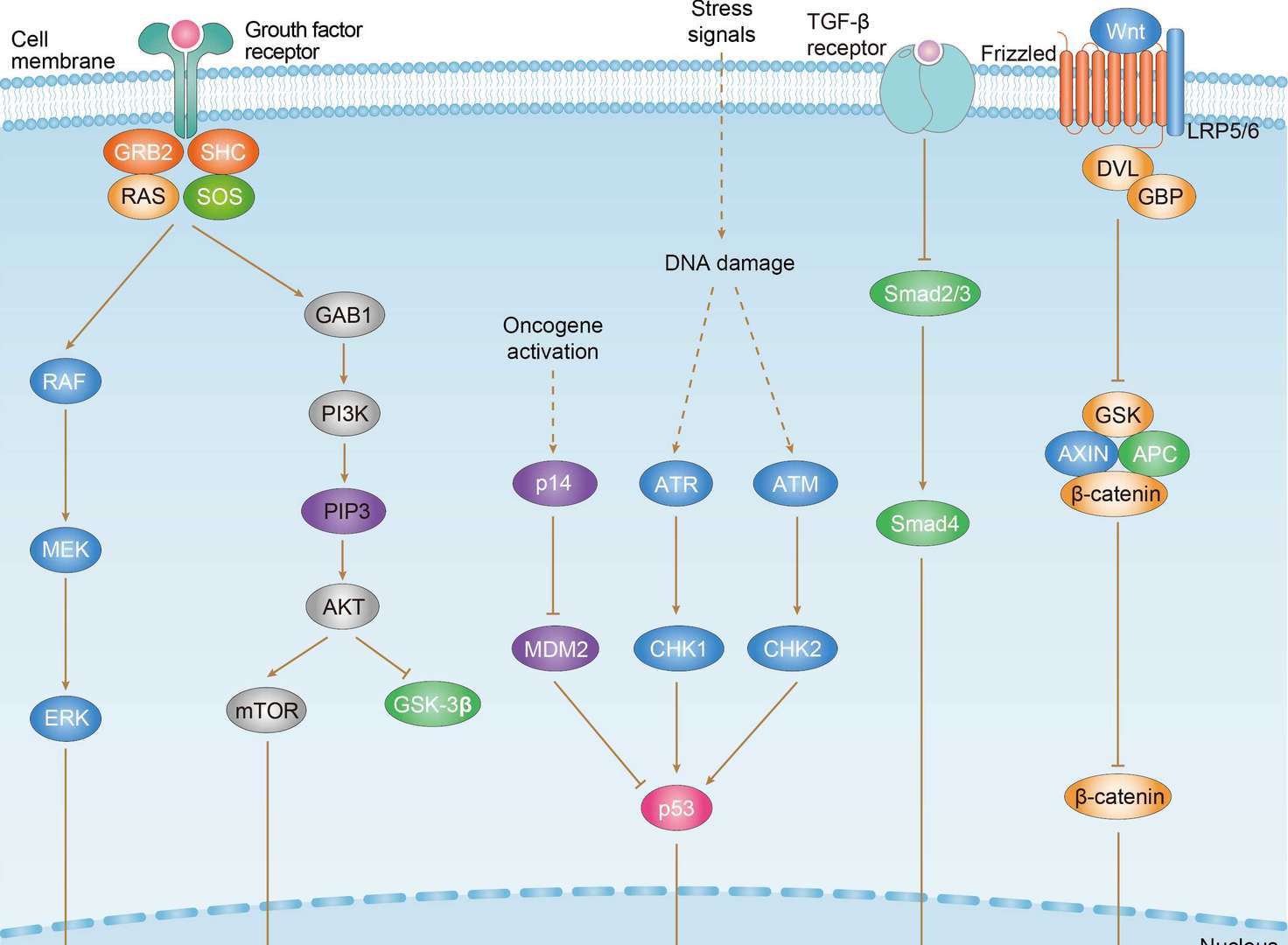 Gastric Cancer
Gastric Cancer