TLR Signaling Pathway
Innate immune receptors, also known as pattern recognition receptors (PRRs), have been identified in the serum, on the cell surface, in endosomes, and in the cytoplasm. Toll-like receptors (TLRs) is one of the particularly important groups of PRRs. TLR paralogs are located on cell surfaces or within endosomes and participate in the first line of defense against invading pathogens and play a crucial role in inflammation, immune cell regulation, survival, and proliferation.
TLR Family and Subfamilies
The TLR family has 10 members (TLR1–TLR10) in humans and 12 (TLR1–TLR9, TLR11–TLR13) in the mouse. In humans, 10 TLRs respond to a variety of pathogen-associated molecular patterns (PAMPs), including lipopolysaccharide (TLR4), lipopeptides (TLR2 associated with TLR1 or TLR6), bacterial flagellin (TLR5), viral dsRNA (TLR3), viral or bacterial ssRNA (TLRs 7 and 8), and CpG-rich unmethylated DNA (TLR9), among others. TLRs can be classified into two subfamilies in terms of their localization, cell surface TLRs and intracellular TLRs. All TLRs are listed in Table 1. Cell surface TLRs can recognize microbial membrane components, such as lipids, lipoproteins, and proteins. Intracellular TLRs are localized in the endosome and can recognize nucleic acids derived from bacteria and viruses, and also recognize self-nucleic acids in disease conditions such as autoimmunity.
Table.1 Summary of Toll-like receptors (TLRs)
| Location | Toll-like receptors (TLRs) | Functions |
| Cell surface TLRS | TLR1 | TLR2 along with TLR1 or TLR6 recognizes a wide variety of PAMPs including lipoproteins, peptidoglycans, lipotechoic acids, zymosan, mannan, and tGPI-mucin |
| TLR2 | ||
| TLR6 | ||
| TLR4 | Recognizes bacterial lipopolysaccharide (LPS) | |
| TLR5 | Recognizes bacterial flagellin | |
| TLR10 |
|
|
| Intracellular TLRs | TLR3 | Recognizes viral double-stranded RNA (dsRNA), small interfering RNAs, and self-RNAs derived from damaged cells |
| TLR7 | TLR7 is predominantly expressed in plasmacytoid DCs (pDCs) and recognizes single-stranded (ss)RNA from viruses. It also recognizes RNA from streptococcus B bacteria in conventional DCs (cDCs). | |
| TLR8 | Human TLR8 responds to viral and bacterial RNA | |
| TLR9 | Recognizes bacterial and viral DNA that is rich in unmethylated CpG-DNA motifs and hemozoin | |
| TLR11 | TLR11 is localized in the endolysosome and recognizes flagellin or an unknown proteinaceous component of uropathogenic Escherichia coli (UPEC) as well as a profilin-like molecule derived from Toxoplasma gondii | |
| TLR12 |
TLR12 is predominantly expressed in myeloid cells and is highly similar to TLR11 and recognizes profilin from T. gondii; Functions either as a homodimer or a heterodimer with TLR11 |
|
| TLR13 | Recognizes bacterial 23S rRNA (16–18) and unknown components of vesicular stomatitis virus. |
The Structure of TLRs
TLRs are type I integral membrane receptors, each with an N-terminal ligand recognition domain, a single transmembrane helix, and a C-terminal cytoplasmic signaling domain. Furthermore, each TLR is composed of an ectodomain with leucine-rich repeats (LRRs) that are short motifs that fold into a characteristic solenoid structure and mediate PAMPs recognition, a transmembrane domain, and a cytoplasmic Toll/IL-1 receptor (TIR) domain that initiates downstream signaling.
TLR Signaling Pathway
The activation of the TLR signaling pathway starts from the cytoplasmic TIR domain that associates with a TIR domain-containing adaptor, MyD88. Upon stimulation with ligands, MyD88 recruits IL-1 receptor-associated kinase-4 (IRAK-4) to TLRs through the interaction of the death domains of both molecules. IRAK-1 is activated via phosphorylation and associates with TRAF6, leading to activate the IKK complex and leading to activation of MAP kinases, including JNK, p38, MAPK, and NF-κB. TOLLIP and IRAK-M interact with IRAK-1 and negatively regulate the TLR-mediated signaling pathways. In addition, there are other modes of regulation for these pathways include TRIF-dependent induction of TRAF6 signaling by RIP1 and negative regulation of TIRAP-mediated downstream signaling by ST2L, TRIAD3A, and SOCS1. Moreover, activation of MyD88-independent pathways occurs via TRIF and TRAF3, resulting in recruitment of IKKε/TBK1, phosphorylation of IRF3, and expression of interferon-β. TIR domain containing adaptors such as TIRAP, TRIF, and TRAM regulates TLR-mediated signaling pathways by providing specificity for individual TLR signaling cascades. TRAF3 plays a critical role in the regulation of both MyD88-dependent and TRIF-dependent signaling via TRAF3 degradation, which activates MyD88-dependent signaling and suppresses TRIF-dependent signaling (and vice versa).