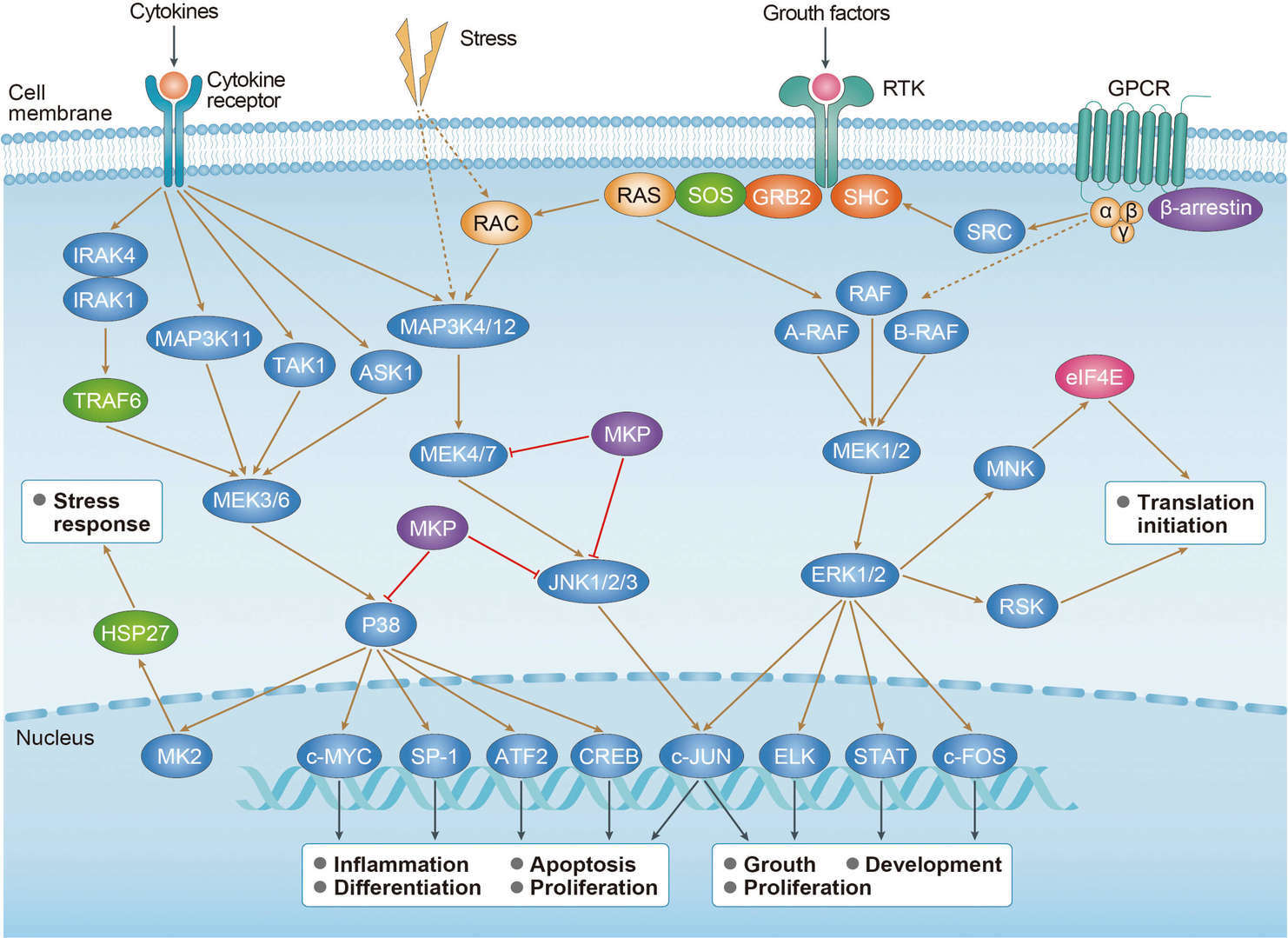 MAPK Pathway
MAPK Pathway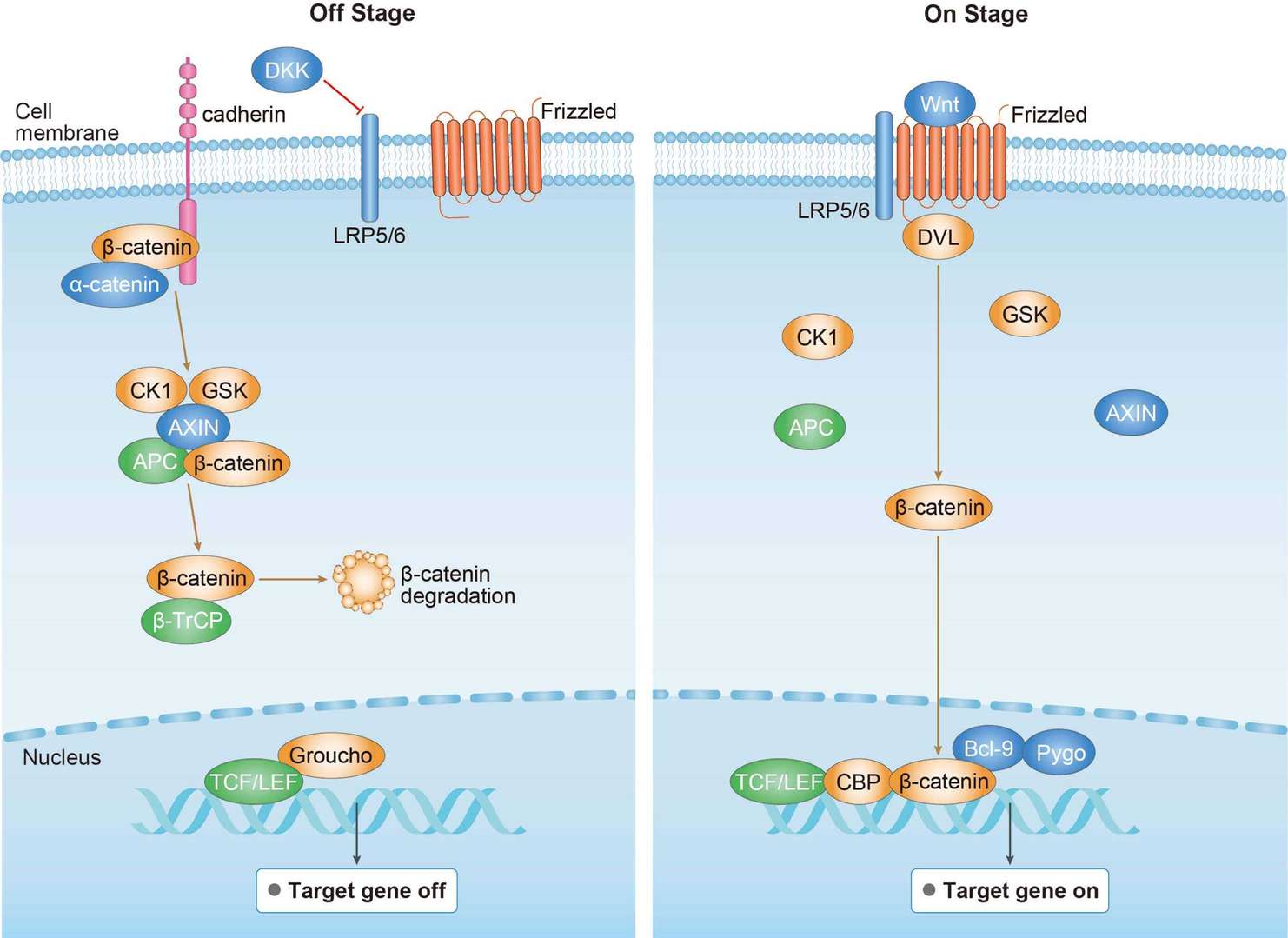 