Related Pathways
For research use only. Not intended for any clinical use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.


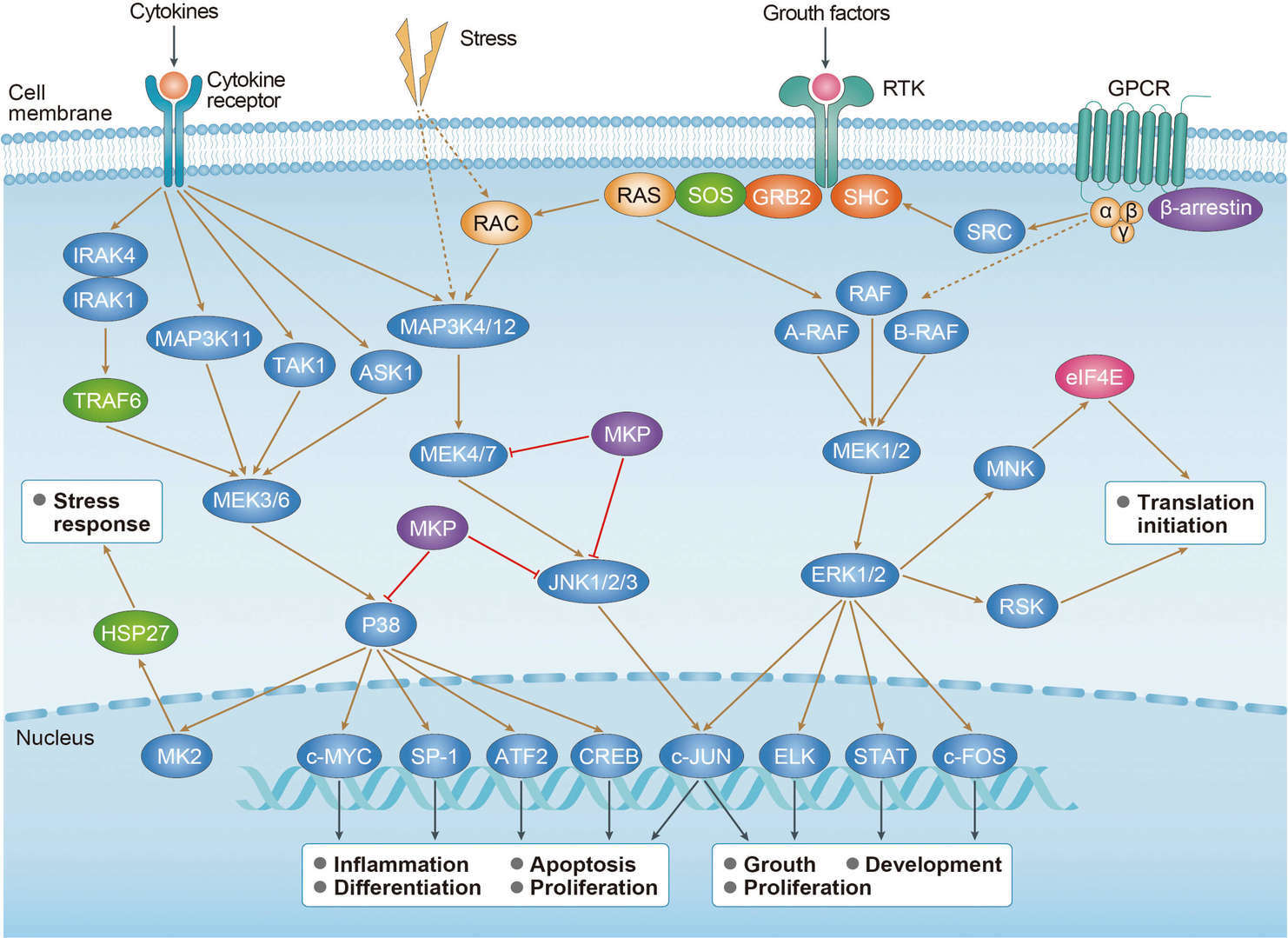 MAPK Signaling Pathway
MAPK Signaling Pathway