Wnt Pathway
Wnt Pathway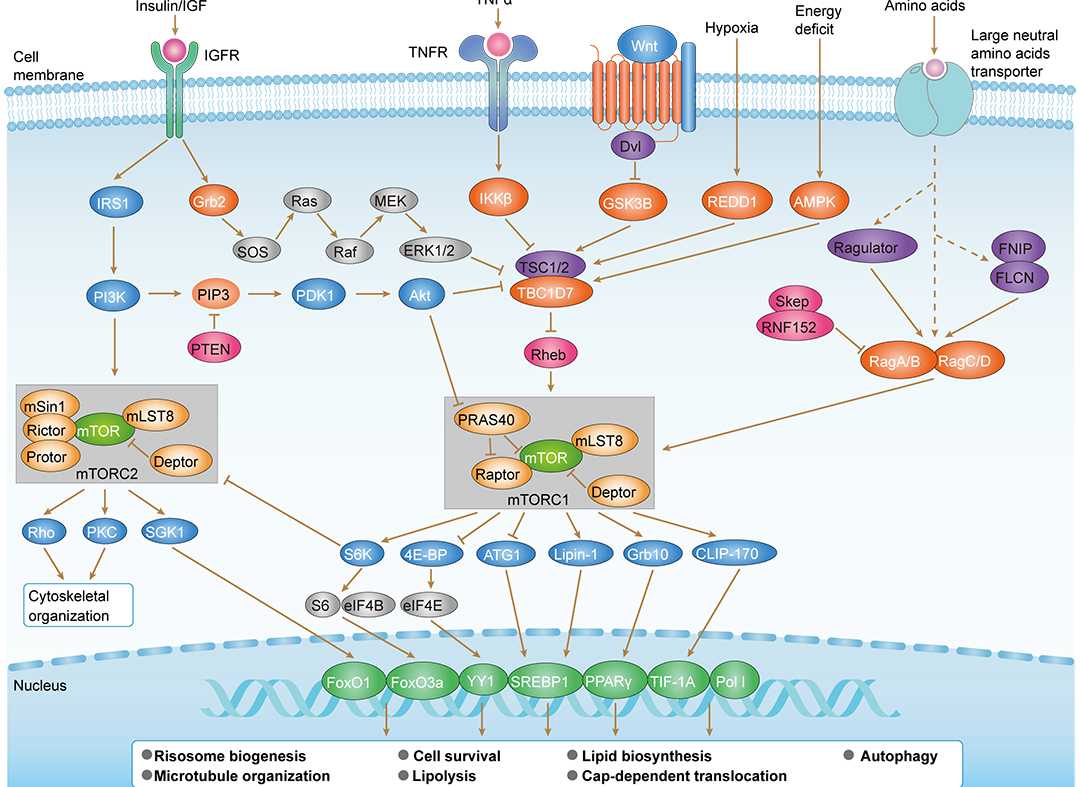 mTOR Pathway
mTOR Pathway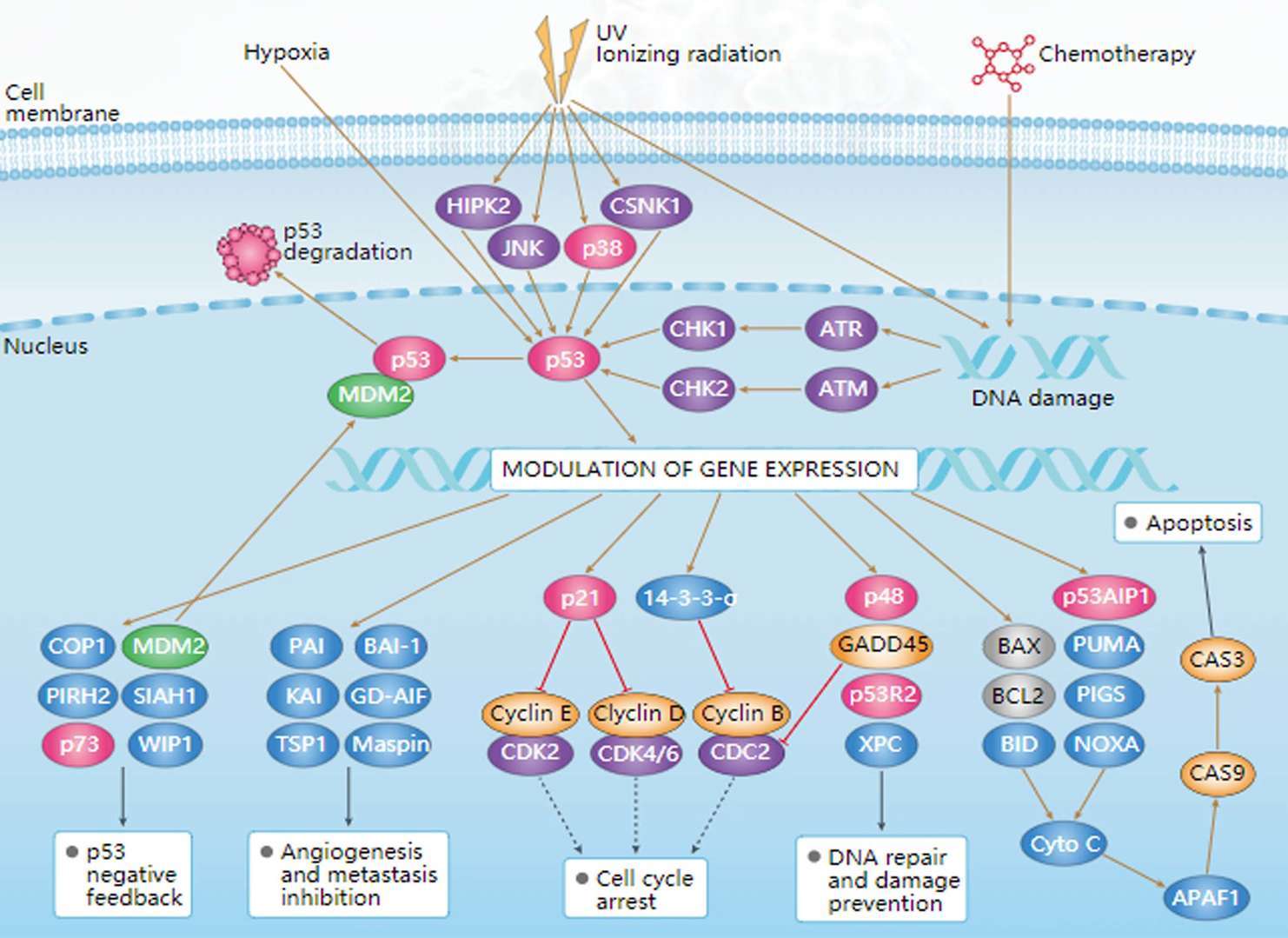 p53 Pathway
p53 Pathway